Abstract
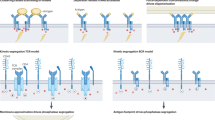
B-cell receptor signaling in response to membrane-bound antigen increases with antigen affinity, a process known as affinity discrimination. We use computational modeling to show that B-cell affinity discrimination requires that kinetic proofreading predominate over serial engagement. We find that if B-cell receptors become signaling-capable immediately upon antigen binding, which results in decreasing serial engagement as affinity increases, then increasing affinity can lead to weaker signaling. Rather, antigen must stay bound to B-cell receptors for a threshold time of several seconds before becoming signaling-capable, a process similar to kinetic proofreading. This process overcomes the loss in serial engagement due to increasing antigen affinity, and replicates the monotonic increase in B-cell signaling with increasing affinity that has been observed in B-cell activation experiments. This finding matches well with the experimentally observed time (∼20 s) required for the B-cell receptor signaling domains to undergo antigen and lipid raft-mediated conformational changes that lead to Src-family kinase recruitment. We hypothesize that the physical basis for a threshold time of antigen binding might lie in the formation timescale of B-cell receptor dimers. The time required for dimer formation decreases with increasing antigen affinity, thereby resulting in shorter threshold antigen binding times as affinity increases. Such an affinity-dependent kinetic proofreading requirement results in affinity discrimination very similar to that observed in biological experiments. B-cell affinity discrimination is critical to the process of affinity maturation and the production of high-affinity antibodies, and thus our results have important implications in applications such as vaccine design.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout









Similar content being viewed by others
References
Lanzavecchia A . Antigen-specific interaction between T and B cells. Nature 1985; 314: 537–539.
Batista FD, Neuberger M . Affinity dependence of the B-cell response to antigen: a threshold, a ceiling, and the importance of off-rate. Immunity 1998; 8: 751–759.
Kouskoff V, Famiglietti S, Lacaud G, Lang P, Rider JE, Kay BK et al. Antigens varying in affinity for the B cell receptor induce differential B lymphocyte responses. J Exp Med 1998; 188: 1453–1464.
Batista FD, Neuberger M . B cells extract and present immobilized antigen: implications for affinity discrimination. EMBO J 2000; 19: 513–520.
Shih TA, Meffre E, Roederer M, Nussenzweig MC . Role of antigen receptor affinity in T cell-independent antibody responses in vivo. Nat Immunol 2002; 3: 399–406.
Shih TA, Meffre E, Roederer M, Nussenzweig MC . Role of BCR affinity in T cell dependent responses in vivo. Nat Immunol 2002; 3: 570–575.
Carrasco YR, Fleire SJ, Cameron T, Dustin ML, Batista FD . LFA-1/ICAM-1 interaction lowers the threshold of B cell activation by facilitating B cell adhesion and synapse formation. Immunity 2004; 20: 589–599.
Fleire SJ, Goldman JP, Carrasco YR, Weber M, Bray D, Batista FD . B cell ligand discrimination through a spreading and contracting response. Science 2006; 312: 738–741.
Liu W, Meckel T, Tolar P, Sohn HW, Pierce SK . Antigen affinity discrimination is an intrinsic function of the B cell receptor. J Exp Med 2010; 207: 1095–1111.
Carrasco YR, Batista FD . B cell recognition of membrane-bound antigen: an exquisite way of sensing ligands. Curr Opin Immunol 2006; 18: 286–291.
Batista FD, Iber D, Neuberger MS . B cells acquire antigen from target cells after synapse formation. Nature 2001; 411: 489–494.
Carrasco YR, Batista FD . B cells acquire particulate antigen in a macrophage-rich area at the boundary between the follicle and the subcapsular sinus of the lymph node. Immunity 2007; 27: 160–171.
Depoil D, Fleire S, Treanor BL, Weber M, Harwood NE . CD19 is essential to B cell activation by promoting B cell receptor–antigen micro-cluster formation in response to membrane-bound ligand. Nat Immunol 2008; 9: 63–72.
Batista FD, Harwood NE . The who, how, and where of antigen presentation to B cells. Nat Rev Immunol 2009; 9: 15–27.
Carrasco YR, Batista FD . B-cell activation by membrane-bound antigens is facilitated by the interaction of VLA-4 with VCAM-1. EMBO J 2006; 25: 889–899.
Bergtold A, Desai DD, Gavhane A, Clynes R . Cell surface recycling of internalized antigen permits dendritic cell priming of B cells. Immunity 2005; 23: 503–514.
Junt T, Moseman EA, Iannacone M, Massberg S, Lang PA, Boes M . Subcapsular sinus macrophages in lymph nodes clear lymph-borne viruses and present them to antiviral B cells. Nature 2007; 450: 110–114.
Phan TG, Grigorova I, Okada T, Cyster JG . Subcapsular encounter and complement dependent transport of immune complexes by lymph node B cells. Nat Immunol 2007; 8: 992–1000.
Qi H, Egen JG, Huang AY, Germain RN . Extrafollicular activation of lymph node B cells by antigen-bearing dendritic cells. Science 2006; 312: 1672–1676.
Schwickert TA, Lindquist RL, Shakhar G, Livshits G . Skokos D. In vivo imaging of germinal centers reveals a dynamic open structure. Nature 2007; 446: 83–87.
Balázs M, Martin F, Zhou T, Kearney J . Blood dendritic cells interact with splenic marginal zone B cells to initiate T-independent immune responses. Immunity 2002; 17: 341–352.
Tolar P, Sohn HW, Pierce SK . Viewing the antigen induced initiation of B cell activation in living cells. Immunol Rev 2008; 221: 64–76.
Sohn HW, Tolar P, Pierce SK . Membrane heterogeneities in the formation of B cell receptor–Lyn kinase microclusters and the immune synapse. J Cell Biol 2008; 182: 367–379.
McKeithan TW . Kinetic proofreading in T-cell receptor signal-transduction. Proc Natl Acad Sci USA 1995; 92: 5042–5046.
Goldstein B, Faeder JR, Hlavacek WS . Mathematical and computational models of immune-receptor signaling. Nat Rev Immunol 2004; 4: 445–456.
Grakoui A, Bromley SK, Sumen C, Davis MM, Shaw AS, Allen PM . The immunological synapse: a molecular machine controlling T cell activation. Science 1999; 285: 221–227.
Coombs D, Kalergis AM, Nathenson SG, Wofsy C, Goldstein B . Activated TCRs remained marked for internalization after dissociation from pMHC. Nat Immunol 2002; 3: 926–931.
Kalergis AM, Boucheron N, Doucey MA, Palmieri E, Goyarts EC, Vegh Z . Efficient cell activation requires an optimal dwell-time of interaction between the TCR and the pMHC complex. Nat Immunol 2001; 2: 229–234.
Coombs D, Dembo M, Wofsy C, Goldstein B . Equilibrium thermodynamics of cell–cell adhesion mediated by multiple receptor–ligand pairs. Biophys J 2004; 86: 1408–1423.
Tsourkas P, Baumgarth N, Simon SI, Raychaudhuri S . Mechanisms of B cell synapse formation predicted by Monte Carlo simulation. Biophys J 2007; 92: 4196–4208.
Tsourkas P, Longo ML, Raychaudhuri S . Monte Carlo simulation of single molecule diffusion can elucidate the mechanism of B cell synapse formation. Biophys J 2008; 95: 1118–1125.
Raychaudhuri S, Tsourkas P, Willgohs E . Computational modeling of receptor–ligand binding and cellular signaling processes. In: JueT(ed.) Handbook of Modern Biophysics, Vol. I: Fundamentals New York: Humana Press 2009: 41–61.
Bell GI . Cell–cell adhesion in the immune system. Immunol Today 1983; 4: 237–240.
Dembo M, Torney TC, Saxman K, Hammer D . The reaction-limited kinetics of membrane-to-surface adhesion and detachment. Proc R Soc Lond B 1988; 234: 55–83.
Hou P, Araujo E, Zhao T, Zhang M, Massenburg D, Veselits M et al. B cell receptor signaling and internalization are mutually exclusive events. PLoS Biol 2006; 4: 1147–1158.
Favier B, Burroughs NJ, Weddeburn L, Valitutti S . T cell antigen receptor dynamics on the surface of living cells. Int Immunol 2001; 13: 1525–1532.
Wofsy C, Torigoe C, Kent UM, Metzger H, Goldstein B . Exploiting the difference between intrinsic and extrinsic kinases: implications for regulation of signaling by immunoreceptors. J Immunol 1997; 159: 5984–5992.
Faeder JR, Hlavacek WS, Reischl I, Blinov ML, Metzger H, Redondo A et al. Investigation of early events in FcεRI-mediated signaling using a detailed mathematical model. J Immunol 2003; 170: 3769–3781.
Tsang E, Giannetti AM, Shaw D, Dinh M, Tse JK, Ghandi S et al. Molecular mechanism of the Syk activation switch. J Biol Chem 2008; 283: 32650-32659.
Tolar P, Sohn HW, Pierce SK . The initiation of antigen induced BCR signaling viewed in living cells by FRET. Nat Immunol 2005; 6: 1168–1176.
Valitutti S, Muller S, Cella M, Padovan E, Lanzavecchia A . Serial triggering of many T-cell receptors by a few peptide–MHC complexes. Nature 1995; 375: 148–151.
Valitutti S, Lanzavecchia A . Serial triggering of TCRs: a basis for the sensitivity and specificity of antigen recognition. Immunol Today 1997; 18: 299–304.
Tolar P, Hanna J, Krueger PD, Pierce SK . The constant region of the membrane immunoglobulin mediates B cell–receptor clustering and signaling in response to membrane antigens. Immunity 2009; 30: 44–55.
Tolar P, Sohn HW, Liu W, Pierce SK . The molecular assembly and organization of signaling active B-cell receptor oligomers. Immunol Rev 2009; 232: 34–41.
Acknowledgements
The authors thank Dr Emanual Maverakis, Dr Aaron Dinner and Dr Stephen Kaattari for proofreading the manuscript and offering valuable advice. PT and SR are supported by NIH grant AI074022.
Author information
Authors and Affiliations
Corresponding author
Supplementary information
Rights and permissions
About this article
Cite this article
Tsourkas, P., Liu, W., Das, S. et al. Discrimination of membrane antigen affinity by B cells requires dominance of kinetic proofreading over serial engagement. Cell Mol Immunol 9, 62–74 (2012). https://doi.org/10.1038/cmi.2011.29
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/cmi.2011.29