Abstract
Germline loss-of-function mutations in USP9X have been reported to cause a wide spectrum of congenital anomalies. Here, we report a Japanese girl with a novel heterozygous nonsense mutation in USP9X who exhibited intellectual disability with characteristic craniofacial abnormalities, including hypotelorism, brachycephaly, hypodontia, micrognathia, severe dental crowding, and an isolated submucous cleft palate. Our findings provide further evidence that disruptions in USP9X contribute to a broad range of congenital craniofacial abnormalities.
Similar content being viewed by others
Ubiquitin signaling plays a wide variety of roles in both embryonic development and general cellular activities; thus, its disruption can result in a wide range of cellular and tissue defects. This signaling pathway is governed by multiple classes of proteins, one of which is deubiquitylases (DUBs), including ubiquitin-specific peptidase 9 X-linked (USP9X)1. Germline loss-of-function mutation of USP9X has been shown to cause intellectual disability as well as other congenital anomalies. Multiple reports have documented the features of developmental delay resulting from USP9X mutations, which is characterized by retardation of neurogenesis, while the etiology of other characteristic features, such as craniofacial anomalies including intraoral features, has not been reported in detail2.
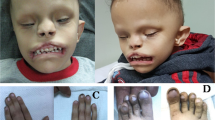
The current patient, a 7-year-old Japanese girl, was referred to the Department of Orthodontics at Osaka University Dental Hospital for the correction of malocclusion. She was born at 40 weeks and 6 days gestation, with a weight of 2768 g, and is the second child of healthy, nonconsanguineous parents. Antenatal ultrasound examinations during pregnancy revealed an umbilical artery. Postnatally, she experienced peripheral circulatory failure due to polycythemia. MRI revealed Dandy–Walker syndrome. She exhibited developmental delays, such as poor weight gain, and underwent intubation feeding at 4 months of age. She also exhibited intellectual disability and required assistance for some subjects at school. Her facial features included hypotelorism, a short columella with a wide nasal base, midfacial deficiency with a thin upper lip (Fig. 1a), low-set ears, and micrognathia (Fig. 1b). Intraoral features included a high-arched palate and malocclusion, such as severe crowding on the lower jaw and an underbite (Fig. 1c–e). Clinical manifestations included hypernasality caused by a submucous cleft palate. In addition, an irregular pigment pattern was detected bilaterally on the upper arms (Fig. 1f). Cephalometric analysis revealed a short anteroposterior maxillary diameter and a retruded mandible (Fig. 2a). Hypodontia (involving the upper right first premolar and bilateral upper second premolars) was evident on panoramic radiography (Fig. 2b). A CT scan of the head revealed plagiocephaly without clear evidence of craniosynostosis and an ectopically positioned upper left canine (Fig. 2c).
Frontal (a) and lateral (b) views of the facial profile. Intraoral photos of the upper (c) and lower (d) jaws. e Underbite was observed in the occlusion. f Abnormal pigment pattern on the arm. g De novo nonsense mutation in the middle of the ubiquitin carboxyl-terminal hydrolase domain of USP9X. h The cryo-EM structure of USP9X (PDB ID: 7YXX). The ubiquitin carboxyl-terminal hydrolase domain is shown in cyan. The faded color indicates the truncated area in the mutated protein. i The crystal structure of the ubiquitin carboxyl-terminal hydrolase domain (PDB ID: 5WCH) is shown in magenta. His1879 and Asp1901 are missing in the mutated USP9X protein.
After comprehensive counseling, explaining the risks and benefits of genetic testing, and obtaining informed consent, whole-exome sequencing was performed using genomic DNA extracted from peripheral lymphocytes of the patient and her parents using the Sure Select Human All Exon Kit V6 (Agilent Technologies, Santa Clara, CA, USA). Sequencing was conducted on the NovaSeq 6000 platform (Illumina, San Diego, CA, USA) to elucidate the causative genes for the patient’s phenotypes. A de novo heterozygous nonsense mutation in USP9X (NM_001039590, hg19: c.4942 C > T: p.Q1648*), which creates a premature stop codon in the middle of the ubiquitin carboxyl-terminal hydrolase (UCH) domain, was identified (Fig. 1g); this mutation has not been documented in previous studies. As a result of this mutation, USP9X is truncated to 1647 amino acids. The USP domain required for the deubiquitination activity of USP9X has a length of 1557 to 1956 amino acids (Fig. 1g). The mutant form of the USP9X protein lacks two crucial amino acid residues within the catalytic triad, strongly suggesting a deficiency in enzymatic activity (Fig. 1h, i)3.
These results indicate that the loss of function of USP9X during embryonic development could result in multiple craniofacial anomalies. However, the mechanism underlying these craniofacial defects is largely unknown. Embryonic craniofacial development relies on coordinated cellular activities, the failure of which could result in a wide variety of morphological and functional defects. Cranial neural crest cells are among the most important cell populations involved in normal craniofacial development. Multiple genetic mutations associated with the developmental process of cranial neural crest cells result in a wide variety of craniofacial defects, including a characteristic facial appearance and orofacial cleft4. Pigment cells are also known to be derived from neural crest cells; therefore, defects in neural crest cells could result in pigment cell defects, as observed in the present case, indicating that neural crest cell defects result from the mutation of USP9X.
Mutations in USP9X in humans have been demonstrated to induce intellectual disability by influencing neurogenesis through the ubiquitin signaling pathway1,5. Interestingly, fibroblasts from patients with USP9X mutations have been demonstrated to exhibit a diminished biological response to the TGFβ signaling pathway, as evidenced by reduced signaling in reporter assays and inhibited cell migration6. In addition, the TGFβ signaling pathway has been shown to phosphorylate USP9X, stabilizing ankyrin-G through deubiquitination in dendritic spine development7,8. TGFβ signaling has also been shown to be critically involved in craniofacial development at various levels. For example, reducing TGFβ signaling in cranial neural crest cells has been demonstrated to increase cell death, subsequently resulting in partial loss of cranial bone9. TGFβ signaling plays critical roles in palatogenesis; loss of function results in cleft palate in both mice and humans10,11. In addition, USP9X has been shown to deubiquitylate DVL2, another key protein regulating the Wnt signaling pathway, altering its activity in the cell12. Canonical Wnt signaling is another central pathway involved in normal craniofacial development. Eliminating beta-catenin, a mediator of canonical Wnt signaling, from neural crest cells causes severe craniofacial development in mice13. Interestingly, polymorphisms in Dvl2 have been associated with susceptibility to orofacial clefts in the Polish population14. Canonical Wnt signaling is also known to regulate the development of dentition, and gene mutations in this pathway could cause the loss of permanent teeth, as in the present case15. Moreover, it has been demonstrated that USP9X is involved in ciliogenesis through the regulation of ubiquitination of key ciliogenic proteins16. Primary cilia serve as sensors for cells, transducing multiple signaling pathways that include critical molecules for embryonic craniofacial development17. The disease spectrum resulting from defects in primary cilia is called ciliopathy, which is associated with a wide variety of congenital defects. Notably, up to 30% of ciliopathies can be primarily defined by craniofacial phenotypes, clearly indicating a biological connection between ciliogenesis and craniofacial development. Craniofacial manifestations of ciliopathy include several phenotypes exhibited in the present case, such as orofacial cleft, facial midline defects such as hypotelorism, micrognathia, and Dandy–Walker malformation, which together are found in the majority of ciliopathy patients18. Considering that the present patient exhibited polydactyly and intellectual disability, which are among the core phenotypic features of ciliopathy, the loss of function of USP9X may be mechanistically related to abnormal ciliogenesis, but further studies are needed to confirm this supposition.
Pathogenic mutations in USP9X are known to exhibit a wide variety of phenotypes. Although no clear genotype‒phenotype correlation has been reported, nonsense-mediated mRNA decay and differential X-chromosome inactivation (XCI) are potential mechanisms that could affect the pathogenesis and phenotypic diversity. Notably, USP9X has been shown to escape XCI, which could influence the phenotype of females19. Studies have highlighted the differences between sexes, with females often exhibiting strong loss-of-function mutations such as premature termination codons, while males tend to have milder forms, such as missense mutations5,6.
HGV database
The relevant data from this Data Report are hosted at the Human Genome Variation Database at https://doi.org/10.6084/m9.figshare.hgv.3402.
References
Murtaza, M., Jolly, L. A., Gecz, J. & Wood, S. A. La FAM fatale: USP9X in development and disease. Cell Mol. Life Sci. 72, 2075–2089 (2015).
Reijnders, M. R. et al. De novo loss-of-function mutations in USP9X cause a female-specific recognizable syndrome with developmental delay and congenital malformations. Am. J. Hum. Genet. 98, 373–381 (2016).
Paudel, P. et al. Crystal structure and activity-based labeling reveal the mechanisms for linkage-specific substrate recognition by deubiquitinase USP9X. Proc. Natl Acad. Sci. USA 116, 7288–7297 (2019).
Trainor, P. A. Craniofacial birth defects: the role of neural crest cells in the etiology and pathogenesis of Treacher Collins syndrome and the potential for prevention. Am. J. Med. Genet. A 152A, 2984–2994 (2010).
Tsurusaki, Y. et al. Novel USP9X variants in two patients with X-linked intellectual disability. Hum. Genome Var. 6, 49 (2019).
Johnson, B. V. et al. Partial loss of USP9X function leads to a male neurodevelopmental and behavioral disorder converging on transforming growth factor beta signaling. Biol. Psychiatry 87, 100–112 (2020).
Yoon, S., Parnell, E. & Penzes, P. TGF-beta-induced phosphorylation of Usp9X stabilizes ankyrin-G and regulates dendritic spine development and maintenance. Cell Rep. 31, 107685 (2020).
Yoon, S. et al. Usp9X controls ankyrin-repeat domain protein homeostasis during dendritic spine development. Neuron 105, 506–21, e7 (2020).
Sasaki, T. et al. TGFbeta-mediated FGF signaling is crucial for regulating cranial neural crest cell proliferation during frontal bone development. Development 133, 371–381 (2006).
Proetzel, G. et al. Transforming growth factor-beta 3 is required for secondary palate fusion. Nat. Genet. 11, 409–414 (1995).
Lidral, A. C. et al. Association of MSX1 and TGFB3 with nonsyndromic clefting in humans. Am. J. Hum. Genet. 63, 557–568 (1998).
Nielsen, C. P., Jernigan, K. K., Diggins, N. L., Webb, D. J. & MacGurn, J. A. USP9X deubiquitylates DVL2 to regulate WNT pathway specification. Cell Rep. 28, 1074–89 e5 (2019).
Brault, V. et al. Inactivation of the beta-catenin gene by Wnt1-Cre-mediated deletion results in dramatic brain malformation and failure of craniofacial development. Development 128, 1253–1264 (2001).
Mostowska, A. et al. Association of DVL2 and AXIN2 gene polymorphisms with cleft lip with or without cleft palate in a Polish population. Birth Defects Res. A Clin. Mol. Teratol. 94, 943–950 (2012).
Yu, M., Wong, S. W., Han, D. & Cai, T. Genetic analysis: Wnt and other pathways in nonsyndromic tooth agenesis. Oral. Dis. 25, 646–651 (2019).
Das, A., Qian, J. & Tsang, W. Y. USP9X counteracts differential ubiquitination of NPHP5 by MARCH7 and BBS11 to regulate ciliogenesis. PLoS Genet 13, e1006791 (2017).
Cortes, C. R., Metzis, V. & Wicking, C. Unmasking the ciliopathies: craniofacial defects and the primary cilium. Wiley Interdiscip. Rev. Dev. Biol. 4, 637–653 (2015).
Schock E. N. & Brugmann S. A. Discovery, diagnosis, and etiology of craniofacial ciliopathies. Cold Spring Harb Perspect Biol. 9, a028258 (2017).
Cotton, A. M. et al. Landscape of DNA methylation on the X chromosome reflects CpG density, functional chromatin state and X-chromosome inactivation. Hum. Mol. Genet. 24, 1528–1539 (2015).
Acknowledgements
We deeply appreciate the patient and her family for participating in this work. This study was supported in part by grants from the Initiative on Rare and Undiagnosed Diseases of the Japanese Agency for Medical and Development (JP23bk0304002) and the Ministry of Education, Science, Sports, and Culture of Japan (no. 19H03858 to H.K.). We also appreciate the support of the Bioinformatics Research Unit, Graduate School of Dentistry, Osaka University, for analyzing the structure of the mutated USP9X.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Nagata, N., Kurosaka, H., Higashi, K. et al. Characteristic craniofacial defects associated with a novel USP9X truncation mutation. Hum Genome Var 11, 21 (2024). https://doi.org/10.1038/s41439-024-00277-w
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41439-024-00277-w