Abstract
Addition of low doses of the atypical antipsychotic drug brexpiprazole with selective serotonin reuptake inhibitors (SSRIs) could promote antidepressant effect in patients with major depressive disorder although the precise mechanisms underlying the action of the combination are unknown. Combination of low dose of brexpiprazole (0.1 mg/kg) and SSRI fluoxetine (10 mg/kg) could promote a rapid antidepressant effect in social defeat stress model although brexpiprazole or fluoxetine alone did not show antidepressant effect. Furthermore, the combination significantly improved alterations in the brain-derived neurotrophic factor (BDNF) - TrkB signaling and dendritic spine density in the prefrontal cortex, hippocampus, and nucleus accumbens in the susceptible mice after social defeat stress. Interestingly, TrkB antagonist ANA-12 significantly blocked beneficial effects of combination of brexpiprazole and fluoxetine on depression-like phenotype. These results suggest that BDNF-TrkB signaling plays a role in the rapid antidepressant action of the combination of brexpiprazole and fluoxetine.
Similar content being viewed by others
Introduction
Substantial clinical data demonstrate that addition of low doses of atypical antipsychotic drugs (e.g., aripiprazole, olanzapine, quetiapine, risperidone, ziprasidone) to selective serotonin reuptake inhibitors (SSRIs) rapidly enhance the antidepressant effects in patients with major depressive disorder (MDD), including treatment-resistant patients1,2,3,4,5,6,7. Although clinical outcome of combined atypical antipsychotic drug and SSRI might be similar to ketamine’s induced rapid antidepressant effect8,9,10, the precise mechanisms underlying rapid antidepressant effect of the combination are currently unclear11,12.
Brexpiprazole (7-{4-[4-(1-benzothiophen-4-yl)piperazin-1-yl]butoxy}quinolin-2(1H)-one) is a serotonin-dopamine activity modulator13. Brexpiprazole binds with high affinity (Ki < 1 nM) to human serotonin (5-HT1A)-, 5-HT2A-, dopamine D2 (D2L)-, α1B-, and α2C-adrenergic receptors. It displays partial agonism at 5-HT1A and D2 receptors, and potent antagonism of 5-HT2A receptors and α1B/2C-adrenoceptors13. Furthermore, brexpiprazole was also shown to potentiate nerve growth factor (NGF)-induced neurite outgrowth in PC12 cells via 5-HT1A and 5-HT2A receptors14, suggesting that brexpiprazole may stimulate neuronal plasticity. Moreover, brexpiprazole showed antipsychotic-like and procognitive effects in rodents15,16,17. Brexpiprazole has been developed to offer efficacious and tolerable therapy for schizophrenia18,19,20,21,22. In addition, brexpiprazole was also developed as adjunctive therapy to antidepressants for the treatment of MDD18,21,23,24,25,26.
The purpose of this study is to examine whether brexpiprazole could demonstrate antidepressant-like effects in combination with sub-threshold dose of the SSRI fluoxetine in depression-like behaviors and alterations in the spine density in stress susceptible mice after repeated social defeat stress. It is well known that brain-derived neurotrophic factor (BDNF) and its receptor TrkB signaling plays a key role in the therapeutic mechanisms of the rapid antidepressants27,28,29,30,31,32,33. Therefore, we examined the role of BDNF-TrkB signaling in the mechanisms of a rapid antidepressant action of combination of brexpiprazole and fluoxetine.
Results
Effects of fluoxetine and brexpiprazole on depression-like behavior in susceptible mice after repeated social defeat stress
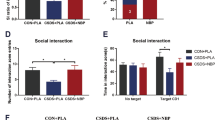
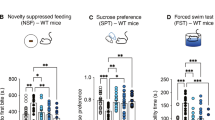
We examined effects of fluoxetine and brexpiprazole on depression-like behavior after repeated social defeat stress. Vehicle, fluoxetine (10 mg/kg), brexpiprazole (0.1 mg/kg), or fluoxetine (10 mg/kg) plus brexpiprazole (0.1 mg/kg) was administered orally into susceptible mice (Fig. 1a). In the locomotion test (LMT), there were no differences (F4,34 = 1.347, P = 0.276) among the five groups (Fig. 1b). One-way ANOVA of TST and FST data revealed a significant result (TST: F4,33 = 6.139, P = 0.001, FST: F4,43 = 2.767, P = 0.043). In the TST and FST, combination of fluoxetine and brexpiprazole significantly reduced the increased immobility time in the susceptible mice after repeated social defeat stress (Fig. 1c,d). One-way ANOVA of SPT data revealed a significant result (F4,38 = 2.650, P = 0.048). In the SPT, combination of fluoxetine and brexpiprazole significantly increased the decreased sucrose preference of susceptible mice (Fig. 1e). In contrast, fluoxetine or brexpiprazole alone did not alter the immobility time for TST and FST, and decreased sucrose preference in the susceptible mice (Fig. 1c–e). These findings suggest that adjunctive treatment of brexpiprazole with fluoxetine showed a rapid antidepressant effect in the susceptible mice after repeated social defeat stress.
Antidepressant effects of combination of brexpiprazole and fluoxetine in social defeat stress model.
(a): Schedule of social defeat stress, treatment, and behavioral tests. Repeated social defeat stress was performed 10 days (day 1- day 10). Social interaction test was performed day 11, and susceptible mice were used subsequent experiments. Vehicle (10 ml/kg), fluoxetine (10 mg/kg), brexpiprazole (0.1 mg/kg), or fluoxetine (10 mg/kg) plus brexpiprazole (0.1 mg/kg) were administered orally. Locomotion (LST), tail-suspension test (TST), and forced swimming test (FST) were performed 2, 4, and 6 hours after oral administration (day 12). One % sucrose preference test (SPT) was performed 24 hours after oral administration (day 13). (b): LMT, (c): TST, (d): FST, (e): SPT. Data are shown as mean ± S.E.M. (n = 6–9). *P < 0.05, **P < 0.01, ***P < 0.001 compared to vehicle-treated stress group (one-way ANOVA, followed post hoc LSD test). N.S.: Not significant.
Effects of fluoxetine and brexpiprazole on BDNF-TrkB signaling in selected brain regions of mice with depression-like phenotype
Since PFC, NAc, striatum, CA1, CA3 and dentate gyrus (DG) of the hippocampus play a role in the depression-like phenotype in rodents34,35,36,37,38,39, we performed Western blot analysis of BDNF (mature form), its precursor proBDNF, TrkB and phosphorylated TrkB (p-TrkB) in selected brain regions (PFC, NAc, striatum, DG, CA1 and CA3). One-way ANOVA of BDNF data revealed the following statistical significances: PFC: F4,27 = 3.705, P = 0.016, NAc: F4,27 = 18.79, P < 0.0001, striatum: F4,27 = 1.934, P = 0.132, CA1: F4,27 = 0.381, P = 0.82; CA3: F4,27 = 4.227, P = 0.009; DG: F4,27 = 5.53, P = 0.002 (Fig. 2a–f). Combination of brexpiprazole and fluoxetine significantly attenuated decreased BDNF levels in the PFC, CA3 and DG regions, but not CA1 region, of susceptible mice (Fig. 2a,d–f). Interestingly, combination of brexpiprazole and fluoxetine significantly attenuated increased BDNF levels in the NAc of susceptible mice (Fig. 2b). However, protein levels of proBDNF in all the regions examined were not different among the five treatment groups (Supplemental Fig. 1a–f).
Effects of brexpiprazole and fluoxetine combination on the alterations in the BDNF-TrkB signaling in the brain regions from susceptible mice after social defeat stress.
(a–f): Forty eight hours after administration of drugs, brain regions from mice were collected. Western blot analysis of BDNF (mature form) and β-actin in the brain regions (PFC, NAc, striatum, CA1, CA3, DG) was performed. The values are expressed as a percentage of that of control mice. Representative data of Western blot analyses of BDNF and β-actin in the mouse brain regions. Data are shown as mean ± S.E.M. (n = 5–8). *P < 0.05, **P < 0.01, ***P < 0.001 compared to vehicle-treated stress group (one-way ANOVA, followed post hoc LSD test). N.S.: Not significant. (g–l): The ratio of p-TrkB to total TrkB in the brain regions is shown. The values are expressed as a percentage of that of control mice. Representative data of Western blot analyses of p-TrkB, and TrkB in the mouse brain regions. Data are shown as mean ± S.E.M. (n = 5–8). *P < 0.05, **P < 0.01, ***P < 0.001 compared to vehicle-treated stress group (one-way ANOVA, followed post hoc LSD test). N.S.: Not significant.
To clarify whether TrkB activation or inhibition is the underlying mechanism of action of brexpiprazole and fluoxetine combination, we performed Western blot analyses of TrkB and phosphorylated TrkB (p-TrkB), an activated form of TrkB, in samples from PFC, NAc, striatum, and CA1, CA3, DG of hippocampus. One-way ANOVA of p-TrkB/TrkB data revealed the following statistical results: PFC: F4,27 = 11.14, P < 0.0001, NAc: F4,27 = 6.095, P = 0.001, striatum: F4,27 = 0.698, P = 0.6, CA1: F4,27 = 0.764, P = 0.558; CA3: F4,27 = 6.149, P = 0.001; DG: F4,27 = 8.26, P = 0.0002 (Fig. 2g–l). Combination of brexpiprazole and fluoxetine significantly attenuated the decrease in p-TrkB/TrkB ratio in the PFC, CA3 and DG regions, but not CA1 region, of susceptible mice (Fig. 2g,j,k,l). Interestingly, combination of brexpiprazole and fluoxetine significantly attenuated the increase of p-TrkB/TrkB ratio in the NAc of susceptible mice (Fig. 2h). However, protein levels of TrkB in all these regions were not different among the five groups (Supplemental Fig. 1g–l).
Effects of combination of fluoxetine and brexpiprazole on dendritic spines density in selected brain regions of mice with depression-like phenotype
Repeated social defeat stress causes alterations in the dendritic spines density in the PFC, CA3 and DG of hippocampus, and NAc37,39. In this study, we examined whether combination of brexpiprazole and fluoxetine could cause alterations in the dendritic spines density in the prelimbic (PrL) and infralimbic (IL) regions of mPFC, shell and core of NAc, CA1, CA3 and DG of the hippocampus (Fig. 3a–h). One-way ANOVA of Golgi staining data revealed the following statistical results: (PrL of mPFC: F4,21 = 158.0, P < 0.0001, IL of mPFC: F4,20 = 2.44, P = 0.08, NAc core: F4,20 = 3.305, P = 0.042, NAc shell: F4,20 = 2.96, P = 0.045, CA1: F4,21 = 2.294, P = 0.093; CA3: F4,21 = 28.79, P < 0.0001; DG: F4,21 = 20.51, P < 0.0001)(Fig. 3b–i). Combination of brexpiprazole and fluoxetine significantly attenuated decreased spine density in the PrL of mPFC, CA3 and DG regions of susceptible mice (Fig. 3b,g,h). Interestingly, combination of brexpiprazole and fluoxetine significantly attenuated the increase in spine density in the core and shell of NAc of susceptible mice (Fig. 3d,e). In contrast, administration of brexpiprazole or fluoxetine alone did not affect the alterations in the dendritic spines density in these regions (Fig. 3b–h).
Effects of brexpiprazole and fluoxetine combination on the alterations in the dendritic spine density in the brain regions from susceptible mice after social defeat stress.
(a): Schedule of social defeat stress, treatment, and behavioral tests. Repeated social defeat stress was performed 10 days (day 1- day 10). Social interaction test was performed day 11, and susceptible mice were used subsequent experiments. Two hours after oral administration of vehicle (10 ml/kg), fluoxetine (10 mg/kg), brexpiprazole (0.1 mg/kg), or fluoxetine (10 mg/kg) plus brexpiprazole (0.1 mg/kg), brains from mice were collected for Golgi staining. (b–h): Golgi staining in the brain regions (PrL and IL regions of mPFC, core and shell of NAc, CA1, CA3, DG of hippocampus) was performed. Representative data of Golgi staining in the mouse brain regions. Data are shown as mean ± S.E.M. (n = 5 or 6). *P < 0.05, **P < 0.01, ***P < 0.001 compared to vehicle-treated stress group (one-way ANOVA, followed post hoc LSD test). N.S.: Not significant.
Role of TrkB in the mechanism of action of brexpiprazole and fluoxetine combination in the social defeat stress model
To assess the role of TrkB on the mechanism of action of combination of brexpiprazole and fluoxetine, we examined the effects of ANA-12, a novel TrkB antagonist40, on depression-like behavior after social defeat stress (Fig. 4a). One-way ANOVA of the behavioral data showed the following statistical results: (LMT: F4,35 = 0.287, P = 0.884, TST: F4,34 = 6.163, P = 0.0008, FST: F4,33 = 12.73, P < 0.0001, SPT: F4,33 = 9.022, P < 0.0001) (Fig. 4b–e). In the TST and FST, treatment with ANA-12 significantly blocked the effects of brexpiprazole plus fluoxetine on the increased immobility time of susceptible mice (Fig. 4c,d). Furthermore, treatment with ANA-12 significantly blocked the effects of brexpiprazole plus fluoxetine on the decreased sucrose preference of susceptible mice (Fig. 4e). In addition, ANA-12 alone showed antidepressant effects in these tests, consistent with previous reports37,38,39.
Effects of TrkB antagonist ANA-12 on antidepressant effect of combination of brexpiprazole and fluoxetine.
(a): Schedule of social defeat stress, treatment, and behavioral tests. Repeated social defeat stress was performed 10 days (day 1- day 10). Social interaction test was performed day 11, and susceptible mice were used subsequent experiments. Vehicle (10 ml/kg), fluoxetine (10 mg/kg), brexpiprazole (0.1 mg/kg), or fluoxetine (10 mg/kg) plus brexpiprazole (0.1 mg/kg) were administered orally 30 min after administration of vehicle or ANA-12 (0.5 mg/kg). Locomotion (LST), tail-suspension test (TST), and forced swimming test (FST) were performed 2, 4, and 6 hours after oral administration (day 12). One % sucrose preference test (SPT) was performed 24 hours after oral administration (day 13). (b): LMT, (c): TST, (d): FST, (e): SPT. Data are shown as mean ± S.E.M. (n = 6–9). *P < 0.05, **P < 0.01, ***P < 0.001 compared to vehicle-treated stress group (one-way ANOVA, followed post hoc LSD test). N.S.: Not significant.
Role of BDNF-TrkB signaling in the mechanism of action of brexpiprazole and fluoxetine combination
Since TrkB antagonist ANA-12 blocked antidepressant effect of brexpiprazole and fluoxetine combination in the social defeat stress model, we performed Western blot analysis of BDNF-TrkB signaling in the brain regions. One-way ANOVA of BDNF data showed the following statistical results: (PFC: F4,28 = 25.33, P < 0.0001, NAc: F4,28 = 7.179, P = 0.001, striatum: F4,28 = 1.203, P = 6.331, CA1: F4,28 = 0.349, P = 0.843, CA3: F4,28 = 5.532, P = 0.002, DG: F4,29 = 4.112, P = 0.009) (Fig. 5a–f). Combination of brexpiprazole and fluoxetine significantly attenuated the decrease in levels of BDNF in the PFC, CA3, and DG from susceptible mice after social defeat stress (Fig. 5a,e,f). However, treatment with ANA-12 did not alter the levels of BDNF in these brain regions.
Effect of TrkB antagonist ANA-12 on the alterations in the BDNF-TrkB signaling in the brain regions from susceptible mice after social defeat stress.
(a–f): Forty eight hours after administration of drugs, brain regions from mice were collected. Western blot analysis of BDNF (mature form) and β-actin in the brain regions (PFC, NAc, striatum, CA1, CA3, DG) was performed. The values are expressed as a percentage of that of control mice. Data are shown as mean ± S.E.M. (n = 6–8). *P < 0.05, **P < 0.01, ***P < 0.001 compared to vehicle-treated stress group (one-way ANOVA, followed post hoc LSD test). N.S.: Not significant. (g–l): The ratio of p-TrkB to total TrkB in the brain regions is shown. Representative data of Western blot analyses of p-TrkB, and TrkB in the mouse brain regions. The values are expressed as a percentage of that of control mice. Data are shown as mean ± S.E.M. (n = 6–8). *P < 0.05, **P < 0.01, ***P < 0.001 compared to vehicle-treated stress group (one-way ANOVA, followed post hoc LSD test). N.S.: Not significant.
One-way ANOVA of p-TrkB/TrkB ratio data showed the following statistical results: (PFC: F4,28 = 4.642, P = 0.005, NAc: F4,29 = 7.305, P < 0.0003, striatum: F4,28 = 0.545, P = 0.704, CA1: F4,28 = 1.063, P = 0.393, CA3: F4,29 =7.056, P = 0.0001, DG: F4,29 = 9.935, P < 0.0001) (Fig. 5g–l). Combination of brexpiprazole and fluoxetine significantly attenuated the decreased p-TrkB/TrkB ratio in the PFC, CA3, and DG from susceptible mice after social defeat stress (Fig. 5g,k,l). Furthermore, treatment with ANA-12 significantly blocked the effect of the combination of brexpiprazole and fluoxetine in these regions (PFC, CA3, DG), suggesting a role of TrkB in the mechanism of action of the combination therapy. Although combination of brexpiprazole and fluoxetine significantly attenuated the increased p-TrkB/TrkB ratio in the NAc of susceptible mice, ANA-12 did not block the effect of the combination (Fig. 5h). However, treatment with ANA-12 alone significantly attenuated the increase of p-TrkB/TrkB ratio in the NAc of susceptible mice, consistent with previous reports37,38,39. Protein levels of proBDNF and TrkB in the all brain regions were not significantly different among the five groups (Supplemental Fig. 2a–l).
Discussion
The key findings of this study demonstrate that although brexpipazole or fluoxetine alone did not show antidepressant effect, their combination could promote a rapid antidepressant effect in the social defeat stress model of depression. Recently, we reported a rapid antidepressant effect of the N-methyl-D-aspartate (NMDA) receptor antagonist ketamine (or R-ketamine) in the same model37,39,41, indicating that the rapid antidepressant effect of combination of brexpiprazole and fluoxetine might be similar to ketamine’s rapid antidepressant action. A recent study demonstrated that, similar to ketamine, a combination of olanzapine and fluoxetine facilitated NMDA- and AMPA-induced currents in pyramidal cells via activation of dopamine D1 receptors42, suggesting that rapid antidepressant effect of both antipsychotic drug and SSRI may be related to a common mechanism of action. To the best of our knowledge, this is the first report showing a rapid antidepressant effect for brexpiprazole plus fluoxetine in the social defeat stress model. Therefore, adjunctive therapy of brexpiprazole with SSRI could promote a rapid antidepressant effect in MDD patients. Interestingly, we showed that TrkB antagonist ANA-12 significantly blocked the rapid antidepressant effect of combination of brexpiprazole and fluoxetine in this model. Given the role of BDNF-TrkB signaling in the antidepressant effects of ketamine37,43,44,45,46, it is therefore likely that BDNF-TrkB signaling may also play a key role in the rapid antidepressant effect of combination of brexpiprazole and fluoxetine in the social defeat stress model.
We previously reported a marked reduction of BDNF-TrkB signaling in the PFC, DG and CA3, but not CA1, of inflammation model38, repeated social defeat stress model37,39,41 and learned helplessness model35,36,47. Direct infusion of BDNF (or TrkB agonist 7,8-dihydroxyflavone (7,8-DHF)48) into the DG and CA3, but not CA1, promoted rapid and sustained antidepressant effects in the rat learned helplessness model of depression35,49, thus implicating the BDNF-TrkB signal pathway in the DG, and CA3, but not CA1, in the antidepressant action of BDNF or TrkB agonist. This is consistent with decreased BDNF protein levels seen in the PFC, DG, CA3, but not CA1, in rat learned helplessness model35,36. In the present study, we found that combination of brexpiprazole and fluoxetine could attenuate decreased BDNF-TrkB signaling in the PFC, CA3, and DG from susceptible mice after social defeat stress. We also reported that 7,8-DHF promoted a rapid antidepressant effect in social defeat stress model39. Therefore, it is likely that combination of brexpiprazole and fluoxetine might promote a rapid antidepressant effect by stimulating BDNF-TrkB pathway in these regions.
The ventral tegmental area (VTA)-NAc pathway plays a critical role in the depression-phenotype35,36,38,39,41,50. We reported that inflammation, social defeat stress and learned helplessness caused an increased BDNF-TrkB signaling within the NAc35,36,37,38,39. Thus, social defeat stress causes decreased BDNF-TrkB signaling in the hippocampus and PFC, but an increased BDNF-TrkB signaling in the NAc, resulting in depression-like behavior in mice. Interestingly, we found that combination of brexpiprazole and fluoxetine could attenuate the alterations in the BDNF-TrkB signaling in the PFC, hippocampus as well as NAc. In contrast, we also found that ketamine did not alter the increased levels of BDNF in the NAc from susceptible mice after social defeat stress37,39, suggesting that ketamine can induce the rapid and long-lasting antidepressant effects by increasing BDNF in the PFC and hippocampus, but not in NAc. It is noteworthy that combination of brexpiprazole and fluoxetine could improve alterations in the BDNF-TrkB signaling in the NAc from susceptible mice after social defeat stress.
Changes in dendritic length and spines density in the PFC and hippocampus are thought to contribute to the neurobiology of depression, and antidepressant treatment is mediated, in part, by blocking or reversing these changes51,52,53. Recently, we reported that ketamine or R-ketamine showed a rapid antidepressant activity by normalizing altered dendritic spines in the PFC and hippocampus, but not NAc37,39. In addition, we also reported that ketamine did not show antidepressant effect in depression-like behavior induced by increased BDNF-TrkB signaling in the NAc after methamphetamine withdrawal34). Together, it is likely that NAc may not be involved in the antidepressant effect of ketamine. A single administration of 7,8-DHF or ANA-12 could normalize alterations in spines density in the social defeat stress model by stimulation at TrkB in the PFC, CA3, and DG, as well as blockade of TrkB in the NAc54. Therefore, it is likely that combination of brexpiprazole and fluoxetine could act by normalizing altered dendritic spines density in regions such as PFC, hippocampus, and NAc. Accordingly, it is likely that, contrary to ketamine, BDNF-TrkB signaling in NAc might be necessary to mediate the antidepressant effect of the combination brexpiprazole plus fluoxetine, although further studies are needed.
In conclusion, this study shows that adjunction of brexpiprazole to fluoxetine can produce a rapid antidepressant effect in the social defeat stress model of depression and that BDNF-TrkB signaling plays a role in the rapid antidepressant action of such combination therapy. Therefore, it is likely that adjunction of brexpiprazole to SSRI could produce a rapid antidepressant effect in treatment-resistant patients with MDD, but without the ketamine-induced psychotomimetic effects and abuse potential.
Methods and Materials
Animals
Male adult C57BL/6 mice 8 weeks old weighing 20–25 g and male adult CD1 mice aged 13–15 weeks (body weight >40 g) were purchased from SLC Japan (Hamamatsu, Shizuoka, Japan). The mice were housed in clear polycarbonate cages (22.5 × 33.8 × 14.0 cm) in groups of 4 or 5 individuals under a controlled 12/12-h light–dark cycle (light from 7:00 AM to 7:00 PM), with the room temperature kept at 23 ± 1 °C and humidity at 55 ± 5%. The mice were given free access to water and food pellets specifically designed for mice. All experiments were carried out in accordance with the Guide for Animal Experimentation of Chiba University. The protocol was approved by the Chiba University Institutional Animal Care and Use Committee.
Drugs and drug administration
Brexpiprazole was synthesized at Otsuka Pharmaceutical Co., Ltd. (Tokyo, Japan). Vehicle (0.5% CMC; 10 ml/kg), fluoxetine (10 mg/kg, Wako Chemical Co., Ltd, Tokyo, Japan), brexpiprazole (0.1 mg/kg), or fluoxetine (10 mg/kg) plus brexpiprazole (0.1 mg/kg) were administered orally into mice. To study the role of BDNF-TrkB signaling in the mechanism of fluoxetine plus brexpiprazole, vehicle (17% dimethyl sulfoxide (DMSO) in phosphate-buffered saline) or ANA-12 (N-[2-[[(Hexahydro-2-oxo-1H-azepin-3-yl) amino] carbonyl] phenyl]-benzo[b]thiophene-2-carboxamide; 0.5 mg/kg, Maybridge, Loughborough, Leicestershire, UK)) was administrated intraperitoneally (i.p.) into mice 30 min before drug administration. The doses of brexpiprazole (0.1 mg/kg), fluoxetine (10 mg/kg) and ANA-12 (0.5 mg/kg) were selected as reported previously11,13,15,16,17,38,39,40,41. Other chemicals were purchased from commercial sources.
Social defeat procedure
The procedure of social defeat stress was performed as previously reported37,39,41,54,55,56,57. Every day the C57BL/6 mice were exposed to a different CD1 aggressor mouse for 10 min, total for 10 days. When the social defeat session ended, the resident CD1 mouse and the intruder mouse were housed in one half of the cage separated by a perforated Plexiglas divider to allow visual, olfactory and auditory contact for the remainder of the 24- h period. At 24 h after the last session, all mice were housed individually. On day 11, a social avoidance test was performed to identify subgroups of mice that were susceptible and unsusceptible to social defeat stress. This was accomplished by placing mice in an interaction test box (42 × 42 cm) with an empty wire-mesh cage (10 × 4.5 cm) located at one end. The movement of the mice was tracked for 2.5 min, followed by 2.5 min in the presence of an unfamiliar aggressor confined in the wire-mesh cage. The duration of the subject’s presence in the ‘interaction zone’ (defined as the 8-cm-wide area surrounding the wire-mesh cage) was recorded by a stopwatch. The interaction ratio was calculated as the time spent in the interaction zone with an aggressor/time spent in the interaction zone without an aggressor. An interaction ratio of 1 was set as the cutoff: mice with scores <1 were defined as ‘susceptible’ to social defeat stress and those with scores ≥1 were defined as ‘unsusceptible’. Only susceptible mice were used in the subsequent experiments.
Behavioral tests of antidepressant effects
Behavioral tests were performed as previously reported37,39,41,54,55,56,57.
Locomotion (LMT)
Mice were placed in experimental cages (L560 × W560 × H330 mm), and locomotor activity was counted by the SCANET MV-40 (MELQUEST, Toyama, Japan). The cumulative locomotor activity was recorded for 60 min. All cages were cleaned between test sessions.
Tail suspension test (TST)
A small piece of adhesive tape was afixed 2 cm from the tip of the tail and punched with a single hole through which mice were hung individually, on a hook. The immobility time of each mouse was recorded for 10 min. Mice were considered immobile only when they hung passively and completely motionless. The TST were performed 2 h after the LMT.
Forced swimming test (FST)
Animals were tested in an automated forced-swim apparatus using SCANET MV-40 (MELQUEST Co., Ltd., Toyama, Japan). The mice were placed individually in a cylinder (Diameter 23 cm; Height 21 cm), containing 15 cm of 23 ± 1 °C warm water. Immobility time was calculated by subtracting active time from total time, using the apparatus analysis software. Cumulative immobility time was scored for 6 min during the test. The TST and FST were performed 2 and 4 h after the LMT, respectively.
Sucrose preference test (SPT)
Mice were exposed to water and 1% sucrose solution for 48 h, followed by 4 h of water and food deprivation and a 1 h exposure to two identical bottles, one containing water and the other 1% sucrose solution. These bottles were weighed before and at the end of the 1 h test period and the sucrose preference (%) was determined.
Western blot analysis of BDNF, and its precursor proBDNF, TrkB, and phosphorylated-TrkB
Western blot analysis was performed as reported previously37,39,41,54,55,56,57. Mice were killed by cervical dislocation and brains were rapidly removed from the skull. Approximately 1-mm-thick coronal sections were cut and bilateral tissue punches of prefrontal cortex (PFC), nucleus accumbens (NAc), striatum, CA1, CA3, and dentate gyrus (DG) of the hippocampus were dissected on ice using a SZ-LED Kenis light microscope (Osaka, Japan), and stored at −80 °C. Basically, tissue samples were homogenized in Laemmli lysis buffer. Aliquots (20 μg) of protein were measured using the DC protein assay kit (Bio-Rad), and incubated for 5 min at 95 °C, with an equal volume of 125 mM Tris-HCl, pH 6.8, 20% glycerol, 0.1% bromophenol blue, 10% β-mercaptoethanol, 4% SDS, and subjected to SDS polyacrylamide gel electrophoresis using AnyKD minigels (Mini-PROTEAN TGX Precast Gel; BioRad). Proteins were transferred onto PVDF membranes using a Trans Blot Mini Cell (Bio-Rad). For immunodetection, the blots were blocked with 2% BSA in TBST (TBS + 0.1% Tween-20) for 1 h at room temperature, and kept with primary antibodies overnight at 4 °C. The following primary antibodies were used: BDNF (1:200; H-117, Cat#: sc-20981, Santa Cruz Biotechnology), phosphor-TrkB (Tyr-706) (1:200; Cat#: sc135645, Santa Cruz Biotechnology), TrkB (80E3) (1:1,000; Cat#: 4603, Cell Signaling Technology). The next day, blots were washed three times in TBST, and incubated with horseradish peroxidase-conjugated anti-rabbit antibody (1:10,000) 1 hour at room temperature. After a final three washes with TBST, bands were detected using enhanced chemiluminescence (ECL) plus the Western Blotting Detection system (GE Healthcare Bioscience). The blots then were washed three times in TBST and incubated with the primary antibody directed against β-actin (1:10,000; Sigma-Aldrich). Images were captured with a Fuji LAS3000-mini imaging system (Fujifilm, Tokyo, Japan), and immunoreactive bands were quantified.
Golgi Staining
Golgi staining was performed using the FD Rapid GolgiStainTM Kit (FD Neuro Technologies, Inc., Columbia, MD), following the manufacturer’s instructions34,38. Twenty four hours after oral administration of vehicle (10 ml/kg), fluoxetine (10 mg/kg), brexpiprazole (0.1 mg/kg), or fluoxetine (10 mg/kg) plus brexpiprazole (0.1 mg/kg), animals were deeply anesthetized with sodium pentobarbital, and brains were removed from the skull and rinsed in double distilled water. Brains were immersed in the impregnation solution, made by mixing equal volumes of Solution A and B, overnight and then stored in fresh solution, for 2 weeks in the dark. Brains were transferred into Solution C overnight and then stored in fresh solution at 4 °C for 1 week, in the dark. Coronal brain sections (100 μm thickness) were cut on a cryostat (3050 S, Leica Microsystems AG, Wetzlar, Germany), with the chamber temperature set at −20 °C. Each section was mounted in Solution C, on saline-coated microscope slides. After absorption of excess solution, sections were dried naturally, at room temperature. Dried sections were processed following the manufacturer’s instructions. Briefly, images of dendrites within CA1, CA3, and DG of the hippocampus, prelimbic (PrL) and inflalimbic (IL) areas of medial PFC (mPFC), and shell and core of NAc were captured using a 100x objective with a Keyence BZ-9000 Generation II microscope (Osaka, Japan). Spines were counted along CA1, CA3, DG, PrL and IL of mPFC and NAc dendrites starting from their point of origin from the primary dendrite, as previously reported34,38. For spine density measurements, all clearly evaluable areas containing 50–100 μm of secondary dendrites from each imaged neuron were used. To determine relative spine density, spines on multiple dendritic branches from a single neuron were counted to obtain an average spine number per 10 μm. For spine number measurements, only spines that emerged perpendicular to the dendritic shaft were counted. Three neurons per section, three sections per animal and six animals were analyzed. The average value for each region, in each individual was obtained. These individual averages were then combined to yield a grand average for each region.
Statistical Analysis
The data show as the mean ± standard error of the mean (S.E.M.). Analysis was performed using PASW Statistics 20 (formerly SPSS Statistics; SPSS). Comparisons between groups were performed using the one-way analysis of variance (ANOVA), followed by post hoc Fisher’s Least Significant Difference (LSD) tests. The P values of less than 0.05 were considered statistically significant.
Additional Information
How to cite this article: Ma, M. et al. Adjunctive treatment of brexpiprazole with fluoxetine shows a rapid antidepressant effect in social defeat stress model: Role of BDNF-TrkB signaling. Sci. Rep. 6, 39209; doi: 10.1038/srep39209 (2016).
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
Barbee, J. G., Conrad, E. J. & Jamhour, N. J. The effectiveness of olanzapine, risperidone, quetiapine, and ziprasidone as augmentation agents in treatment-resistant major depressive disorder. J Clin Psychiatry. 65, 975–981 (2004).
Nelson, J. C. & Papakostas, G. I. Atypical antipsychotic augmentation in major depressive disorder: a meta-analysis of placebo-controlled randomized trials. Am J Psychiatry. 166, 980–991 (2009).
Papakostas, G. I. et al. Aripiprazole augmentation of selective serotonin reuptake inhibitors for treatment-resistant major depressive disorder. J Clin Psychiatry. 66, 1326–1330 (2005).
Papakostas, G. I. et al. Augmentation of antidepressants with atypical antipsychotic medications for treatment-resistant major depressive disorder: a meta-analysis. J Clin Psychiatry. 68, 826–831 (2007).
Shelton, R. C. & Papakostas, G. I. Augmentation of antidepressants with atypical antipsychotics for treatment-resistant major depressive disorder. Acta Psychiatr Scand. 117, 253–259 (2008).
Brunner, E. Efficacy and safety of olanzapine/fluoxetine combination vs fluoxetine monotherapy following successful combination therapy of treatment-resistant major depressive disorder. Neuropsychopharmacology. 39, 2549–2559 (2014).
Papakostas, G. I. et al. Ziprasidone augmentation of escitalopram for major depressive disorder: efficacy results From a randomized, double-blind, placebo-controlled study. Am J Psychiatry. 172, 1251–1258 (2015).
Abdallah, C. G. et al. Ketamine and rapid-acting antidepressants: a window into a new neurobiology for mood disorder therapeutics. Annu Rev Med. 66, 509–523 (2015).
Newport, D. J. et al. Ketamine and other NMDA antagonists: early clinical trials and possible mechanisms in depression. Am J Psychiatry. 172, 950–966 (2015).
Kishimoto, T. et al. Single-dose infusion ketamine and non-ketamine N-methyl-D-aspartate receptor antagonists for unipolar and bipolar depression: a meta-analysis of efficacy, safety and time trajectories. Psychol Med. 46, 1459–1472 (2016).
Rogóz, Z. Combined treatment with atypical antipsychotics and antidepressants in treatment-resistant depression: preclinical and clinical efficacy. Pharmacol Rep. 65, 1536–1544 (2013).
Blier, P. Rational site-directed pharmacotherapy for major depressive disorder. Int J Neuropsychopharmacol. 17, 997–1008 (2014).
Maeda, K. et al. Brexpiprazole I: in vitro and in vivo characterization of a novel serotonin-dopamine activity modulator. J Pharmacol Exp Ther. 350, 589–604 (2014).
Ishima, T. et al. Potentiation of neurite outgrowth by brexpiprazole, a novel serotonin-dopamine activity modulator: a role for serotonin 5-HT1A and 5-HT2A receptors. Eur Neuropsychopharmacol. 25, 505–511 (2015).
Maeda, K. et al. Brexpiprazole II: antipsychotic-like and procognitive effects of a novel serotonin-dopamine activity modulator. J Pharmacol Exp Ther. 350, 605–614 (2014).
Yoshimi, N. et al. Effects of brexpiprazole, a novel serotonin-dopamine activity modulator, on phencyclidine-induced cognitive deficits in mice: a role for serotonin 5-HT1A receptors. Pharmacol Biochem Behav. 124, 245–249 (2014).
Yoshimi, N., Futamura, T. & Hashimoto, K. Improvement of dizocilpine-induced social recognition deficits in mice by brexpiprazole, a novel serotonin-dopamine activity modulator. Eur Neuropsychopharmacol. 25, 356–364 (2015).
Citrome, L. Brexpiprazole: a new dopamine D2receptor partial agonist for the treatment of schizophrenia and major depressive disorder. Drugs Today (Barc). 51, 397–414 (2015).
Citrome, L., Stensbøl, T. B. & Maeda, K. The preclinical profile of brexpiprazole: what is its clinical relevance for the treatment of psychiatric disorders? Expert Rev Neurother. 5, 1219–1229 (2015).
Correll, C. U. Efficacy and safety of brexpiprazole for the treatment of acute schizophrenia: A 6-week randomized, double-blind, placebo-controlled trial. Am J Psychiatry. 172, 870–880 (2015).
Das, S. et al. Brexpiprazole: so far so good. Ther Adv Psychopharmacol. 6, 39–54 (2016).
Kane, J. M. et al. A multicenter, randomized, double-blind, controlled phase 3 trial of fixed-dose brexpiprazole for the treatment of adults with acute schizophrenia. Schizophr Res. 164, 127–135 (2015).
McKeage, K. Adjunctive brexpiprazole: A review in major depressive disorder. CNS Drugs. 30, 91–99 (2016).
Stahl, S. M. Mechanism of action of brexpiprazole: comparison with aripiprazole. CNS Spectr. 21, 1–6 (2016).
Thase, M. E. et al. Efficacy and safety of adjunctive brexpiprazole 2 mg in major depressive disorder: a phase 3, randomized, placebo-controlled study in patients with inadequate response to antidepressants. J Clin Psychiatry. 76, 1224–1231 (2015).
Thase, M. E. et al. Efficacy and safety of adjunctive brexpiprazole 2 mg in major depressive disorder: a phase 3, randomized, placebo-controlled study in patients with inadequate response to antidepressants. J Clin Psychiatry. 76, 1232–1240 (2015).
Nestler, E. J. et al. Neurobiology of depression. Neuron. 34, 13–25 (2002).
Hashimoto, K., Shimizu, E. & Iyo, M. Critical role of brain-derived neurotrophic factor in mood disorder. Brain Res Brain Res Rev. 45, 104–114 (2004).
Duman, R. S. & Monteggia, L. M. A neurotrophic model for stress-related mood disorders. Biol Psychiatry. 59, 1116–1127 (2006).
Hashimoto, K. Brain-derived neurotrophic factor as a biomarker for mood disorders: An historical overview and future directions. Psychiatry Clin Neurosci. 64, 341–357 (2010).
Hashimoto, K. Sigma-1 receptor chaperone and brain-derived neurotrophic factor: Emerging links between cardiovascular disease and depression. Prog Neurobiol. 100, 15–29 (2013).
Castrén, E. Neurotrophins and psychiatric disorders. Handb Exp Pharmacol. 220, 461–479 (2014).
Zhang, J. C., Yao, W. & Hashimoto, K. Brain-derived neurotrophic factor (BDNF) – TrkB signaling in inflammation-related depression and potential therapeutic targets. Curr Neuropharmacol. 14, 721–731 (2016).
Ren, Q. et al. BDNF-TrkB signaling in the nucleus accumbens shell of mice has key role in methamphetamine withdrawal symptoms. Transl Psychiatry. 5, e666 (2015).
Shirayama, Y. et al. Alterations in brain-derived neurotrophic factor (BDNF) and its precursor proBDNF in the brain regions of a learned helplessness rat model and the antidepressant effects of a TrkB agonist and antagonist. Eur Neuropsychopharmacol. 25, 2449–2458 (2015).
Yang, C. et al. Regional differences in brain-derived neurotrophic factor levels and dendritic spine density confer resilience to inescapable stress. Int J Neuropsychopharmacol. 18, pyu121 (2015).
Yang, C. et al. R-ketamine: A rapid-onset and sustained antidepressant without psychotomimetic side effects. Transl Psychiatry. 5, e632 (2015).
Zhang, J. C. et al. Antidepressant effects of TrkB ligands on depression-like behavior and dendritic changes in mice after inflammation. Int J Neuropsychophamacol. 18, pyu077 (2015).
Zhang, J. C. et al. Comparison of ketamine, 7,8-dihydroxyflavone, and ANA-12 antidepressant effects in the social defeat stress model of depression. Psychopharmacology (Berl). 232, 4325–4335 (2015).
Cazorla, M. et al. Identification of a low–molecular weight TrkB antagonist with anxiolytic and antidepressant activity in mice. J Clin Invest. 121, 1846–1857 (2011).
Yang, B. et al. Comparison of R-ketamine and rapastinel antidepressant effects in the social defeat stress model of depression. Psychopharmacology (Berl). 233, 3647–3657 (2016).
Björkholm, C. et al. Ketamine-like effects of a combination of olanzapine and fluoxetine on AMPA and NMDA receptor-mediated transmission in the medial prefrontal cortex of the rat. Eur Neuropsychopharmacol. 25, 1842–1847 (2015).
Autry, A. E. et al. NMDA receptor blockade at rest triggers rapid behavioural antidepressant responses. Nature. 475, 91–95 (2011).
Lepack, A. E. et al. BDNF release is required for the behavioral actions of ketamine. Int J Neuropsychopharmacol. 18, pyu033 (2014).
Liu, W. X. et al. Regulation of glutamate transporter 1 via BDNF-TrkB signaling plays a role in the anti-apoptotic and antidepressant effects of ketamine in chronic unpredictable stress model of depression. Psychopharmacology (Berl). 233, 405–415 (2016).
Sun, H. L. et al. Role of hippocampal p11 in the sustained antidepressant effect of ketamine in the chronic unpredictable mild stress model. Transl Psychiatry. 6, e741 (2016).
Yang, B. et al. Regional differences in the expression of brain-derived neurotrophic factor (BDNF) pro-peptide, proBDNF, and preproBDNF in the brain confer stress resilience. Eur Arch Psychiatry Clin Neurosci., 266, 765–769 (2016) doi: 10.1007/s00406-016-0693-6.
Jang, S. W. et al. A selective TrkB agonist with potent neurotrophic activities by 7,8-dihydroxyflavone. Proc Natl Acad Sci USA 107, 2687–2692 (2010).
Shirayama, Y. et al. Brain-derived neurotrophic factor produces antidepressant effects in behavioral models of depression. J Neurosci. 22, 3251–3261 (2002).
Nestler, E. J. & Carlezon, W. A. Jr. The limbic dopamine reward circuit in depression. Biol Psychiatry. 59, 1151–1159 (2006).
Duman, R. S. & Aghajanian, G. K. Synaptic dysfunction in depression: Potential therapeutic targets. Science. 338, 68–72 (2012).
Ohgi, Y., Futamura, T. & Hashimoto, K. Glutamate signaling in synaptogenesis and NMDA receptors as potential therapeutic targets for psychiatric disorders. Curr Mol Med. 15, 206–221 (2015).
McEwen, B. S. Physiology and neurobiology of stress and adaptation: central role of the brain. Physiol Rev. 87, 873–904 (2007).
Ren, Q. et al. 7,8-Dihydroxyflavone, a TrkB agonist, attenuates behavioral abnormalities and neurotoxicity in mice after administration of methamphetamine. Psychopharmacology (Berl). 231, 159–166 (2014).
Berton, O. et al. Essential role of BDNF in the mesolimbic dopamine pathway in social defeat stress. Science. 311, 864–868 (2006).
Golden, S. A. et al. A standardized protocol for repeated social defeat stress in mice. Nat Protoc. 6, 1183–1191 (2011).
Ren, Q. et al. Gene deficiency and pharmacological inhibition of soluble epoxide hydrolase confers resilience to repeated social defeat stress. Proc Natl Acad Sci USA 113, E1944–E1952 (2016).
Acknowledgements
The authors would like to thank Prof. Toshinori Nakayama (Department of Immunology, Chiba University Graduate School of Medicine) for the use of Keyence BZ-X700 Generation II microscope. Ms. Min Ma was supported by the Nurture of Creative Research Leaders in Immune System Regulation and Innovative Therapeutics Program of Chiba University. Dr. Qian Ren and Dr. Chun Yang were supported by Research Fellowship of the Japan Society for the Promotion of Science (Tokyo, Japan). Dr. Wei Yao was supported by Ishidsu Shun Memorial Scholarship (Tokyo, Japan). Dr. Chao Dong was supported by the Uehara Research Foundation (Tokyo, Japan). This study was supported by the research grant from Otsuka Pharmaceutical Co, Ltd. (Tokyo, Japan), and a Grant-in-Aid for Scientific Research on Innovative Areas of the Ministry of Education, Culture, Sports, Science and Technology, Japan (to K.H.).
Author information
Authors and Affiliations
Contributions
K.H. designed the study and wrote the protocol. M.M., Q.R., C.Y., J.C.Z., W.Y. and C.D. performed all the pharmacological experiments. Y.O. and T.F. contributed the new compound brexpiprazole. M.M. and Q.R. undertook the statistical analysis. M.M., Q.R. and K.H. wrote the first draft of the manuscript. All authors contributed to and have approved the final manuscript.
Ethics declarations
Competing interests
Dr. Kenji Hashimoto received research support from Dainippon-Sumitomo, Mochida, Otsuka, and Taisho. Dr. Ohigi and Dr. Futamura are employees of Otsuka Pharmaceutical Co, Ltd. The other authors declare no conflict of interest.
Electronic supplementary material
Rights and permissions
This work is licensed under a Creative Commons Attribution 4.0 International License. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in the credit line; if the material is not included under the Creative Commons license, users will need to obtain permission from the license holder to reproduce the material. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/
About this article
Cite this article
Ma, M., Ren, Q., Yang, C. et al. Adjunctive treatment of brexpiprazole with fluoxetine shows a rapid antidepressant effect in social defeat stress model: Role of BDNF-TrkB signaling. Sci Rep 6, 39209 (2016). https://doi.org/10.1038/srep39209
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/srep39209
This article is cited by
-
Involvement of PI3K/AKT Pathway in the Rapid Antidepressant Effects of Crocetin in Mice with Depression-Like Phenotypes
Neurochemical Research (2024)
-
Molecular and neurocircuitry mechanisms of social avoidance
Cellular and Molecular Life Sciences (2021)
-
Stress-primed secretory autophagy promotes extracellular BDNF maturation by enhancing MMP9 secretion
Nature Communications (2021)
-
Brain-derived neurotrophic factor-TrkB signaling in the medial prefrontal cortex plays a role in the anhedonia-like phenotype after spared nerve injury
European Archives of Psychiatry and Clinical Neuroscience (2020)
-
YL-0919, a dual 5-HT1A partial agonist and SSRI, produces antidepressant- and anxiolytic-like effects in rats subjected to chronic unpredictable stress
Acta Pharmacologica Sinica (2018)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.