Abstract
Bacterial pathogens possess complex type III effector (T3E) repertoires that are translocated inside the host cells to cause disease. However, only a minor proportion of these effectors have been assigned a function. Here, we show that the T3E AWR5 from the phytopathogen Ralstonia solanacearum is an inhibitor of TOR, a central regulator in eukaryotes that controls the switch between cell growth and stress responses in response to nutrient availability. Heterologous expression of AWR5 in yeast caused growth inhibition and autophagy induction coupled to massive transcriptomic changes, unmistakably reminiscent of TOR inhibition by rapamycin or nitrogen starvation. Detailed genetic analysis of these phenotypes in yeast, including suppression of AWR5-induced toxicity by mutation of CDC55 and TPD3, encoding regulatory subunits of the PP2A phosphatase, indicated that AWR5 might exert its function by directly or indirectly inhibiting the TOR pathway upstream PP2A. We present evidence in planta that this T3E caused a decrease in TOR-regulated plant nitrate reductase activity and also that normal levels of TOR and the Cdc55 homologues in plants are required for R. solanacearum virulence. Our results suggest that the TOR pathway is a bona fide T3E target and further prove that yeast is a useful platform for T3E function characterisation.
Similar content being viewed by others
Introduction
Many bacterial pathogens use a type III secretion system (T3SS) to inject a suite of proteins inside the host cell1. These proteins are referred to as type III effectors (T3Es) and play a central role in bacterial survival and disease development2. T3Es manipulate host cell pathways by mimicking key host proteins or mediating changes in their subcellular localization, by targeting plant-specific transcription factors, by inhibiting translation and metabolic stress pathways or exploiting a specific form of host-mediated fatty acid modification3,4,5. The functional study of T3Es from phytopathogenic bacteria has raised a tremendous interest in the last years6,7. The number of T3Es identified is growing at a very fast pace as more bacterial genomes become available, revealing complex repertoires that feature internal redundancy, which complicates their study6. However, only in a few cases the function of this kind of effectors in planta has been identified.
Heterologous production in Saccharomyces cerevisiae has offered promising and effective strategies to characterize bacterial T3Es8. Seminal work with YopE showed that this T3E caused specific growth inhibition and cytoskeletal alteration, an activity conserved in yeast and mammalian cells9. Functional analyses of plant-associated T3E in yeast have revealed other effector-triggered phenotypes including cell death, suppression of apoptosis or perturbation of host cellular processes, such as MAPK signalling or sphingolipid synthesis10,11,12. All these findings strengthen the premise that many bacterial T3E target universal eukaryotic processes so thatS. cerevisiae can be exploited to elucidate their molecular function and to investigate target-effector interactions8,13.
The TOR complex 1 (TORC1) is a central regulator of cell growth in response to nutrient availability and stress conditions by controlling diverse cellular processes, including transcriptional activation, ribosome biogenesis or autophagy14 (Fig. 1a). This complex contains the Tor1 or Tor2 protein kinases and can be inhibited by the drug rapamycin. In yeast, TORC1 acts by controlling three major cell components: the kinase Sch9, Tap42 and its associated phosphatases and the ATG1 complex14,15. Thus, TORC1 modulates nitrogen catabolite repression and diverse stress responses by controlling the activity of several phosphatases, such as protein phosphatase 2A (PP2A) or Sit 4, often by modifying their interaction with regulatory subunits (Fig. 1a15).
Expression of awr5 effector inhibits yeast growth.
(a) Schematic view of theSaccharomyces cerevisiae TORC1-regulated pathways. The TORC1 complex is a central growth regulator, controlling the balance between growth and quiescence. Continuous and dotted lines represent, respectively, signaling events regulated by active and inactive TORC1. (b) Growth on solid medium of yeast strains expressing awr effectors. Yeast strains bearing awr genes fused to GFP tag or a GFP control were subjected to serial 10-fold dilutions and spotted onto solid SD-Ura+doxycycline (repressing medium) and SD-Ura (inducing medium). Photographs were taken after 2 days of growth. (c) Growth kinetics in liquid medium of yeast cells harboring awr5 or a GFP control. Yeast cells harboring awr5 or a GFP control were grown in SD-Ura+dox (−AWR5) and SD-Ura (+AWR5) liquid media and dispersed on SD-Ura+dox plates. The logarithm of colony forming units (CFU) per ml is shown over time. Error bars indicate standard errors for 2 biological replicates. (d) Immunoblot analysis of AWR5 protein levels. Total protein was extracted from cultures shown in Fig. 1c and immunoblotted using an anti-GFP antibody. The black arrowhead indicates AWR5-GFP protein. All experiments were performed at least three times, with similar results.
Ralstonia solanacearum is emerging as a model system to study plant-pathogen molecular interactions and T3E function16. This soil-borne bacterium is the causing agent of bacterial wilt, a disease caused when the bacterium growing in plant extracellular spaces (apoplast) infects the xylem vessels, where it multiplies extensively and blocks water flow17. R. solanacearum has been ranked as the second most important bacterial plant pathogen18, due to its high persistence and wide geographical distribution and host range, as it infects more than 200 plant species, including important agricultural crops such as tomato and potato19. Of more than 70 T3Es identified in the reference strain GMI1000, only for two of them a defined rolein planta has been assigned16. AWRs (named after a conserved alanine-tryptophan-arginine tryad and also called RipAs) are one of the multigenic families of T3Es conserved in all R. solanacearum strains17, with orthologues in other bacterial pathogens such as Xanthomonas strains, Acidovorax avenae or Burkholderia spp.20. A low protein similarity has also been described between AWRs and the Xanthomonas oryzae pv. oryzae effector XopZ, which was shown to be involved in virulence and suppression of host basal defence21. Translocation assays have proven AWRs asbona fide R. solanacearum type III secreted effectors20,22,23. However, sequence information on AWR proteins gives no clue on their putative function. In a previous study, we showed that the AWR T3E family collectively contributes to R. solanacearum virulence, as a mutant bacterium devoid of all AWR multiplies 50-fold less than the wild-type strain on eggplant and tomato plants. Functional analysis of AWRs also demonstrated that their expression in different plant species triggers varying defence responses20. Functional analyses for each AWR showed that AWR5 had an important contribution in virulence and also caused the most dramatic plant responses. In addition, we have recently found that awr5 is one of the most highly expressed genes whenR. solanacearum grows inside the plant host (Marina Puigvert, unpublished results). Association genetics combining genomic data from R. solanacearum strains and their pathogenicity on eggplant, pepper and tomato accessions identified AWR5 amongst the three T3Es highly associated to virulence24.
In this work we take advantage of the yeast system to characterize AWR5 function. Heterologous expression of AWR5 inS. cerevisiae resulted in dramatic growth inhibition of yeast cells. We show that this effect on yeast growth is caused by inhibition of the central regulatory TOR pathway. Importantly, AWR5 impact on the TOR pathway is conserved in both yeast and plants, revealing a previously unknown T3E mode of action maintained in evolutionary distant organisms. Moreover, our work further validates yeast as an excellent platform to uncover T3E function.
Results
Expression of the R. solanacearum awr type III effector family in yeast causes growth inhibition
To investigate the function of the AWR bacterial effectors in eukaryotic cells, we expressed the five awr genes fromR. solanacearum GMI1000 in S. cerevisiae. In a first step, awrs were cloned in the high-copy-number vector pAG426GAL, where they are transcribed from the strong galactose-inducible GAL1 promoter. The resulting plasmids were introduced in yeast and the transformed strains grown overnight, then serially diluted and plated either in repressing media (glucose) or inducing media (galactose). It was observed that, except for AWR4, these effectors inhibited growth to different extents, as observed by the inability to form macroscopic colonies on inducing media (Supplementary Fig. S1). AWR1, 2, 3 and 5 caused a strong toxicity upon induction, but AWR5 showed the most dramatic effect, inhibiting yeast growth even in non-inducing conditions. The phenotype seemed specific for AWR effectors, as it was not observed when a control gene (GFP) was expressed (Supplementary Fig. S1). The full-length AWR5 protein was required for functionality, as expression of split variants of AWR5 (N-terminal or C-terminal halves, or the central region) did not cause toxicity (Supplementary Fig. S1).
To evaluate the phenotype in more physiological conditions and ensure construct stability and tight control of effector transcription, we integrated the bacterial genes in the yeast genome under the control of a repressible Tet-Off promoter. When the resulting strains bearing awrs or a control GUS gene were plated in the absence of the repressor doxycycline, only expression of awr5 reproduced the dramatic growth arrest (Fig. 1b). The absence of toxicity for AWR1, 2 and 4 could not be attributed to a lack of expression, as the full-length proteins were readily detected in yeast cells (Supplementary Fig. S2). Thus, we concentrated on the characterization of the growth inhibition caused by awr5 expression.
Characterization of the AWR5-dependent growth inhibition phenotype
Yeast growth inhibition was also apparent upon AWR5 production in liquid cultures as indicated by a rapid stagnation of cell density over time (not shown) and a clear decrease in the number of growing cells (Fig. 1c). Growth inhibition kinetics paralleled with an increase in awr5 RNA (Supplementary Fig. S3) and protein levels (Fig. 1d). Microscopic observation of strains producing AWR5 revealed the presence of budding cells at similar proportions to cells not producing the bacterial effector (Supplementary Fig. S4a). Thus, it could be ruled out that this protein specifically alters the cell cycle.
Expression of awr5 caused strong growth inhibition but not cell death, as deduced from methylene blue staining of cells bearing awr5 in the absence of doxycycline (Supplementary Fig. S4a) and from counting of viable cells able to form colonies after 6 h of awr5 expression (Supplementary Fig. S4b). Similarly, growth arrest in cells expressing awr5 was not likely caused by defects in cell wall construction leading to cell lysis, since it was not eliminated by osmotic stabilization with 10% sorbitol (Supplementary Fig. S5). In contrast, determination of cell size upon expression of awr5 showed significant changes, visible after 8 h of induction, with AWR5-producing cells showing an average diameter of 4.96 ± 0.03 μm, while that of non-expressing cells was over 5.3 ± 0.06 μm (Supplementary Fig. S4c).
Previous reports studying effectors from Pseudomonas syringae or Xanthomonas euvesicatoria had shown that some of them caused growth arrest when yeast was forced to respire10,11. To verify if respiration affected AWR5 toxicity in yeast, we grew serial dilutions of the strain producing this protein or a control gene (β-glucuronidase, GUS) onto solid medium containing the non-fermentable carbon sources ethanol and glycerol. As observed in Supplementary Fig. S5, the toxic effect due to AWR5 was maintained under these conditions.
In summary, we established that production of the full-length AWR5 protein in yeast targeted a cellular process leading to growth inhibition and decreased cell size, but not involving an evident cell cycle arrest or cell death.
Expression of awr5 mimics the transcriptional changes induced by the TORC1 inhibitor rapamycin
To understand the molecular basis of awr5 toxicity in yeast and to highlight putative functional targets, we considered the identification of possible changes at the mRNA level caused by expression of the effector. To this end, we carried out a genome-wide transcriptomic analysis using DNA microarrays in yeast cells with awr5 expression induced for 2, 4 and 6 h. This time-course was selected according to the previously characterized growth effect (Fig. 1c). DNA microarray analysis yielded 3763 genes with valid data for all 3 time-points. We observed that induction of awr5 expression produced relevant time-dependent changes in the transcriptomic profile that, in most cases, could be observed after 4 and 6 h of induction. The mRNA level of 766 genes was modified at least 2-fold, with 319 genes induced and 447 repressed. The functional assignment of induced genes revealed a striking excess of genes subjected to nitrogen catabolite repression (NCR)25, such as MEP2, GAP1, DAL5, CPS1 or DUR1,2, whereas among the repressed genes there was a vast excess of genes encoding ribosomal proteins or involved in ribosome biogenesis. This profile was reminiscent of that reported by several laboratories for inhibition of the TORC1 pathway15.
We took advantage of recent work in our laboratory in which the transcriptomic profile in response to 1 h of exposure to rapamycin had been generated26. Combination of this data with that obtained here after awr5 expression yielded 2774 genes with expression information in both conditions. Figure 2a shows the correspondence between changes produced in response to awr5 with those caused by rapamycin. It can be observed that whereas the correlation is relatively poor shortly afterawr5 induction (correlation coefficient = 0.402), the similarity between both responses becomes evident after 4 h and, particularly, after 6 h of awr5 induction (correlation coefficients 0.569 and 0.739, respectively). We then selected among the 766 genes whose expression changed at least 2-fold those with data for the rapamycin treatment (596 genes) and subjected this set of genes to clustering analysis. Figure 2b clearly documents that the time-dependent transcriptional response to expression ofawr5 matches that provoked by rapamycin treatment (correlation coefficient of 0.872 when compared with awr5 data after 6 h of expression). It can be observed that clusters 1 and 2 -and to some extent also cluster 3- are enriched in induced genes related to metabolism of nitrogen (mostly amino acids), whereas regarding the repressed genes, cluster 5 includes genes involved in translation and cluster 6 is enriched in genes encoding ribosomal proteins or members of the RiBi (ribosome biogenesis) regulon. All these results indicate that expression of bacterial awr5 in yeast triggers a response that mimics the inhibition of the TORC1 pathway.
Expression of bacterial awr5 in yeast mimics the transcriptomic changes caused by inhibition of the TORC1 pathway.
(a) Changes in mRNA levels caused by expression of awr5 (X-axis, log2 space) for the set of 2774 genes with valid data for all three time-points were plotted against the corresponding values after 1 h treatment with 200 ng/ml rapamycin (Y axis, log2 space). “CC” figures indicate the calculated correlation coefficient among both sets of data for each time-point. (b) The set of 596 genes presenting at least 2-fold changes in mRNA levels upon expression ofawr5 and with valid data for the rapamycin treatment were clustered (Euclidean distance, average linkage) using Cluster 3.0 software57 and are represented with the Java Treeview software, version 1.1.6r458. Numbers in red denote selected clusters referred to in the main text and number between parentheses designate the p-value for the indicated GO annotations.
These transcriptomic data were validated by performing quantitative RT-PCR analysis on a subset of genes from different TORC1-regulated pathways, which showed altered expression levels in response to awr5 (Fig. 3b). As expected,awr5 expression resulted in a decrease of the levels of the TOR-activated STM1 and NSR1 genes, which are involved in yeast growth27,28. In contrast, the levels of the TOR-repressedGAP1 and MEP2, which control nitrogen catabolite repression29, increased in response to awr5 expression. Similar results were obtained when promoter activity was measured using fusions to the β-galactosidase reporter:awr5 expression resulted in increasedGAP1 and MEP2 promoter output (Fig. 3b).
Transcriptional response of TORC1-related genes to awr5 expression.
(a) qRT-PCR experiments showing relative gene expression of TORC1 downstream targets. Gene expression of nitrogen catabolite repression (NCR)-sensitive GAP1 and MEP2 and ribosomal biogenesis STM1 and NSR1 genes was tested in yeast strains expressing awr5 (+AWR5 (−dox)) 6 hours after induction. Error bars represent standard errors from 2 biological replicates. (b) β-galactosidase activities from yeast cells bearing awr5. Promoter activities of GAP1 and MEP2 were determined 6 hours after growth in SD-Ura+dox (−AWR5) and SD-Ura (+AWR5). Data represent the means and standard errors of 4 independent clones. All assays were repeated at least twice with similar results.
Mutations in two genes involved in the TORC1 pathway rescue the yeast growth inhibition caused by AWR5
Since AWR5 mimicked rapamycin treatment in yeast, we tested whether disruption of FPR1-encoding the rapamycin-binding protein Fpr1 that inhibits the TORC1 kinase in the presence of rapamycin15 rescued the AWR5-triggered phenotype. Growth inhibition caused by AWR5 was maintained in the fpr1 strain (Fig. 4a), indicating that the bacterial effector acts on TORC1 through a different mechanism than rapamycin.
cdc55 and tpd3 mutations affecting PP2A protein phosphatase activity suppress AWR5-induced yeast growth inhibition.
(a) Growth on solid medium of control (WT) and an fpr1 mutant carrying awr5 under the control of a Tet-Off promoter. Serial 10-fold dilutions were spotted onto solid SD-Ura+doxycycline (−AWR5) and SD-Ura (+AWR5). (b) Growth on solid medium of control (WT) and TORC1-related yeast mutants containing plasmid carrying awr5. Serial 10-fold dilutions were spotted onto solid SD-Ura+doxycycline (−AWR5) and SD-Ura (+AWR5). Photographs were taken after 3 days of growth. (c) GAP1 promoter activity from plasmid pGAP1-LacZ in wild-type (WT) and mutantcdc55 yeast cells bearing awr5 or a control gene (GFP). β-galactosidase activity was measured 6 hours after growth in SD-Ura+dox (−AWR5) and SD-Ura (+AWR5). Values represent the means and standard errors of 4 independent clones. All experiments were performed three times with similar results.
In order to ascertain which point of the TOR-controlled pathways was targeted by AWR5 we analysed yeast strains with altered levels of different genes mediating TORC1 signalling. Interestingly, the strains mutated in the PP2A regulatory or scaffold subunits cdc55 or tpd3 did not show AWR5-triggered growth inhibition (Fig. 4b). This indicated that these PP2A subunits are essential for AWR5 to cause its phenotype. These results were also corroborated by testing promoter activity of GAP1 fused to the β-galactosidase reporter in wild type andcdc55 mutant strains. Our results clearly showed that CDC55 was required for the increase in GAP1 promoter activity that occurs in response to awr5 expression (Fig. 4c).
On the contrary, AWR5 did not seem to target the PP2A catalytic subunit, since AWR5-mediated growth inhibition could not be rescued by overexpression or conditional mutation of the two redundant genes (pph21, 22) encoding this subunit (Supplementary Fig. S6a,b). Any other mutant (rts1, tip41, ppm1 and gln3) or overexpressor (SIT4) in genes related to signalling through the TORC1 pathway that we tested did not show reversion of AWR5-mediated growth inhibition. However, we could not detect interaction between Cdc55 or Tpd3 and AWR5 in yeast using co-immunoprecipitation (Supplementary Fig. S7a,b). Although the transcription profile was specifically compatible with TORC1 inhibition, we checked whether AWR5 had any impact on TORC2. As shown in supplementary Fig. S8, AWR5 does not interfere with TORC2, because a dominant active ypk2 mutant (one of the major downstream components of the TORC2 pathway) did not rescue growth inhibition caused by AWR5 (Fig. S8a) and expression of the effector did not alter the actin cytoskeleton, a target of the TORC2 pathway (Fig. S8b). In addition, AWR5 also did not co-immunoprecipitate with the Lst8, a shared component of TORC1 and TORC230 (Supplementary Fig. S7c).
To determine whether Cdc55 was required for downstream AWR5-mediated responses, we carried out a new transcriptomic analysis, in this case by direct sequencing of RNAs (RNA-seq) in wild type and cdc55 cells expressing awr5 for 6 h. Analysis of the wild type strain showed a response congruent with that observed previously using DNA microarrays, with a correlation coefficient of 0.63 in the genes detected as induced by both methodologies (Supplementary Fig. S9). In addition, among the top 25 most induced genes detected by microarray analysis, 13 were also ranked as such by RNA-seq. Comparison of the profiles of the wild type and the cdc55 strains after 6 h of awr5 induction showed that mutation in CDC55 dramatically attenuated the transcriptomic effects caused byawr5 expression. As illustrated in Fig. 5a, 512 genes were induced in the wild type strain upon awr5 expression and only 212 in the cdc55 strain (of which only 144 were also induced in wild type cells). This effect was particularly evident in repressed genes, since the cdc55 mutation affected almost 90% of the genes repressed by awr5 expression in the wild type strain. The attenuation of the transcriptional response to AWR5 could clearly be observed by plotting the 100 genes showing highest induction (Fig. 5b, upper panel) or repression (Fig. 5b, lower panel) in wild-type cells and comparing to their expression in cdc55 cells.
Mutation of CDC55 greatly attenuates AWR5 impact on the yeast transcriptional profile.
(a) Venn diagram showing the number of genes whose expression was considered to be induced (top) or repressed (bottom) by expression of AWR5 in wild-type and cdc55 cells for a set of 5732 genes with valid data for both strains. (b) Plots of the log2 values for the changes in the level of expression induced by expression of AWR5 in both wild-type (open circles) and cdc55 strains for the 100 most upregulated (top) and 100 most downregulated (bottom) genes in the wild-type strain (open circles). Symbols for the expression values for the cdc55 strain are depicted as follows. For the induced genes: open triangles, the NCR family, as defined previously25; the RTG group (open squares) comprises the genes described as documented targets for the Rtg1 or Rtg3 transcription factors as defined in59. Genes not included in these categories are designated as “others” (closed circles). The genes downregulated in the wild-type strain are classified into one of three possible families: Ribi regulon (open squares), ribosomal proteins (open triangles), protein translation (open diamonds) and others (closed circles), as defined in59.
It was apparent that many of the highly induced genes in response to AWR5 expression, which belong to the NCR and the mitochondrial retrograde pathways, decreased their expression in the absence of the regulatory subunit of PP2A. Indeed, 26 out of 28 NCR and RTG genes31 ranking as top 100 induced decreased their expression more than 50% in cdc55 cells. Similarly, a significant number of genes whose expression was decreased in response to AWR5 were clearly no longer repressed in cdc55 cells. However, the effect was not homogeneous. For instance the transcripts showing little or no change in awr5-induced repression upon deletion of CDC55 are largely enriched in genes involved in ribosome biogenesis and rRNA processing (Supplementary Fig. S10). This could be expected, as TOR-regulated expression of these genes is mostly PP2A-independent.
Taken together, these results indicate that the inability to form PP2A complexes containing Cdc55 not only neutralizes the severe growth defect caused by expression of awr5, but also substantially minimizes the transcriptional alterations derived from such expression. These data further supported the notion that the PP2A complex might mediate the phenotype caused by the AWR5 effector.
awr5 expression constitutively activates autophagy
It is known that TORC1 regulates autophagy in yeast via inhibition of the ATG1 complex (Fig. 1a and32). Our microarray data showed that expression ofawr5 increased the expression of diverse autophagy genes, such as ATG8 or ATG14, which indicates activation of this process. In order to confirm whether autophagy was affected by awr5 expression, autophagic flux was monitored in yeast cells constitutively expressing GFP-ATG8 (Fig. 6). Proteolysis of GFP-ATG8 in the vacuole during autophagy results in the accumulation of the GFP moiety. Hence, detection of free GFP levels by western blot analysis can be used as readout of the autophagic rate33. Expression of awr5 led to a dramatic accumulation of GFP in yeast cells, indicating an increased autophagic flux (Fig. 6a). As a control, we subjected yeast cells to nitrogen starvation, which resulted, as expected, in an increase of free GFP levels (Fig. 6b). Interestingly, free GFP levels in awr5-expressing cells relative to GFP-ATG8 were higher than in nitrogen-starved cells, indicating that AWR5 expression induces autophagy more potently than nitrogen starvation does. Next, we tested whether Cdc55 was involved in AWR5-triggered autophagy in yeast. Although GFP-ATG8 levels were slightly higher in cdc55 mutant cells expressing awr5, autophagy was similarly induced in both strains (Fig. 6a). awr5 expression was analysed and similar levels were detected in wild type and cdc55 mutant cells (Fig. 6c). These findings indicated that AWR5-mediated autophagy induction occurs independently of Cdc55 in yeast.
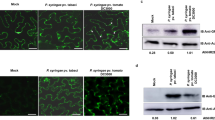
awr5 expression induces constitutive autophagy, independently of Cdc55-PP2A activity.
(a) Immunodetection of GFP-ATG8 processing in wild-type and mutant cdc55 yeast strains expressing awr5. Wild-type (WT) and mutant cdc55 yeast cells bearing awr5 gene were grown in SD-Ura+dox (−AWR5) and SD-Ura (+AWR5). Total protein extracts were immunoblotted using anti-GFP antibody. The black and the empty arrowhead indicate, respectively, GFP-ATG8 fusion protein and cleaved GFP. The asterisk denotes a degradation product of AWR5-GFP protein. (b) Wild-type cells carrying GFP-ATG8 grown in nitrogen-rich (N+) or nitrogen-depleted (N−) medium were used as a control of GFP-ATG8 processing and induction of autophagy in N− conditions. (c) AWR5 protein levels in wild-type and mutantcdc55 yeast cells. Total protein was extracted and immunoblotted using anti-GFP antibody. The black arrow indicates AWR5-GFP protein. All experiments were performed at least three times, with similar results.
AWR5 alters the TOR pathway in plants
Since heterologous expression of a T3E from R. solanacearum in yeast altered the TORC1 pathway, it was plausible that the effector had a similar effect in its natural context, i.e. when translocated inside the cells of plants infected by the pathogen. In plants, it has been shown that TOR silencing results in activation of nitrogen recycling activities and reduces primary nitrogen assimilation, measured by nitrate reductase activity34. In order to test whether awr5 expression resulted in TOR inhibition in plants we thus used this activity as readout. Transient expression of awr5 in Nicotiana benthamiana leaves resulted in a significant reduction of nitrate reductase activity compared to the control (GUS) (Fig. 7a). Leaky expression of awr5 prior to induction may account for the slightly lower nitrate reductase activity values in leaves transformed with awr5.awr5 expression did not significantly affect the activity of the TOR-independent, constitutive enzyme glucose-6-phosphate dehydrogenase (Fig. 7b). This clearly indicates that the decrease in the TOR- dependent nitrate reductase activity is specifically caused by awr5 expression in plants.
Interplay between AWR5 and TOR in planta.
Effect of awr5 transient expression on (a) nitrate reductase (NR) activity or (b) glucose-6-phosphate dehydrogenase (G6PdH) in Nicotiana benthamiana. Full leaves of N. benthamiana were agroinfiltrated with constructs bearingawr5 or a control gene (GUS). Total protein extracts were used to determine NR and G6PdH activity at 0 and 1 hours post-estradiol induction (hpi). Error bars indicate standard errors of 2 biological replicates for NR and 3 for G6PdH. TOR (c) and its signalling component B55 (d) are involved in plant defence responses against R. solanacearum invasion. Five-week old plants grown in Jiffy pots were inoculated with R. solanacearum GMI1000 at an OD600 = 0.1 and wilting symptoms were recorded over time according to a disease index scale (0: no wilting, 1: 25% wilted leaves, 2: 50%, 3: 75%, 4: death). The experiment was repeated twice using at least 20 plants in each. Error bars indicate standard errors.
The mechanisms by which AWR5 alters the TOR pathway in plants remains to be determined. Transient expression of awr5 did not result in autophagy induction in N. benthamiana leaves expressing the autophagy marker GFP:ATG8a (Supplementary Fig. S11a). In addition, we could not detect direct interaction between AWR5 and TOR1 by co-immunoprecipitation using N. benthamiana leaves transiently over-expressing tagged versions of the two proteins (Supplementary Fig. S11b).
To further prove that AWR5 impacts the plant TOR pathway we infected Arabidopsis thaliana wild-type Col-0 plants, TOR1-silenced plants (TOR RNAi)35 and two mutant lines disrupted in the genes encoding either of the CDC55 homologues (b55α and b55β)36 with R. solanacearum and recorded the appearance of wilting symptoms over time. TOR1-silenced lines were slightly more resistant to bacterial infection (Fig. 7c) and the two lines mutated in the CDC55 homologues showed a striking resistance to infection as compared to the wild-type Arabidopsis (Fig. 7d), indicating that AWR5 effector may be targeting the TOR pathway in both plants and yeast. Although TOR RNAi lines have been previously reported to be slightly reduced in growth compared35, in our growing conditions both TOR RNAi and b55 mutants were indistinguishable from wild-type plants (Fig. S12), ruling out the possibility that their altered response to R. solanacearum infection was due to reduced surface of interaction.
Discussion
In this work, we have produced R. solanacearum AWR effectors in yeast and have found that AWR5 impacts the TORC1 pathway, an essential component of eukaryotic cells. The premise for usingSaccharomyces cerevisiae was that this organism carries out most eukaryotic processes and, unlike the host cells where T3E are naturally injected, it shows less gene redundancy and lacks resistance components that counteract and mask effector function37. For instance, gain-of-function analyses of T3E in plants are often hampered by a hypersensitive response (HR), a programmed cell death associated with recognition of effectors or effector virulence activities38.
A number of studies have successfully used S. cerevisiae as a model to identify T3E targets8,13. Toxicity -ranging from growth arrest to cell death- is the most common phenotype observed in these studies. However, this is not a widespread phenomenon when R. solanacearum T3E are expressed in yeast, as only 6 out of 36 effectors representing the repertoire of strain GMI1000 caused substantial growth inhibition (this work and39). Interestingly, four out of the six toxic T3E encode AWR proteins, suggesting a distinct function for this effector family in bacterial-host interactions. Cell growth inhibition caused by T3E has been traced back to interference on vesicle trafficking, disruption of the cytoskeleton or MAP Kinase alteration8, providing important clues on T3E function. In the case of AWR5, we show that it targets a novel cellular process, namely, the TORC1 pathway.
As mentioned above, the TORC1 protein complex regulates the transition between growth and quiescence in response to nutrient status and can be inhibited by rapamycin. TORC1 acts by controlling three major cell components: the kinase Sch9, Tap42, its associated phosphatases and the ATG1 complex14,15. Active TORC1 directly phosphorylates Sch9 -the orthologue of the mammalian S6 kinase-, which induces RiBi genes, such as STM1 and NSR1, to increase translation and promote growth (Fig. 1a). In addition, when TORC1 is active, the essential downstream regulatory protein, Tap42, is phosphorylated and associates with the catalytic subunits of the PP2A and PP2A-like phosphatases, which are retained in membranes interacting with TORC114. Finally, active TORC1 can inhibit autophagy by phosphorylation of ATG13, which prevents association with the ATG1 kinase and subsequent autophagy induction32. On the contrary, when TORC1 is inactivated by rapamycin treatment or nitrogen starvation, Tap42 and the PP2A and PP2A-like phosphatases are released to the cytosol and activated, allowing expression of stress genes and NCR genes such as GAP1 andMEP215 (Fig. 1a). This gene reprogramming takes place through PP2A-mediated inhibition of nuclear export of the Msn2/4 factors and PP2A/Sit4-mediated dephosphorylation and subsequent translocation of Gln3 to the nucleus. Our gene expression analyses and biochemical characterizations showed that the bacterial effector AWR5 interferes with the TORC1-regulated pathways, repressing ribosome biogenesis and translation and activating autophagy and stress responses. Activation of the latter, which are incompatible with growth, could explain the dramatic growth defects triggered by AWR5 in yeast. Our findings that mutants in two PP2A subunits (cdc55 and tpd3) totally rescued this phenotype strongly support that AWR5 impacts TORC1-regulated pathways in eukaryotic cells.
As mentioned before, most TORC1-controlled effects occur through two major effector branches, mediated by the Sch9 kinase and by complexes of Tap42 and the phosphatases (mainly PP2A and Sit4). The wide transcriptomic impact of AWR5 on all TORC1-controlled pathways, mimicking the effect of rapamycin or nitrogen starvation, could be explained by assuming that AWR5 targets multiple hits downstream the pathway. Along this line, downstream components of the TOR pathway have already been involved in plant defense: PP2A was found to negatively regulate pathogen perception40 and PP1A is targeted by a Phytophtora infestans effector41,42. However, the most likely scenario is that AWR5 would target a few or even a single target controlling all these processes. If this were the case, AWR5 would exert its function inhibiting TORC1 upstream of PP2A, thus causing Sch9 inhibition, autophagy activation and the release of Tap42 and PP2A phosphatase subunits. The notion of a single target is reinforced by the fact that only a limited number of T3E molecules are injected into the host cell to exert their function. Along this line, leaky expression of awr5 from a tet-off promoter in the presence of the repressor doxycycline had a detectable effect on yeast growth.
The observation that deletion of genes encoding two components of the PP2A heterotrimeric forms, CDC55 and TPD3, abolishes the dramatic growth defect of cells expressing awr5 suggests that in spite of the wide transcriptional effect caused by awr5 expression, the major reason for AWR5 toxicity lies downstream PP2A and indicates that the formation of this heterotrimer is essential for the negative effect of AWR5 to take place. In this regard, it is worth noting that deletion of TPD3 and of CDC55 yields yeast cells resistant to rapamycin, whereas that of RTS1 does not. Moreover, it has been proposed that an active TORC1 pathway promotes the association of Tap42 with PP2A catalytic subunits Pph21/22 to form complexes necessary for sustaining cell growth, whereas Cdc55 and Tpd3 would inhibit such association43. Although our study does not allow pointing to a specific TOR-regulated event to explain the inhibitory effect of AWR5, the observation that deletion of CDC55 only normalizes the expression of specific subsets of genes altered by awr5 expression (i.e. NCR genes but not ribosomal protein encoding genes) or the fact that AWR5-mediated autophagy promotion was not dependent on Cdc55 contribute to narrow the possible candidates.
Interestingly, during the course of this work, the cdc55 mutant has been also isolated in a screen for suppressors of the yeast growth inhibition caused by the Erwinia amylovora T3E DspA12. This could suggest that the PP2A phosphatase has evolved as a cellular hub, targeted by different pathogens to interfere with plant host cell homeostasis. However, DspA caused a specific alteration of the yeast sphingolipid biosynthesis, showing no overlap with AWR5-triggered phenotypes other than the Cdc55-dependent growth inhibition. In addition, AWR5 still caused its toxicity on strains with mutations in the small GTPase rho2 and in the sphingolipid biosynthesis gene sur1 (data not shown), which strongly supressed DspA-triggered growth defects12. All these data support a different mode of action for these T3Es, only sharing Cdc55 as an intermediate in signal transduction.
TOR functions are conserved across kingdoms; in plants TOR is also a master regulator of the cell, controlling the switch between stress and growth44,45. Our data clearly supports the idea that AWR5 alters the TOR pathway in plants.
First, awr5 expression in planta results in nitrate reductase activity inhibition. This enzyme has a central role in nitrogen metabolism and its inhibition has been previously linked to TOR deficiency and activated nitrogen recycling34. Noteworthy, even a minimal escape in awr5 expression visibly impacted plant nitrate reductase activity, similar to the yeast growth inhibition caused by leaky expression ofawr5. This strengthens the notion of a conserved AWR5 function as an extremely efficient modulator of the TOR pathway in disparate eukaryotic contexts. Second, TOR-deficient plants were more susceptibleto R. solanacearum infection and plants lacking the CDC55 homologues B55α or B55β, showed enhanced resistance to the pathogen. These opposite results are expected if the bacterium inhibits TOR signalling, as the B55 activity is repressed by TOR and demonstrate a novel role for the TOR complex in plant defence.
In the context of Ralstonia solanacearum infection it remains a mystery why a bacterial T3E would mimic the effect of nitrogen starvation on infected tissues. Interestingly, there are several instances in the literature showing modulation of the host metabolism by T3Es. For example, the R. solanacearum effector RipTPS was shown to possess trehalose-6-phosphate synthase activity46 and the effector WtsE fromPantoea stewartii was shown to alter phenylpropanoid metabolism47.
Furthermore, group A Streptococcus enhances its growth by activation of asparagine metabolism via ER stress induction in mammalian cells48. Since ER stress responses are intimately connected with TOR signalling49, it is tempting to speculate that AWR5 modulates the TOR pathway to induce ER stress responses and stimulate growth by an analogous mechanism to the one proposed in Streptococcus. On an alternative hypothetic scenario, AWR5-mediated inhibition of TOR (nitrogen recycling, autophagy, inhibition of protein synthesis…) would be beneficial for the bacterium during the last stages of infection, as it would facilitate plant cell dismissal and consequently nutrient availability.
Methods
Plasmids, strains and gene cloning
All strains and plasmids used in this study are described in Supplementary Table S1. For heterologous expression of awrs under the control of the galactose inducible promoter (GAL1), expression vectors were constructed by recombining entry clones carrying each of the awr ORFs into the Gateway destination vector pAG426GAL-ccdb-HA50 through a Gateway LR reaction (Invitrogen, Waltham, Massachusetts, USA). For expression of awr5 fragments in yeast, N-terminal (1368 bp) and C-terminal (1821 bp) halves of awr5 as well as a central (1425 bp) fragment overlapping them were amplified from genomic DNA.
For integration of the awr genes fused to a C-terminal GFP tag at the locus of URA3 gene in the yeast chromosome, each of them was cloned by Gateway recombination or ligation into the integrative vector pYI-GWY, a URA3 plasmid in which the heterologous genes are under the control of a Tet-off promoter created in this study. Following linearization with BstBI that cuts inside in URA3 cassette, pYI-GWY derivatives carrying genes awr1 to awr5 were integrated into the yeast chromosome by double recombination into the URA3 locus in yeast. To this end, the wild type strain JA-100 containing a ura3 point mutation was used as recipient, giving rise to uracyl autotrophs after awr integration. For expression of awr5 gene in the cdc55Δ mutant yeast strain, cloning was performed in two steps. Firstly, a cdc55::KanMX4 cassette from the cdc55Δ strain in the BY4741 background was amplified and subsequently introduced into the genome of strain JA-100. Secondly, the awr5 gene fused to the C-terminal GFP was integrated into the newly constructed cdc55 strain as described above.
Yeast strains and growth conditions
For expression of awrs or their fragments under the control of the galactose promoter, yeast cells were grown for 2 days in SD-Ura + raffinose 2%, then diluted to optical density at 600 nm of 0.4 in water and plated either in repressing media (glucose) or inducing media (galactose) to monitor the effects of AWRs in cell growth/viability. For standard growth inhibition experiments on plates, strains were incubated overnight with shaking in selective medium with doxycycline 20 μg/ml. Cultures were then normalized to OD600 = 0.1–0.2 and grown until exponential phase. 1 OD600 of cells were then harvested, washed 2 times with sterile water, re-suspended in 1 ml water and 10-fold serially diluted in water four times. Each suspension (5 or 10 μl) was dropped either in non-inducing media (+doxycycline) or inducing media (no doxycycline) onto agar plates and then incubated for 2–3 days before photographs were taken.
To test growth viability in liquid media over time and for sample harvesting for RNA isolation, yeast strains were grown overnight in rich YPD medium with doxycycline 15 μg/ml (repressing conditions), then normalized to OD600 = 0.05 and grown for 2, 4, 6 or 8 hours in YPD+dox (non-inducing conditions) and YPD (inducing conditions). Similar growth conditions were carried out for protein extraction and beta-galactosidase assays, using selective medium in this case. To test viability of yeast cells expressingawr5 after doxycycline addition, strains were grown overnight in either SD-Ura+dox (non-inducing conditions) or SD-Ura (inducing). Cells were recovered and normalized to OD600 = 0.05 and grown in liquid in SD-Ura+dox. Samples were harvested at different time points, serially 10-fold diluted and plated onto solid SD-Ura+dox and incubated for 2 days at 28 °C until colonies were counted.
For methylene blue staining, yeast cells carrying awr5 were harvested at 6 hours after induction and stained for 5 minutes with a 0.01% methylene blue solution in glycine buffer. In parallel, the same cells were fixed with formaldehyde 37% for 10 mins before methylene blue addition as a positive staining control. Images were obtained using a Dapi 395–440/FT 460/LP470 filterset.
To measure yeast cell size, wild-type yeast strains (JA-100) and strains bearing awr5 were grown overnight in YPD medium with and without doxycycline (15 μg/ml). The next day, cultures were normalized to OD600 = 0.05 and grown in liquid either in YPD+dox or YPD during 6 and 8 hours. Cells were analyzed with a Scepter Handheld Automated Cell Counter (Merck Millipore, Darmstadt, Germany).
To measure induction of autophagy, wild-type and cdc55Δ strains carrying awr5 and ATG8-GFP were grown overnight in selective media plus doxycycline. Cultures were then normalized to an OD600 = 0.2, grown until exponential phase, normalized again to OD600 = 0.05 and finally grown overnight with or without doxycycline until samples were harvested. For autophagy induction after nitrogen starvation JA-100 cells were grown overnight in SD medium without ammonium sulfate (BD Difco, Franklin Lakes, NJ, USA) and 2% glucose.
DNA microarray analysis
Aliquots of the same samples harvested to test viability of cells expressingawr5 in liquid media at 2, 4 and 6 hours after induction were used for microarray analysis. For microarray hybridization, total RNA (8 μg) was employed for cDNA synthesis and labelling using the indirect labelling kit (CyScribe Post-Labeling kit; GE Healthcare, Wauwatosa, WI, USA) with Cy3–dUTP and Cy5–dUTP fluorescent nucleotides. The cDNA obtained was dried, re-suspended in hybridization buffer and evaluated with a Nanodrop spectrophotometer (Nanodrop Technologies, Thermo Scientific, Waltham, MA, USA). The combined fluorescently labelled cDNAs were hybridized to yeast genomic microchips constructed in our laboratory by arraying 6014 different PCR-amplified open reading frames from S. cerevisiae51. Microarrays were processed as described previously52, scanned with a ScanArray 4000 apparatus (Packard BioChip Technologies, Perkin Elmer, Waltham, MA, USA) and the output was analysed using GenePix Pro 6.0 software. Data collected from 2 biological replicates (two microarrays each, with dye swap) after 2, 4 and 6 h of doxycycline removal (thus triggering expression of awr5) were combined. Genes were considered induced or repressed by AWR5 expression when the minus/plus doxycycline ratio was ≥2.0 or ≤0.5, respectively, for both biological replicates. All data has been added to the Gene Expression Omnibus (GEO) database under numbers GSE70202, GSE70331 and GSE70835.
qRT-PCR
Two independent biological replicas of the strain carrying awr5 grown in inducing and non-inducing conditions were harvested at 4 and 6 hours after induction and subjected to RNA extraction to quantify awr5 mRNA levels, whereas of GAP1, MEP2, STM1 and NSR1 levels were only tested from samples obtained 6 h after induction. For quantitative real-time PCR, a Light Cycler 480 (Roche, Basel, Switzerland) with SYBR Green chemistry was used with three technical replicas. Actin was used as a housekeeping gene to normalize samples.
RNA-seq experiments
Libraries were prepared with the QuantSeq 3′ mRNA kit (Lexogen, Greenland, NH, USA) using 0.5 μg of total RNA purified as above. Sequencing was performed in an Illumina MiSeq machine with Reagent Kit v3 (single end, 80–125 nt/read). Two biological replicates were sequenced, obtaining a total number of 8.4–12.9 million reads per condition. Mapping of fastq files to generate SAM files was carried out with the Bowtie2 software53 in local mode (95.1–97.3% mapped reads). The SAM files were analyzed with the SeqMonk software (www.bioinformatics.bbsrc.ac.uk/projects/seqmonk). Mapped reads were counted using CDS probes (extended 100 nt downstream the open reading frame because the library is biased towards the 3′-end of mRNAs) and corrected for the largest dataset. Raw data was subjected to diverse filters to remove sequences with a low number of reads.
Protein assays
For immunoblots, 30 or 40 OD600 units from overnight yeast cultures grown in non-inducing or inducing conditions were were resuspended in 500 μl of extraction buffer (50 mM Tris-HCl pH7.5, 1 mM EDTA, 0.1% Nonidet P-40, 1% glycerol, with complete protease inhibitor (Roche, Basel, Switzerland) and subjected to 10 cycles of 1 minute sonication and 1 minute pauses. Supernatants were recovered after centrifugation at 500 g for 10 min at 4 °C. 125 μg of total protein extracts were separated on polyacrylamide gels and immunoblot was performed using anti-GFP mouse monoclonal antibody (clone B-2; Santa Cruz Biotechnology, Dallas, TX, USA).
Beta-galactosidase activity was measured from, 2 ml of cultures pelleted 6 hours after induction as described54.
Plant material
Wild type (Wt) Columbia 0, TOR-silenced 35-7 (TOR RNAi)35, b55α andb55β Arabidopsis mutant lines36 were used. 3 to 4-week-old N. benthamiana plants were used for transient expression experiments.
Enzymatic activity determinations
To measure nitrate reductase activity, N. benthamiana plants were treated two times a week with 2mM-15mM KNO3, then, transient Agrobacterium-mediated transformation was performed as previously described20 using the estradiol-inducible vector pMDC7 carrying AWR5 or GUS. Protein expression was induced by painting the leaves 14 hours post-agrobacterium infiltration with 20 μM estradiol and Silwet L-77 adjuvant. Whole leaves (1 g) were harvested at 0 and 1 hour after post-estradiol induction and homogenized in 3 ml of 0.1 M HEPES-KOH, pH 7.5, 3% polyvinylpolypyrrolidone, 1 mM EDTA and 10 mM cysteine. The extracts were filtered through four layers of Miracloth (Merk Millipore, Billerica, USA) and centrifuged for 15 minutes at 30,000 × g at 4 °C and nitrate reductase activity measured as described in55.
To measure glucose-6-phosphate dehydrogenase activity N. benthamiana leaves were transiently transformed as previously described20 using the estradiol-inducible vector pMDC7 carrying AWR5 or GUS. Protein expression was induced by painting the leaves with 20 μM estradiol and Silwet L-77 adjuvant 14 hours post-agrobacterium infiltration. Half-leaves (500 mg) were harvested at 0 and 1 hour after post-estradiol induction and homogenized in 500 μl of 20 mM imidazol, pH 7. The extracts were centrifuged 15 minutes at 1000 × g at 4 °C and the supernatant was transferred to a new tube and kept on ice. To determine the activity of glucose-6-phosphate dehydrogenase activity 170 μl of 2x assay buffer (0.1 M imidazol, 0.2 M KCl, 20 mM MgCl2, 2 mM EDTA), 131 μl H2O, 7 μl of 10 mM NADP and 25 μl of cell-free extract were sequentially added to a spectrophotometer cuvette and the A340 was monitored for a few minutes until stabilization. Then 7 μl of 50 mM glucose-6-phosphate were added and the A340 was recorded, as a measure of Glucose-6-phosphate dehydrogenase activity (expressed as nmoles min−1 mg−1 protein).
Pathogenicity assays
R. solanacearum pathogenicity tests were carried out using the soil-drench method as described56.
Additional Information
How to cite this article: Popa, C. et al. The effector AWR5 from the plant pathogenRalstonia solanacearum is an inhibitor of the TOR signalling pathway. Sci. Rep.6, 27058; doi: 10.1038/srep27058 (2016).
Change history
26 November 2020
An amendment to this paper has been published and can be accessed via a link at the top of the paper.
References
Charro, N. & Mota, L. J. Approaches targeting the type III secretion system to treat or prevent bacterial infections. Expert Opin Drug Discov 10, 373–387, 10.1517/17460441.2015.1019860 (2015).
Macho, A. P. & Zipfel, C. Targeting of plant pattern recognition receptor-triggered immunity by bacterial type-III secretion system effectors. Curr Opin Microbiol 23, 14–22, 10.1016/j.mib.2014.10.009 (2015).
Dean, P. Functional domains and motifs of bacterial type III effector proteins and their roles in infection. FEMS Microbiol Rev 35, 1100–1125, 10.1111/j.1574-6976.2011.00271.x (2011).
Boch, J., Bonas, U. & Lahaye, T. TAL effectors–pathogen strategies and plant resistance engineering. New Phytol 204, 823–832 (2014).
Boyle, P. C. & Martin, G. B. Greasy tactics in the plant-pathogen molecular arms race. J Exp Bot 66, 1607–1616, 10.1093/jxb/erv059 (2015).
Deslandes, L. & Rivas, S. Catch me if you can: bacterial effectors and plant targets. Trends Plant Sci 17, 644–655, 10.1016/j.tplants.2012.06.011 (2012).
Marin, M. & Ott, T. Intrinsic disorder in plant proteins and phytopathogenic bacterial effectors. Chem Rev 114, 6912–6932, 10.1021/cr400488d (2014).
Popa, C., Coll, N. S., Valls, M. & Sessa, G. Yeast as a Heterologous Model System to Uncover Type III Effector Function. PLoS pathogens 12, e1005360, 10.1371/journal.ppat.1005360 (2016).
Von Pawel-Rammingen, U. et al. GAP activity of the Yersinia YopE cytotoxin specifically targets the Rho pathway: a mechanism for disruption of actin microfilament structure. Mol Microbiol 36, 737–748 (2000).
Munkvold, K. R., Martin, M. E., Bronstein, P. A. & Collmer, A. A survey of the Pseudomonas syringae pv. tomato DC3000 type III secretion system effector repertoire reveals several effectors that are deleterious when expressed in Saccharomyces cerevisiae. Mol Plant Microbe Interact 21, 490–502 (2008).
Salomon, D., Bosis, E., Dar, D., Nachman, I. & Sessa, G. Expression of Pseudomonas syringae type III effectors in yeast under stress conditions reveals that HopX1 attenuates activation of the high osmolarity glycerol MAP kinase pathway. Microbiology 158, 2859–2869, 10.1099/mic.0.062513-0 (2012).
Siamer, S. et al. Expression of the bacterial type III effector DspA/E in Saccharomyces cerevisiae down-regulates the sphingolipid biosynthetic pathway leading to growth arrest. The Journal of biological chemistry 289, 18466–18477, 10.1074/jbc.M114.562769 (2014).
Curak, J., Rohde, J. & Stagljar, I. Yeast as a tool to study bacterial effectors. Curr Opin Microbiol 12, 18–23 (2009).
Eltschinger, S. & Loewith, R. TOR Complexes and the Maintenance of Cellular Homeostasis. Trends Cell Biol 26, 148–159, 10.1016/j.tcb.2015.10.003 (2016).
Conrad, M. et al. Nutrient sensing and signaling in the yeast Saccharomyces cerevisiae. FEMS Microbiol Rev 38, 254–299, 10.1111/1574-6976.12065 (2014).
Coll, N. S. & Valls, M. Current knowledge on the Ralstonia solanacearum type III secretion system. Microb Biotechnol 6, 614–620, 10.1111/1751-7915.12056 (2013).
Peeters, N., Guidot, A., Vailleau, F. & Valls, M. Ralstonia solanacearum, a widespread bacterial plant pathogen in the post-genomic era. Mol Plant Pathol 14, 651–662, 10.1111/mpp.12038 (2013).
Mansfield, J. et al. Top 10 plant pathogenic bacteria in molecular plant pathology. Mol Plant Pathol 13, 614–629, 10.1111/j.1364-3703.2012.00804.x (2012).
Salanoubat, M. et al. Genome sequence of the plant pathogen Ralstonia solanacearum. Nature 415, 497–502, 10.1038/415497a (2002).
Sole, M. et al. The awr gene family encodes a novel class of Ralstonia solanacearum type III effectors displaying virulence and avirulence activities. Mol Plant Microbe Interact 25, 941–953, 10.1094/MPMI-12-11-0321 (2012).
Song, C. & Yang, B. Mutagenesis of 18 type III effectors reveals virulence function of XopZ(PXO99) in Xanthomonas oryzae pv. oryzae. Mol Plant Microbe Interact 23, 893–902, 10.1094/MPMI-23-7-0893 (2010).
Cunnac, S., Occhialini, A., Barberis, P., Boucher, C. & Genin, S. Inventory and functional analysis of the large Hrp regulon in Ralstonia solanacearum: identification of novel effector proteins translocated to plant host cells through the type III secretion system. Mol Microbiol 53, 115–128, 10.1111/j.1365-2958.2004.04118.x (2004).
Mukaihara, T., Tamura, N. & Iwabuchi, M. Genome-wide identification of a large repertoire of Ralstonia solanacearum type III effector proteins by a new functional screen. Mol Plant Microbe Interact 23, 251–262, 10.1094/MPMI-23-3-0251 (2010).
Pensec, F. et al. Towards the identification of Type III effectors associated to Ralstonia solanacearum virulence on tomato and eggplant. Phytopathology, 10.1094/PHYTO-06-15-0140-R (2015).
Godard, P. et al. Effect of 21 different nitrogen sources on global gene expression in the yeast Saccharomyces cerevisiae. Mol Cell Biol 27, 3065–3086, 10.1128/MCB.01084-06 (2007).
Gonzalez, A., Casado, C., Arino, J. & Casamayor, A. Ptc6 is required for proper rapamycin-induced down-regulation of the genes coding for ribosomal and rRNA processing proteins in S. cerevisiae. PLoS One 8, e64470, 10.1371/journal.pone.0064470 (2013).
Homma, T., Iwahashi, H. & Komatsu, Y. Yeast gene expression during growth at low temperature. Cryobiology 46, 230–237 (2003).
Van Dyke, N., Chanchorn, E. & Van Dyke, M. W. The Saccharomyces cerevisiae protein Stm1p facilitates ribosome preservation during quiescence. Biochem Biophys Res Commun 430, 745–750, 10.1016/j.bbrc.2012.11.078 (2013).
Conway, M. K., Grunwald, D. & Heideman, W. Glucose, nitrogen and phosphate repletion in Saccharomyces cerevisiae: common transcriptional responses to different nutrient signals. G3 (Bethesda) 2, 1003–1017, 10.1534/g3.112.002808 (2012).
Maegawa, K., Takii, R., Ushimaru, T. & Kozaki, A. Evolutionary conservation of TORC1 components, TOR, Raptor and LST8, between rice and yeast. Mol Genet Genomics 290, 2019–2030, 10.1007/s00438-015-1056-0 (2015).
Dilova, I., Chen, C. Y. & Powers, T. Mks1 in concert with TOR signaling negatively regulates RTG target gene expression in S. cerevisiae. Curr Biol 12, 389–395 (2002).
Kamada, Y. et al. Tor directly controls the Atg1 kinase complex to regulate autophagy. Mol Cell Biol 30, 1049–1058, 10.1128/MCB.01344-09 (2010).
Cheong, H. & Klionsky, D. J. Biochemical methods to monitor autophagy-related processes in yeast. Methods Enzymol 451, 1–26, 10.1016/S0076-6879(08)03201-1 (2008).
Ahn, C. S., Han, J. A., Lee, H. S., Lee, S. & Pai, H. S. The PP2A regulatory subunit Tap46, a component of the TOR signaling pathway, modulates growth and metabolism in plants. Plant Cell 23, 185–209, 10.1105/tpc.110.074005 (2011).
Deprost, D. et al. The Arabidopsis TOR kinase links plant growth, yield, stress resistance and mRNA translation. EMBO Rep 8, 864–870, 10.1038/sj.embor.7401043 (2007).
Heidari, B. et al. Protein phosphatase 2A B55 and A regulatory subunits interact with nitrate reductase and are essential for nitrate reductase activation. Plant Physiol 156, 165–172, 10.1104/pp.111.172734 (2011).
Costanzo, M. et al. The genetic landscape of a cell. Science 327, 425–431, 10.1126/science.1180823 (2010).
Coll, N. S., Epple, P. & Dangl, J. L. Programmed cell death in the plant immune system. Cell Death Differ 18, 1247–1256, 10.1038/cdd.2011.37 (2011).
Fujiwara, S. et al. RipAY, a plant pathogen effector protein exhibits robust gamma-glutamyl cyclotransferase activity when stimulated by eukaryotic thioredoxins. The Journal of biological chemistry, 10.1074/jbc.M115.678953 (2016).
Segonzac, C. et al. Negative control of BAK1 by protein phosphatase 2A during plant innate immunity. EMBO J 33, 2069–2079, 10.15252/embj.201488698 (2014).
Boevink, P. C. et al. A Phytophthora infestans RXLR effector targets plant PP1c isoforms that promote late blight disease. Nat Commun 7, 10311, 10.1038/ncomms10311 (2016).
Yerlikaya, S. et al. TORC1 and TORC2 work together to regulate ribosomal protein S6 phosphorylation in Saccharomyces cerevisiae. Mol Biol Cell 27, 397–409, 10.1091/mbc.E15-08-0594 (2016).
Jiang, Y. & Broach, J. R. Tor proteins and protein phosphatase 2A reciprocally regulate Tap42 in controlling cell growth in yeast. EMBO J 18, 2782–2792, 10.1093/emboj/18.10.2782 (1999).
Dobrenel, T. et al. TOR Signaling and Nutrient Sensing. Annu Rev Plant Biol, 10.1146/annurev-arplant-043014-114648 (2016).
Xiong, Y. & Sheen, J. Novel links in the plant TOR kinase signaling network. Curr Opin Plant Biol 28, 83–91, 10.1016/j.pbi.2015.09.006 (2015).
Poueymiro, M. et al. A Ralstonia solanacearum type III effector directs the production of the plant signal metabolite trehalose-6-phosphate. MBio 5, 10.1128/mBio.02065-14 (2014).
Asselin, J. A. et al. Perturbation of maize phenylpropanoid metabolism by an AvrE family type III effector from Pantoea stewartii. Plant Physiol 167, 1117–1135, 10.1104/pp.114.253120 (2015).
Baruch, M. et al. An extracellular bacterial pathogen modulates host metabolism to regulate its own sensing and proliferation. Cell 156, 97–108, 10.1016/j.cell.2013.12.007 (2014).
Crespo, J. L. BiP links TOR signaling to ER stress in Chlamydomonas. Plant Signal Behav 7, 273–275, 10.4161/psb.18767 (2012).
Alberti, S., Gitler, A. D. & Lindquist, S. A suite of Gateway cloning vectors for high-throughput genetic analysis in Saccharomyces cerevisiae. Yeast 24, 913–919, 10.1002/yea.1502 (2007).
Alberola, T. M. et al. A new set of DNA macrochips for the yeast Saccharomyces cerevisiae: features and uses. Int Microbiol 7, 199–206 (2004).
Hegde, P. et al. A concise guide to cDNA microarray analysis. Biotechniques 29, 548–550, 552–544, 556 passim (2000).
Langmead, B. & Salzberg, S. L. Fast gapped-read alignment with Bowtie 2. Nat Methods 9, 357–359, 10.1038/nmeth.1923 (2012).
Reynolds, A., Lundblad, V., Dorris, D. & Keaveney, M. Yeast vectors and assays for expression of cloned genes. Curr Protoc Mol Biol Chapter 13, Unit13 16, 10.1002/0471142727.mb1306s39 (2001).
Reed, A. J. & Hageman, R. H. Relationship between Nitrate Uptake, Flux and Reduction and the Accumulation of Reduced Nitrogen in Maize (Zea mays L.): II. Effect of nutrient nitrate concentration. Plant Physiol 66, 1184–1189 (1980).
Monteiro, F., Genin, S., van Dijk, I. & Valls, M. A luminescent reporter evidences active expression of Ralstonia solanacearum type III secretion system genes throughout plant infection. Microbiology 158, 2107–2116, 10.1099/mic.0.058610-0 (2012).
de Hoon, M. J., Imoto, S., Nolan, J. & Miyano, S. Open source clustering software. Bioinformatics 20, 1453–1454, 10.1093/bioinformatics/bth078 (2004).
Saldanha, A. J. Java Treeview–extensible visualization of microarray data. Bioinformatics 20, 3246–3248, 10.1093/bioinformatics/bth349 (2004).
Gonzalez, A., Ruiz, A., Casamayor, A. & Arino, J. Normal function of the yeast TOR pathway requires the type 2C protein phosphatase Ptc1. Mol Cell Biol 29, 2876–2888, 10.1128/MCB.01740-08 (2009).
Acknowledgements
Asier González, Albert Serra-Cardona, Diego Velázquez, Saúl Lema A., Montserrat Solé and Marina Puigvert are thanked for their advice and technical assistance. The authors thank Christian Meyer (Institut Jean-Pierre Bourgin, INRA, France) for the Arabidopsis TOR-silenced mutant and helpful comments; Eulàlia de Nadal (Universitat Pompeu Fabra, Spain) for the TAP constructs and technical advice; Ethel Queralt (IDIBELL, Spain) for the 3HA-Cdc55 construct; Catherine Lillo (University of Stavanger, Norway) for the Arabidopsis b55 mutants; Erica Washington (University of North Carolina at Chapel Hill, USA) for the pMDC7 Estradiol::HA-citrine plasmid; and Chang Sook Ahn (Yonsei University, South Korea) for the Myc-TOR construct. We kindly thank Jeff Dangl (University of North Carolina at Chapel Hill, USA) for critical reading of the manuscript. This work was funded by projects AGL2013-46898-R (MINECO, Spain) to N.S.C. and M.V., AGL2010-21870 (MICINN, Spain) to M.V., EU-Marie Curie Actions (PCDMC-321738 and PIIF-331392) and BP_B 00030 from the Catalan Government to N.S.C.; grants BFU2011-30197-C3-01 and BFU2014-54591-C2-1-P (MINECO, Spain) and 2014SGR4 from the Catalan Government to J.A.
Author information
Authors and Affiliations
Contributions
C.P. performed and designed the experiments, analyzed data and wrote the manuscript. L.L. performed the experiments. S.G. performed the experiments. L.T. performed the experiments. K.H. performed the experiments. M.T. designed the research. N.S.C. designed the experiments, analyzed data and wrote the manuscript. J.A. designed the experiments, analyzed data and wrote the manuscript. M.V. designed the experiments, analyzed data and wrote the manuscript. All authors reviewed the manuscript.
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Electronic supplementary material
Rights and permissions
This work is licensed under a Creative Commons Attribution 4.0 International License. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in the credit line; if the material is not included under the Creative Commons license, users will need to obtain permission from the license holder to reproduce the material. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/
About this article
Cite this article
Popa, C., Li, L., Gil, S. et al. The effector AWR5 from the plant pathogen Ralstonia solanacearum is an inhibitor of the TOR signalling pathway. Sci Rep 6, 27058 (2016). https://doi.org/10.1038/srep27058
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/srep27058
This article is cited by
-
Unravelling bacterial virulence factors in yeast: From identification to the elucidation of their mechanisms of action
Archives of Microbiology (2024)
-
The Minichromosome Maintenance Complex Component 2 (MjMCM2) of Meloidogyne javanica is a potential effector regulating the cell cycle in nematode-induced galls
Scientific Reports (2022)
-
Dynamic expression of Ralstonia solanacearum virulence factors and metabolism-controlling genes during plant infection
BMC Genomics (2021)
-
Puccinia melanocephala and Salt Stress Modified the Inositol Phosphate and TOR Signalling Pathway in Sugarcane
Sugar Tech (2021)
-
Yeast Ppz1 protein phosphatase toxicity involves the alteration of multiple cellular targets
Scientific Reports (2020)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.