Abstract
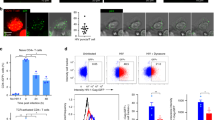
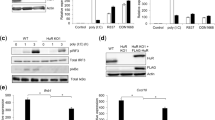
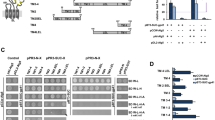
Pattern-recognition receptors (PRRs) elicit antiviral immune responses to human immunodeficiency virus type 1 (HIV-1). Here we show that HIV-1 required signaling by the PRRs Toll-like receptor 8 (TLR8) and DC-SIGN for replication in dendritic cells (DCs). HIV-1 activated the transcription factor NF-κB through TLR8 to initiate the transcription of integrated provirus by RNA polymerase II (RNAPII). However, DC-SIGN signaling was required for the generation of full-length viral transcripts. Binding of the HIV-1 envelope glycoprotein gp120 to DC-SIGN induced kinase Raf-1–dependent phosphorylation of the NF-κB subunit p65 at Ser276, which recruited the transcription-elongation factor pTEF-b to nascent transcripts. Transcription elongation and generation of full-length viral transcripts was dependent on pTEF-b-mediated phosphorylation of RNAPII at Ser2. Inhibition of either pathway abrogated replication and prevented HIV-1 transmission. Thus, HIV-1 subverts crucial components of the immune system for replication that might be targeted to prevent infection and dissemination.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
References
Steinman, R.M. & Banchereau, J. Taking dendritic cells into medicine. Nature 449, 419–426 (2007).
Medzhitov, R. Recognition of microorganisms and activation of the immune response. Nature 449, 819–826 (2007).
Akira, S., Uematsu, S. & Takeuchi, O. Pathogen recognition and innate immunity. Cell 124, 783–801 (2006).
Kanneganti, T.D., Lamkanfi, M. & Núñez, G. Intracellular NOD-like receptors in host defense and disease. Immunity 27, 549–559 (2007).
O'Neill, L.A.J. When signaling pathways collide: positive and negative regulation of Toll-like receptor signal transduction. Immunity 29, 12–20 (2008).
Geijtenbeek, T.B.H. & Gringhuis, S.I. Signalling through C-type lectin receptors: shaping immune responses. Nat. Rev. Immunol. 9, 465–479 (2009).
Meier, A. et al. MyD88-dependent immune activation mediated by human immunodeficiency virus type 1-encoded Toll-like receptor ligands. J. Virol. 81, 8180–8191 (2007).
Beignon, A.S. et al. Endocytosis of HIV-1 activates plasmacytoid dendritic cells via Toll-like receptor-viral RNA interactions. J. Clin. Invest. 115, 3265–3275 (2005).
Heil, F. et al. Species-specific recognition of single-stranded RNA via Toll-like receptor 7 and 8. Science 303, 1526–1529 (2004).
Meier, A. et al. Sex differences in the Toll-like receptor-mediated response of plasmacytoid dendritic cells to HIV-1. Nat. Med. 15, 955–959 (2009).
Hardy, A.W., Graham, D.R., Shearer, G.M. & Herbeuval, J.P. HIV turns plasmacytoid dendritic cells (pDC) into TRAIL-expressing killer pDC and down-regulates HIV coreceptors by Toll-like receptor 7-induced IFN-α. Proc. Natl. Acad. Sci. USA 104, 17453–17458 (2007).
de Witte, L. et al. Langerin is a natural barrier to HIV-1 transmission by Langerhans cells. Nat. Med. 13, 367–371 (2007).
Geijtenbeek, T.B.H. et al. DC-SIGN, a dendritic cell-specific HIV-1-binding protein that enhances trans-infection of T cells. Cell 100, 587–597 (2000).
Gringhuis, S.I. et al. C-type lectin DC-SIGN modulates toll-like receptor signaling via Raf-1 kinase-dependent acetylation of transcription factor NF-κB. Immunity 26, 605–616 (2007).
Gringhuis, S.I., den Dunnen, J., Litjens, M., van der Vlist, M. & Geijtenbeek, T.B.H. Carbohydrate-specific signaling through the DC-SIGN signalosome tailors immunity to Mycobacterium tuberculosis, HIV-1 and Helicobacter pylori. Nat. Immunol. 10, 1081–1088 (2009).
Hodges, A. et al. Activation of the lectin DC-SIGN induces an immature dendritic cell phenotype triggering Rho-GTPase activity required for HIV-1 replication. Nat. Immunol. 8, 569–577 (2007).
Royce, R.A., Sena, A., Cates, W. & Cohen, M.S. Sexual transmission of HIV. N. Engl. J. Med. 336, 1072–1078 (1997).
UNAIDS. Global AIDS epidemic just beginning. AIDS Policy Law 20, 1 (2005).
de Witte, L., Nabatov, A. & Geijtenbeek, T.B. Distinct roles for DC-SIGN+-dendritic cells and Langerhans cells in HIV-1 transmission. Trends Mol. Med. 14, 12–19 (2008).
Wu, L. & KewalRamani, V.N. Dendritic-cell interactions with HIV: infection and viral dissemination. Nat. Rev. Immunol. 6, 859–868 (2006).
Turville, S.G. et al. Immunodeficiency virus uptake, turnover, and 2-phase transfer in human dendritic cells. Blood 103, 2170–2179 (2004).
Perreau, M., Pantaleo, G. & Kremer, E.J. Activation of a dendritic cell-T cell axis by Ad5 immune complexes creates an improved environment for replication of HIV in T cells. J. Exp. Med. 205, 2717–2725 (2008).
Goff, S.P. Host factors exploited by retroviruses. Nat. Rev. Microbiol. 5, 253–263 (2007).
Greene, W.C. & Peterlin, B.M. Charting HIV's remarkable voyage through the cell: Basic science as a passport to future therapy. Nat. Med. 8, 673–680 (2002).
Perkins, N.D. et al. A cooperative interaction between NF-κB and Sp1 is required for HIV-1 enhancer activation. EMBO J. 12, 3551–3558 (1993).
Nabel, G. & Baltimore, D. An inducible transcription factor activates expression of human immunodeficiency virus in T cells. Nature 326, 711–713 (1987).
Peterlin, B.M. & Trono, D. Hide, shield and strike back: how HIV-infected cells avoid immune eradication. Nat. Rev. Immunol. 3, 97–107 (2003).
Barboric, M. & Peterlin, B.M. A new paradigm in eukaryotic biology: HIV Tat and the control of transcriptional elongation. PLoS Biol. 3, e76 (2005).
Egloff, S. & Murphy, S. Cracking the RNA polymerase II CTD code. Trends Genet. 24, 280–288 (2008).
Adams, M. et al. Cellular latency in human immunodeficiency virus-infected individuals with high CD4 levels can be detected by the presence of promoter-proximal transcripts. Proc. Natl. Acad. Sci. USA 91, 3862–3866 (1994).
Kao, S.Y., Calman, A.F., Luciw, P.A. & Peterlin, B.M. Anti-termination of transcription within the long terminal repeat of HIV-1 by tat gene product. Nature 330, 489–493 (1987).
Burleigh, L. et al. Infection of dendritic cells (DCs), not DC-SIGN-mediated internalization of human immunodeficiency virus, is required for long-term transfer of virus to T cells. J. Virol. 80, 2949–2957 (2006).
Kwon, D.S., Gregorio, G., Bitton, N., Hendrickson, W.A. & Littman, D.R. DC-SIGN-mediated internalization of HIV is required for trans-enhancement of T cell infection. Immunity 16, 135–144 (2002).
Brussel, A. & Sonigo, P. Evidence for gene expression by unintegrated human immunodeficiency virus type 1 DNA species. J. Virol. 78, 11263–11271 (2004).
Wellbrock, C., Karasarides, M. & Marais, R. The RAF proteins take centre stage. Nat. Rev. Mol. Cell Biol. 5, 875–885 (2004).
Yedavalli, V.S.R.K., Neuveut, C., Chi, Y.h., Kleiman, L. & Jeang, K.T. Requirement of DDX3 DEAD box RNA helicase for HIV-1 Rev-RRE export function. Cell 119, 381–392 (2004).
Gringhuis, S.I. et al. Dectin-1 directs T helper cell differentiation by controlling noncanonical NF-κB activation through Raf-1 and Syk. Nat. Immunol. 10, 203–213 (2009).
Nowak, D.E. et al. RelA Ser276 phosphorylation is required for activation of a subset of NF-κB-dependent genes by recruiting cyclin-dependent kinase 9/cyclin T1 complexes. Mol. Cell. Biol. 28, 3623–3638 (2008).
Pion, M. et al. APOBEC3G/3F mediates intrinsic resistance of monocyte-derived dendritic cells to HIV-1 infection. J. Exp. Med. 203, 2887–2893 (2006).
Publicover, J., Ramsburg, E., Robek, M. & Rose, J.K. Rapid pathogenesis induced by a vesicular stomatitis virus matrix protein mutant: viral pathogenesis is linked to induction of tumor necrosis factor α. J. Virol. 80, 7028–7036 (2006).
Vermeulen, L., De Wilde, G., Van Damme, P., Vanden Berghe, W. & Haegeman, G. Transcriptional activation of the NF-κB p65 subunit by mitogen- and stress-activated protein kinase-1 (MSK1). EMBO J. 22, 1313–1324 (2003).
de Jong, M.A. et al. TNF-α and TLR agonists increase susceptibility to HIV-1 transmission by human Langerhans cells ex vivo. J. Clin. Invest. 118, 3440–3452 (2008).
Toossi, Z. Virological and immunological impact of tuberculosis on human immunodeficiency virus type 1 disease. J. Infect. Dis. 188, 1146–1155 (2003).
Ranjbar, S. et al. HIV-1 replication is differentially regulated by distinct clinical strains of Mycobacterium tuberculosis. PLoS ONE 4, e6116 (2009).
Lawn, S.D. AIDS in Africa: the impact of coinfections on the pathogenesis of HIV-1 infection. J. Infect. 48, 1–12 (2004).
Hester, R.A. & Kennedy, S.B. Candida infection as a risk factor for HIV transmission. J. Womens Health 12, 487–494 (2003).
Collins, K.R., Quinones-Mateu, M.E., Toossi, Z. & Arts, E.J. Impact of tuberculosis on HIV-1 replication, diversity, and disease progression. AIDS Rev. 4, 165–176 (2002).
Kim, Y.K. et al. Recruitment of TFIIH to the HIV LTR is a rate-limiting step in the emergence of HIV from latency. EMBO J. 25, 3596–3604 (2006).
Sgarbanti, M. et al. Modulation of human immunodeficiency virus 1 replication by interferon regulatory factors. J. Exp. Med. 195, 1359–1370 (2002).
Geijtenbeek, T.B.H. et al. DC-SIGN-ICAM-2 interaction mediates dendritic cell trafficking. Nat. Immunol. 1, 353–357 (2000).
Geijtenbeek, T.B. et al. Identification of DC-SIGN, a novel dendritic cell-specific ICAM-3 receptor that supports primary immune responses. Cell 100, 575–585 (2000).
Lackey, K. et al. The discovery of potent cRaf1 kinase inhibitors. Bioorg. Med. Chem. Lett. 10, 223–226 (2000).
Obrig, T.G., Culp, W.J., McKeehan, W.L. & Hardesty, B. The mechanism by which cycloheximide and related glutarimide antibiotics inhibit peptide synthesis on reticulocyte ribosomes. J. Biol. Chem. 246, 174–181 (1971).
Gallay, P., Hope, T., Chin, D. & Trono, D. HIV-1 infection of nondividing cells through the recognition of integrase by the importin/karyopherin pathway. Proc. Natl. Acad. Sci. USA 94, 9825–9830 (1997).
Chatterji, U. et al. Naturally occurring capsid substitutions render HIV-1 cyclophilin A independent in human cells and TRIM-cyclophilin-resistant in Owl monkey cells. J. Biol. Chem. 280, 40293–40300 (2005).
Duh, E.J., Maury, W.J., Folks, T.M., Fauci, A.S. & Rabson, A.B. Tumor necrosis factor α activates human immunodeficiency virus type 1 through induction of nuclear factor binding to the NF-κB sites in the long terminal repeat. Proc. Natl. Acad. Sci. USA 86, 5974–5978 (1989).
Acknowledgements
We thank M. Oudhoff (Academic Centre for Dentistry Amsterdam) for C. albicans; P. Gallay (The Scripps Research Institute) and C. Aiken (Vanderbilt University School of Medicine) for HIV-1 proviral plasmids; K. Kristiansen (University of Southern Denmark) for the GST-p65 expression plasmid; J. Belisle (Colorado State University; National Institute of Allergy and Infectious Diseases contract HHSN266200400091C) for M. tuberculosis; and J. Kappes and X. Wu (Tranzyme) for TZM-bl cells (obtained through the National Institutes of Health AIDS Research and Reference Reagent Program). Supported by the Netherlands Organisation for Scientific Research (NWO 917-46-367 to M.L. and NWO 912-04-025 to J.d.D.), the AIDS Foundation (2007036 to M.v.d.V.), Dutch Burns Foundation (08.109 to L.M.v.d.B) and the Dutch Asthma Foundation (3.2.03.39 to S.I.G.).
Author information
Authors and Affiliations
Contributions
S.I.G. designed, executed and interpreted most experiments and prepared the manuscript; M.v.d.V. executed HIV-1 stimulation, infection and transmission experiments, isolated myeloid DCs and helped with the nuclear extract preparations; L.M.v.d.B. isolated dermal DCs; J.d.D. helped with flow cytometry; M.L. helped with GST-p65 purification and setting up the GST enzyme-linked immunosorbent assay (ELISA); and T.B.H.G. supervised all aspects of this study, including study design, execution and interpretation, and manuscript preparation.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–4 and Supplementary Table 1 (PDF 547 kb)
Rights and permissions
About this article
Cite this article
Gringhuis, S., van der Vlist, M., van den Berg, L. et al. HIV-1 exploits innate signaling by TLR8 and DC-SIGN for productive infection of dendritic cells. Nat Immunol 11, 419–426 (2010). https://doi.org/10.1038/ni.1858
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ni.1858
This article is cited by
-
Functions of MAP3Ks in antiviral immunity
Immunologic Research (2023)
-
HIV transmitting mononuclear phagocytes; integrating the old and new
Mucosal Immunology (2022)
-
Human anogenital monocyte-derived dendritic cells and langerin+cDC2 are major HIV target cells
Nature Communications (2021)
-
Cathepsin S: investigating an old player in lung disease pathogenesis, comorbidities, and potential therapeutics
Respiratory Research (2020)
-
HIV-1 subtype C transmitted founders modulate dendritic cell inflammatory responses
Retrovirology (2020)