Article
|
Open Access
Featured
-
-
Article |
 A light-driven enzymatic enantioselective radical acylation
A light-driven enzymatic enantioselective radical acylation
Enzyme-bound ketyl radicals derived from thiamine diphosphate are selectively generated through single-electron oxidation by a photoexcited organic dye and shown to lead to enantioselective radical acylation reactions.
- Yuanyuan Xu
- , Hongwei Chen
- & Xiaoqiang Huang
-
Article |
 Enantioconvergent Cu-catalysed N-alkylation of aliphatic amines
Enantioconvergent Cu-catalysed N-alkylation of aliphatic amines
The chemoselective and enantioconvergent N-alkylation of aliphatic amines, including ammonia, is achieved using chiral tridentate anionic ligands and a copper catalyst; the method shows excellent enantioselectivity and functional-group tolerance.
- Ji-Jun Chen
- , Jia-Heng Fang
- & Xin-Yuan Liu
-
Article |
 Enantioselective synthesis of ammonium cations
Enantioselective synthesis of ammonium cations
Enantioselective supramolecular recognition allows for the asymmetric synthesis of nitrogen stereocentres, providing chiral ammonium cations in a dynamic crystallization process.
- Mark P. Walsh
- , Joseph M. Phelps
- & Matthew O. Kitching
-
Article |
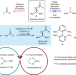 Photoinduced copper-catalysed asymmetric amidation via ligand cooperativity
Photoinduced copper-catalysed asymmetric amidation via ligand cooperativity
In the presence of three ligands and light, two distinct copper catalysts combine to produce enantioenriched secondary amides from racemic alkyl electrophiles and primary amide nucleophiles.
- Caiyou Chen
- , Jonas C. Peters
- & Gregory C. Fu
-
Article |
 Concise asymmetric synthesis of (−)-bilobalide
Concise asymmetric synthesis of (−)-bilobalide
A synthetic route to the Ginkgo biloba metabolite (−)-bilobalide is described.
- Meghan A. Baker
- , Robert M. Demoret
- & Ryan A. Shenvi
-
Letter |
 Asymmetric α-arylation of amino acids
Asymmetric α-arylation of amino acids
This approach introduces an aryl substituent to the α-carbon of amino acids without a transition-metal catalyst, and uses the inherent chirality of the amino acid itself as the source of asymmetry.
- Daniel J. Leonard
- , John W. Ward
- & Jonathan Clayden
-
Letter |
 Building C(sp3)-rich complexity by combining cycloaddition and C–C cross-coupling reactions
Building C(sp3)-rich complexity by combining cycloaddition and C–C cross-coupling reactions
Combining cycloaddition and carbon–carbon cross-coupling offers a way of simplifying the enantioselective preparation of chemical building blocks, natural products and medicines such as the antipsychotic asenapine.
- Tie–Gen Chen
- , Lisa M. Barton
- & Phil S. Baran
-
Letter |
 Site-selective and stereoselective functionalization of non-activated tertiary C–H bonds
Site-selective and stereoselective functionalization of non-activated tertiary C–H bonds
The functionalization of specific inert C–H bonds avoids the need for functional groups in organic synthesis and here the challenges of this approach are overcome using a dirhodium catalyst that is capable of C–H bond site-selectivity.
- Kuangbiao Liao
- , Thomas C. Pickel
- & Huw M. L. Davies
-
News & Views |
 Thin films with a hidden twist
Thin films with a hidden twist
Many naturally occurring substances have a 'handedness' that enables them to interact highly specifically with matter or light. The helical features responsible for this can now be replicated in solid, porous films. See Letter p.422
- Andreas Stein

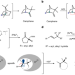 Catalytic asymmetric cationic shifts of aliphatic hydrocarbons
Catalytic asymmetric cationic shifts of aliphatic hydrocarbons