Abstract
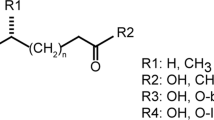
The ability to sense prey-derived cues is essential for predatory lifestyles. Under low-nutrient conditions, Arthrobotrys oligospora and other nematode-trapping fungi develop dedicated structures for nematode capture when exposed to nematode-derived cues, including a conserved family of pheromones, the ascarosides. A. oligospora senses ascarosides via conserved MAPK and cAMP–PKA pathways; however, the upstream receptors remain unknown. Here, using genomic, transcriptomic and functional analyses, we identified two families of G protein-coupled receptors (GPCRs) involved in sensing distinct nematode-derived cues. GPCRs homologous to yeast glucose receptors are required for ascaroside sensing, whereas Pth11-like GPCRs contribute to ascaroside-independent nematode sensing. Both GPCR classes activate conserved cAMP–PKA signalling to trigger trap development. This work demonstrates that predatory fungi use multiple GPCRs to sense several distinct nematode-derived cues for prey recognition and to enable a switch to a predatory lifestyle. Identification of these receptors reveals the molecular mechanisms of cross-kingdom communication via conserved pheromones also sensed by plants and animals.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Data availability
The A. oligospora TWF154 genome database used in this study is from National Center for Biotechnology Information GenBank under the accession number SOZJ00000000. RNA sequencing data used in this study are from the National Center for Biotechnology Information Gene Expression Omnibus database under the accession number GSE233568. References to these accession numbers can also be found throughout this paper. Source data are provided with this paper.
References
Brown, N. A., Schrevens, S., Van Dijck, P. & Goldman, G. H. Fungal G-protein-coupled receptors: mediators of pathogenesis and targets for disease control. Nat. Microbiol. 3, 402–414 (2018).
van Dijck, P. et al. Nutrient sensing at the plasma membrane of fungal cells. Microbiol. Spectr. 5, 2 (2017).
Martín, J., Van Den Berg, M., van Themaat, E. V. L. & Liras, P. Sensing and transduction of nutritional and chemical signals in filamentous fungi: impact on cell development and secondary metabolites biosynthesis. Biotechnol. Adv. 37, 1–15 (2019).
Jiang, C. et al. An expanded subfamily of G-protein-coupled receptor genes in Fusarium graminearum required for wheat infection. Nat. Microbiol. 4, 1582–1591 (2019).
Johns, L. E., Goldman, G. H., Ries, L. N. & Brown, N. A. Nutrient sensing and acquisition in fungi: mechanisms promoting pathogenesis in plant and human hosts. Fungal Biol. Rev. 36, 1–14 (2021).
Lorenz, M. C. et al. The G protein-coupled receptor Gpr1 is a nutrient sensor that regulates pseudohyphal differentiation in Saccharomyces cerevisiae. Genetics 154, 609–622 (2000).
Maidan, M. M. et al. The G protein-coupled receptor Gpr1 and the Gα protein Gpa2 act through the cAMP–protein kinase A pathway to induce morphogenesis in Candida albicans. Mol. Biol. Cell 16, 1971–1986 (2005).
Ballou, E. R. et al. Lactate signalling regulates fungal β-glucan masking and immune evasion. Nat. Microbiol. 2, 1–9 (2016).
DeZwaan, T. M., Carroll, A. M., Valent, B. & Sweigard, J. A. Magnaporthe grisea pth11p is a novel plasma membrane protein that mediates appressorium differentiation in response to inductive substrate cues. Plant Cell 11, 2013–2030 (1999).
Shang, J., Shang, Y., Tang, G. & Wang, C. Identification of a key G-protein coupled receptor in mediating appressorium formation and fungal virulence against insects. Sci. China Life Sci. 64, 466–477 (2021).
Nordbring-Hertz, B., Jansson, H. B. & Tunlid, A. Nematophagous fungi. eLS https://doi.org/10.1038/npg.els.0000374 (2001).
Yang, Y., Yang, E., An, Z. & Liu, X. Evolution of nematode-trapping cells of predatory fungi of the Orbiliaceae based on evidence from rRNA-encoding DNA and multiprotein sequences. Proc. Natl Acad. Sci. USA 104, 8379–8384 (2007).
Yang, E. et al. Origin and evolution of carnivorism in the Ascomycota (fungi). Proc. Natl Acad. Sci. USA 109, 10960–10965 (2012).
Youssar, L. et al. Intercellular communication is required for trap formation in the nematode-trapping fungus Duddingtonia flagrans. PLoS Genet. 15, e1008029 (2019).
Choe, A. et al. Ascaroside signaling is widely conserved among nematodes. Curr. Biol. 22, 772–780 (2012).
Hsueh, Y.-P., Mahanti, P., Schroeder, F. C. & Sternberg, P. W. Nematode-trapping fungi eavesdrop on nematode pheromones. Curr. Biol. 23, 83–86 (2013).
Srinivasan, J. et al. A modular library of small molecule signals regulates social behaviors in Caenorhabditis elegans. PLoS Biol. 10, e1001237 (2012).
Srinivasan, J. et al. A blend of small molecules regulates both mating and development in Caenorhabditis elegans. Nature 454, 1115–1118 (2008).
Pungaliya, C. et al. A shortcut to identifying small molecule signals that regulate behavior and development in Caenorhabditis elegans. Proc. Natl Acad. Sci. USA 106, 7708–7713 (2009).
Yamada, K. et al. Olfactory plasticity is regulated by pheromonal signaling in Caenorhabditis elegans. Science 329, 1647–1650 (2010).
Butcher, R. A., Fujita, M., Schroeder, F. C. & Clardy, J. Small-molecule pheromones that control dauer development in Caenorhabditis elegans. Nat. Chem. Biol. 3, 420–422 (2007).
Kim, K. et al. Two chemoreceptors mediate developmental effects of dauer pheromone in C. elegans. Science 326, 994–998 (2009).
McGrath, P. T. et al. Parallel evolution of domesticated Caenorhabditis species targets pheromone receptor genes. Nature 477, 321–325 (2011).
Park, D. et al. Interaction of structure-specific and promiscuous G-protein-coupled receptors mediates small-molecule signaling in Caenorhabditis elegans. Proc. Natl Acad. Sci. USA 109, 9917–9922 (2012).
Manosalva, P. et al. Conserved nematode signalling molecules elicit plant defenses and pathogen resistance. Nat. Commun. 6, 7795 (2015).
Shinoda, K. et al. Nematode ascarosides attenuate mammalian type 2 inflammatory responses. Proc. Natl Acad. Sci. USA 119, e2108686119 (2022).
Yang, C.-T. et al. Natural diversity in the predatory behavior facilitates the establishment of a robust model strain for nematode-trapping fungi. Proc. Natl Acad. Sci. USA 117, 6762–6770 (2020).
Chen, S.-A., Lin, H.-C. & Hsueh, Y.-P. The cAMP–PKA pathway regulates prey sensing and trap morphogenesis in the nematode-trapping fungus Arthrobotrys oligospora. G3 12, jkac217 (2022).
Chen, S.-A., Lin, H.-C., Schroeder, F. C. & Hsueh, Y.-P. Prey sensing and response in a nematode-trapping fungus is governed by the MAPK pheromone response pathway. Genetics 217, iyaa008 (2021).
Artyukhin, A. B. et al. Metabolomic ‘dark matter’ dependent on peroxisomal β-oxidation in Caenorhabditis elegans. J. Am. Chem. Soc. 140, 2841–2852 (2018).
Fox, B. W. et al. C. elegans as a model for inter-individual variation in metabolism. Nature 607, 571–577 (2022).
Butcher, R. A. et al. Biosynthesis of the Caenorhabditis elegans dauer pheromone. Proc. Natl Acad. Sci. USA 106, 1875–1879 (2009).
Krogh, A., Larsson, B., Von Heijne, G. & Sonnhammer, E. L. Predicting transmembrane protein topology with a hidden Markov model: application to complete genomes. J. Mol. Biol. 305, 567–580 (2001).
Käll, L., Krogh, A. & Sonnhammer, E. L. Advantages of combined transmembrane topology and signal peptide prediction—the Phobius web server. Nucleic Acids Res. 35, W429–W432 (2007).
Dilks, T., Halsey, K., De Vos, R. P., Hammond-Kosack, K. E. & Brown, N. A. Non-canonical fungal G-protein coupled receptors promote Fusarium head blight on wheat. PLoS Pathog. 15, e1007666 (2019).
Dean, R. A. et al. The genome sequence of the rice blast fungus Magnaporthe grisea. Nature 434, 980–986 (2005).
Lin, H.-C. et al. Key processes required for the different stages of fungal carnivory by a nematode-trapping fungus. PLoS Biol. 21, e3002400 (2023).
Hanyaloglu, A. C. & Zastrow, M. V. Regulation of GPCRs by endocytic membrane trafficking and its potential implications. Annu. Rev. Pharmacol. Toxicol. 48, 537–568 (2008).
Jumper, J. et al. Highly accurate protein structure prediction with AlphaFold. Nature 596, 583–589 (2021).
Zhang, Y. & Skolnick, J. TM-align: a protein structure alignment algorithm based on the TM-score. Nucleic Acids Res. 33, 2302–2309 (2005).
Grosdidier, A., Zoete, V. & Michielin, O. SwissDock, a protein-small molecule docking web service based on EADock DSS. Nucleic Acids Res. 39, W270–W277 (2011).
Bai, M. Dimerization of G-protein-coupled receptors: roles in signal transduction. Cell Signal. 16, 175–186 (2004).
Bulenger, S., Marullo, S. & Bouvier, M. Emerging role of homo- and heterodimerization in G-protein-coupled receptor biosynthesis and maturation. Trends Pharmacol. Sci. 26, 131–137 (2005).
Kuo, C.-Y., Chen, S.-A. & Hsueh, Y.-P. The high osmolarity glycerol (HOG) pathway functions in osmosensing, trap morphogenesis and conidiation of the nematode-trapping fungus Arthrobotrys oligospora. J. Fungi 6, 1–11 (2020).
Zhen, Z. et al. MAP kinase Slt2 orthologs play similar roles in conidiation, trap formation and pathogenicity in two nematode-trapping fungi. Fungal Genet. Biol. 116, 42–50 (2018).
Thompson, J. N. The evolution of species interactions. Science 284, 2116–2118 (1999).
Nürnberger, T., Brunner, F., Kemmerling, B. & Piater, L. Innate immunity in plants and animals: striking similarities and obvious differences. Immunol. Rev. 198, 249–266 (2004).
Abisado, R. G., Benomar, S., Klaus, J. R., Dandekar, A. A. & Chandler, J. R. Bacterial quorum sensing and microbial community interactions. MBio 9, e02331–17 (2018).
Chisholm, S. T., Coaker, G., Day, B. & Staskawicz, B. J. Host–microbe interactions: shaping the evolution of the plant immune response. Cell 124, 803–814 (2006).
Zhao, L. et al. Ascarosides coordinate the dispersal of a plant-parasitic nematode with the metamorphosis of its vector beetle. Nat. Commun. 7, 12341 (2016).
Huang, L. et al. NILR1 perceives a nematode ascaroside triggering immune signaling and resistance. Curr. Biol. 33, 3992–3997.e3993 (2023).
Xu, X., Li, G., Li, L., Su, Z. & Chen, C. Genome-wide comparative analysis of putative Pth11-related G protein-coupled receptors in fungi belonging to Pezizomycotina. BMC Microbiol. 17, 1–11 (2017).
Priest, S. J., Yadav, V. & Heitman, J. Advances in understanding the evolution of fungal genome architecture. F1000Res. 9, F1000 (2020).
Conesa, A. et al. Blast2GO: a universal tool for annotation, visualization and analysis in functional genomics research. Bioinformatics 21, 3674–3676 (2005).
Taly, J.-F. et al. Using the T-Coffee package to build multiple sequence alignments of protein, RNA, DNA sequences and 3D structures. Nat. Protoc. 6, 1669–1682 (2011).
Price, M. N., Dehal, P. S. & Arkin, A. P. FastTree: computing large minimum evolution trees with profiles instead of a distance matrix. Mol. Biol. Evol. 26, 1641–1650 (2009).
Letunic, I. & Bork, P. Interactive Tree Of Life (iTOL): an online tool for phylogenetic tree display and annotation. Bioinformatics 23, 127–128 (2007).
de Ulzurrun, G. V.-D., Huang, T.-Y., Chang, C.-W., Lin, H.-C. & Hsueh, Y.-P. Fungal Feature Tracker (FFT): a tool for quantitatively characterizing the morphology and growth of filamentous fungi. PLoS Comput. Biol. 15, e1007428 (2019).
Zhang, Y. K., Sanchez-Ayala, M. A., Sternberg, P. W., Srinivasan, J. & Schroeder, F. C. Improved synthesis for modular ascarosides uncovers biological activity. Org. Lett. 19, 2837–2840 (2017).
Hsueh, Y.-P., Xue, C. & Heitman, J. G protein signaling governing cell fate decisions involves opposing Gα subunits in Cryptococcus neoformans. Mol. Biol. Cell. 18, 3237–3249 (2007).
Schumann, U., Smith, N. A. & Wang, M.-B. A fast and efficient method for preparation of high-quality RNA from fungal mycelia. BMC Res. Notes 6, 1–5 (2013).
Hsu, P.-C. et al. Plastic rewiring of Sef1 transcriptional networks and the potential of nonfunctional transcription factor binding in facilitating adaptive evolution. Mol. Biol. Evol. 38, 4732–4747 (2021).
Kang, Y.-S., Kane, J., Kurjan, J., Stadel, J. M. & Tipper, D. J. Effects of expression of mammalian G alpha and hybrid mammalian-yeast G alpha proteins on the yeast pheromone response signal transduction pathway. Mol. Cell. Biol. 10, 2582–2590 (1990).
Xue, C., Wang, Y. & Hsueh, Y.-P. Assessment of constitutive activity of a G protein-coupled receptor, CPR2, in Cryptococcus neoformans by heterologous and homologous methods. Methods Enzymol. 484, 397–412 (2010).
Bradford, M. M. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal. Biochem. 72, 248–254 (1976).
Acknowledgements
We thank S.-C. Cheng for providing the anti-HA antibody and J.-Y. Leu for assistance with yeast transformation. We thank C.-Y. Lin and T.-H. Chang at the Metabolomics Core Facility, Agricultural Biotechnology Research Center at Academia Sinica for assisting with UPLC–tandem mass spectrometry parameter optimization and data analysis. We thank S.-P. Lee at the Imaging Core at the Institute of Molecular Biology, Academia Sinica for technical assistance with imaging. We are grateful to L.-M. Hsu and A.-M. Yang for their technical assistance in the laboratory. Funding for this work was provided by the Academia Sinica Investigator Award (to Y.-P.H., AS-IA-111-L02); the Ministry of Science and Technology MOST grant (to Y.-P.H., 110-2311-B-001-047-MY3); the Academia Sinica Postdoctoral Scholar Program (to R.J.T., AS-PD 11301-L06); National Science and Technology Council NSTC grant (to Y.-C.C., 108-2113-M-038-002-MY3); and the National Institutes of Health (to F.C.S., R35GM131877). We also thank support from the EMBO YIP and GIN programs.
Author information
Authors and Affiliations
Contributions
C.-Y.K. and Y.-P.H. conceived and designed the research. C.-Y.K., H.-C.L. and S.-C.J. performed the experiments. C.-Y.K., H.-C.L., S.-C.J., G.V.-D.U., Y.-C.C. and Y.-P.H. analysed the data. Y.-C.C., J.H. and F.C.S contributed reagents, materials and analysis tools. C.-Y.K., R.J.T., F.C.S. and Y.-P.H. wrote the paper.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Microbiology thanks Gustavo Goldman, Xingzhong Liu and the other, anonymous, reviewer(s) for their contribution to the peer review of this work. Peer reviewer reports are available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data
Extended Data Fig. 1 Phylogenetic tree of GPCRs from carbon receptor family.
A maximum-likelihood phylogenetic tree of GPCR protein sequences belonging to carbon receptor family from A. oligospora and orthologs from model fungi. Ani: Aspergillus nidulans. Ao: A. oligospora. Cal: Candida albicans. Cne: Cryptococcus neoformans. Fgr: F. graminearum. Mor: M. oryzae. Ncr: Neurospora crassa. Sce: S. cerevisiae. Uma: Ustilago maydis.
Extended Data Fig. 2 Expression of GINs and GPRs is up-regulated after exposure to C. elegans.
a, Transcripts per kilobase million (TPM) values of the differentially expressed Pth11-like GPCRs, with arrows indicating the five highly upregulated Pth11-like GPCRs. (The values represent the average of three independent biological replicates.). b, TPM values of the differentially expressed GPR2 and GPR3. (The values represent the average of three independent biological replicates.). c, d, Protein sequence alignment of Gins (c) and Gprs (d). Three levels of shading and three different symbols are used to indicate degrees of sequence similarity: black background with asterisk (*) indicates identical amino acids, intermediate grey background with colon (:) indicates conserved amino acids, and light grey with single dots (.) indicates semi-conserved amino acids.
Extended Data Fig. 3 Expression of GPR3 and GINs is regulated by Ste12.
a, b, The expression level of GPR (a) and GIN (b) genes in the WT and ste12 mutant was evaluated using qPCR under with or without exposure to C. elegans. GPD1 was used as normalization control. (Data represent mean ± SEM; n shown along the x axis; two-tailed unpaired Student’s t-test; P values are unadjusted.).
Extended Data Fig. 4 The Gins and Gprs are not required for fungal growth.
a, Colonies of the WT, gin, and gpr mutants grown on PDA plates (5.5-cm diameter) for 3 days (Scale bar, 1 cm; the images are representative of three independent biological repeats.). b, Quantification of colony diameter for the A. oligospora WT and gpcr mutants. (Data represent mean ± SEM; n shown along the x axis; two-tailed unpaired Student’s t-test; P values are noted.).
Extended Data Fig. 5 Protein structural alignment of Gpr2, Gpr3, and SRBC-66.
a, b, TM-align was utilized to compare the protein structure of Gpr3 (depicted in green) with Gpr2 (depicted in blue) (a) and SRBC-66 (depicted in red) (b). (0.5 < TM-score < 1.00, in about the same fold). An overall map of the protein structural alignment between Gpr2 (blue), Gpr3 (green), and SRBC-66 (red) was based on the structural predictions from AlphaFold.
Extended Data Fig. 6 The prediction of ascaroside binding pocket in Gpr3.
Using SwissDock to predict the ascaroside binding pocket in Gpr3. An overall map of the docking interactions between Gpr3 (green) and and ascr#3 (a) and ascr#7 (b), based on the structural predictions from AlphaFold. The structural display of the amino acid region from 212 to 264 has been omitted due to its prediction as a random coil. Additionally, an enlarged schematic diagram highlights the potential ascaroside docking region in Gpr3.
Extended Data Fig. 7 Western blot analysis with S. cerevisiae transformants expressing each GPCR-3×HA and Gpa-Myc.
Total proteins were extracted from S. cerevisiae expressing each GPCR-3×HA or each Gpa-Myc construct. The expected 48-kD GPCR-3×HA and 42-kD Gpa-Myc bands were detected in each transformants. Detection with the anti-tubulin antibody was used as the loading control. The blots are representative of two independent biological repeats.
Extended Data Fig. 8 The Gins are not required for ascaroside sensing.
a, Quantification of the trap numbers induced by ascarosides for the A. oligospora WT and gin mutants. (Data represent mean ± SEM; n shown along the x axis; two-tailed unpaired Student’s t-test; P values are noted.). b, Representative brightfield images of the traps induced by C. elegans in the A. oligospora WT and gin mutants. (Scale bar, 200 μm. Black arrow indicates trap; the images are representative of three independent biological repeats.). c, Quantification of the trap numbers induced by C. elegans for the A. oligospora WT and gin mutants. (Data represent ± SEM; n shown along the x axis; two-tailed unpaired Student’s t-test; P values are noted.). d, Images of traps formed by the A. oligospora WT and gin mutants after 24 hours of continuous induction with 400 C. elegans. Vegetative hyphae and traps of A. oligospora were stained with SR2200, which specifically bound to fungal cell walls. (Scale bar, 20 μm; the images are representative of three independent biological repeats.). e, The localization of Gin1-GFP was displayed at 0, 2, and 10 hours after C. elegans induction. Merged image shows the GFP channel, FM4-64, and CMAC. (Scale bar, 10 μm; the images are representative of three independent biological repeats.) f, The expression level of GIN genes in the WT and gin3 mutant was evaluated using qPCR under with or without exposure to C. elegans. GPD1 was used as normalization control. (Data represent ± SEM; n shown along the x axis; two-tailed unpaired Student’s t-test; P values are noted.).
Extended Data Fig. 9 Distribution of GPCRs across the nine A. oligospora chromosomes.
GPCR-encoding genes from Pth11-like family (pink), carbon receptor (blue), and other classes (green) distribute across the nine A. oligospora chromosomes.
Extended Data Fig. 10 Hypothetical model of GPCRs-governed prey sensing and trap development in A. oligospora.
When Gpr2 and Gpr3 recognize ascr#3 and ascr#7, Gpa2 dissociates from the GPCRs and subsequently activates the downstream cAMP-PKA pathway. Additionally, Gin3 is activated by other unknown nematode-derived signals and also operates upstream of the cAMP-PKA pathway. PKA then phosphorylates the downstream substrates required for trap morphogenesis. Additionally, Gpr2, Gpr3, and Gin3 may be involved in the modulation of phosphorylation of Hog1. Gin3 may also partially activate the pheromone response MAPK pathway to induce the expression of GINs and GPR3, which may indicate their potential role as nematode-responsive genes and as receptors for unidentified nematode signals in A. oligospora.
Supplementary information
Supplementary Information
Supplementary Fig. 1 and Tables 1–4.
41564_2024_1679_MOESM4_ESM.xlsx
Supplementary Table 1 Manual annotation of 83 putative GPCR genes. Supplementary Table 2 Wilcoxon test result of Pth11-like GPCR expression. Supplementary Table 3 The primers used in this study. Supplementary Table 4 The strains and plasmids used in this study.
Source data
Source Data Fig. 1
Statistical source data.
Source Data Fig. 2
Statistical source data.
Source Data Fig. 3
Statistical source data.
Source Data Fig. 4
Uncropped western blots and statistical source data.
Source Data Fig. 4
Uncropped western blots and statistical source data.
Source Data Fig. 5
Uncropped western blots and statistical source data.
Source Data Fig. 5
Uncropped western blots and statistical source data.
Source Data Extended Data Fig. 2/Table 2
Graph source data.
Source Data Extended Data Fig. 3/Table 3
Statistical source data.
Source Data Extended Data Fig. 4/Table 4
Statistical source data.
Source Data Extended Data Fig. 7/Table 7
Uncropped western blots and/or gels.
Source Data Extended Data Fig. 8/Table 8
Statistical source data.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Kuo, CY., Tay, R.J., Lin, HC. et al. The nematode-trapping fungus Arthrobotrys oligospora detects prey pheromones via G protein-coupled receptors. Nat Microbiol (2024). https://doi.org/10.1038/s41564-024-01679-w
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41564-024-01679-w