Abstract
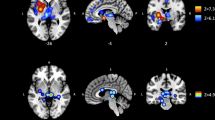
Substance use disorders are associated with neuroimaging abnormalities, but results are heterogeneous across studies and vary across substances, and the causal interpretation of these abnormalities is unknown. We have used network mapping approaches and a functional connectome from a large cohort of healthy participants (n = 1,000) to test whether neuroimaging abnormalities across substance use disorders map to a common brain network. Starting with coordinates of regional brain atrophy from 45 studies (3,791 participants), we found that 91% of the neuroimaging findings mapped to a common brain network. This network was specific to substance use disorder compared to atrophy associated with normal aging and neurodegenerative disease (PFWE < 0.05). Coordinates of functional MRI abnormalities from 99 studies (5,256 participants) mapped to a similar brain network. We found no differences in networks across different substance use categories. We combined all substance use disorder data (144 studies, 9,047 participants) to generate an overall coordinate-based network for substance use disorder, which included positive connectivity to the anterior cingulate, bilateral insulae, dorsolateral prefrontal cortices and thalamus, and negative connectivity to the medial prefrontal and occipital cortices. Lesions resulting in remission from nicotine use disorder (n = 34) intersected this network significantly more than control lesions (n = 69; P < 0.0084). We conclude that neuroimaging abnormalities across substance use disorders map to a common brain network that is similar across imaging modalities, substances and lesion locations that cause remission from substance use disorders.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 digital issues and online access to articles
$79.00 per year
only $6.58 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Data availability
All atrophy and fMRI coordinates used in our study are available in published studies as identified in previous meta-analyses9,10,27. De-identified lesion masks of stroke lesions causing remission from nicotine use disorder are publicly available25,56, and clinical data from the patients may be shared upon request to the corresponding author of the original lesion network mapping study25. A version of the connectome along with preprocessing details is publicly available47. Voxel-wise imaging results were projected onto a publicly available ultrahigh-resolution ex vivo brain aligned to MNI space58.
Code availability
Code to conduct connectivity analyses is available as part of the open-access Lead-DBS software package (https://lead-dbs.org)57, and supporting code for this study is available at https://github.com/nimlab/NMH_Stubbs2023/.
References
Degenhardt, L. & Hall, W. Extent of illicit drug use and dependence, and their contribution to the global burden of disease. Lancet 379, 55–70 (2012).
GBD 2019 Risk Factors Collaborators. Global burden of 87 risk factors in 204 countries and territories, 1990–2019: a systematic analysis for the Global Burden of Disease Study 2019. Lancet 396, 1223–1249 (2020).
Lipari, R. N., Hedden, S. L. & Hughes, A. Substance use and mental health estimates from the 2013 National Survey on Drug Use and Health: overview of findings. In The CBHSQ Report (Substance Abuse and Mental Health Services Administration (US), 2013).
Volkow, N. D., Koob, G. F. & McLellan, A. T. Neurobiologic advances from the brain disease model of addiction. N. Engl. J. Med. 374, 363–371 (2016).
Wollman, S. C. et al. Gray matter abnormalities in opioid-dependent patients: a neuroimaging meta-analysis. Am. J. Drug Alcohol Abuse 43, 505–517 (2017).
Martín-Santos, R. et al. Neuroimaging in cannabis use: a systematic review of the literature. Psychol. Med. 40, 383–398 (2010).
Mackey, S. & Paulus, M. Are there volumetric brain differences associated with the use of cocaine and amphetamine-type stimulants? Neurosci. Biobehav. Rev. 37, 300–316 (2013).
Ersche, K. D., Williams, G. B., Robbins, T. W. & Bullmore, E. T. Meta-analysis of structural brain abnormalities associated with stimulant drug dependence and neuroimaging of addiction vulnerability and resilience. Curr. Opin. Neurobiol. 23, 615–624 (2013).
Pando-Naude, V. et al. Gray and white matter morphology in substance use disorders: a neuroimaging systematic review and meta-analysis. Transl. Psychiatry 11, 29 (2021).
Klugah-Brown, B. et al. Common and separable neural alterations in substance use disorders: a coordinate-based meta-analyses of functional neuroimaging studies in humans. Hum. Brain Mapp. 41, 4459–4477 (2020).
Spindler, C., Mallien, L., Trautmann, S., Alexander, N. & Muehlhan, M. A coordinate-based meta-analysis of white matter alterations in patients with alcohol use disorder. Transl. Psychiatry 12, 40 (2022).
Mackey, S. et al. Mega-analysis of gray matter volume in substance dependence: general and substance-specific regional effects. Am. J. Psychiatry 176, 119–128 (2019).
Fox, M. D. Mapping symptoms to brain networks with the human connectome. N. Engl. J. Med. 379, 2237–2245 (2018).
Siddiqi, S. H., Kording, K. P., Parvizi, J. & Fox, M. D. Causal mapping of human brain function. Nat. Rev. Neurosci. 23, 361–375 (2022).
Siddiqi, S. H. et al. Brain stimulation and brain lesions converge on common causal circuits in neuropsychiatric disease. Nat. Hum. Behav. 5, 1707–1716 (2021).
Ferguson, M. A. et al. A human memory circuit derived from brain lesions causing amnesia. Nat. Commun. 10, 3497 (2019).
Taylor, J. J., Siddiqi, S. H. & Fox, M. D. Coordinate network mapping: an emerging approach for morphometric meta-analysis. Am. J. Psychiatry 178, 1080–1081 (2021).
Weil, R. S., Hsu, J. K., Darby, R. R., Soussand, L. & Fox, M. D. Neuroimaging in Parkinson’s disease dementia: connecting the dots. Brain Commun. 1, fcz006 (2019).
Burke, M. J. et al. Mapping migraine to a common brain network. Brain J. Neurol. 143, 541–553 (2020).
Darby, R. R., Joutsa, J. & Fox, M. D. Network localization of heterogeneous neuroimaging findings. Brain J. Neurol. 142, 70–79 (2019).
Zhukovsky, P. et al. Coordinate-based network mapping of brain structure in major depressive disorder in younger and older adults: a systematic review and meta-analysis. Am. J. Psychiatry 178, 1119–1128 (2021).
Peng, S., Xu, P., Jiang, Y. & Gong, G. Activation network mapping for integration of heterogeneous fMRI findings. Nat. Hum. Behav. https://doi.org/10.1038/s41562-022-01371-1 (2022).
Neiman, J., Haapaniemi, H. M. & Hillbom, M. Neurological complications of drug abuse: pathophysiological mechanisms. Eur. J. Neurol. 7, 595–606 (2000).
Cadet, J. L., Bisagno, V. & Milroy, C. M. Neuropathology of substance use disorders. Acta Neuropathol. (Berl.) 127, 91–107 (2014).
Joutsa, J. et al. Brain lesions disrupting addiction map to a common human brain circuit. Nat. Med. 28, 1249–1255 (2022).
Taylor, J. et al. A convergent brain network for psychiatric illness. Nat. Hum. Behav. 7, 420–429 (2023).
Minkova, L. et al. Gray matter asymmetries in aging and neurodegeneration: a review and meta-analysis. Hum. Brain Mapp. 38, 5890–5904 (2017).
Miller, J. M. et al. Anhedonia after a selective bilateral lesion of the globus pallidus. Am. J. Psychiatry 163, 786–788 (2006).
Moussawi, K., Kalivas, P. W. & Lee, J. W. Abstinence from drug dependence after bilateral Globus Pallidus hypoxic ischemic injury. Biol. Psychiatry 80, e79–e80 (2016).
Muskens, J. B., Schellekens, A. F. A., de Leeuw, F. E., Tendolkar, I. & Hepark, S. Damage in the dorsal striatum alleviates addictive behavior. Gen. Hosp. Psychiatry 34, 702–702 (2012).
Goldstein, R. Z. & Volkow, N. D. Dysfunction of the prefrontal cortex in addiction: neuroimaging findings and clinical implications. Nat. Rev. Neurosci. 12, 652–669 (2011).
Koob, G. F. & Volkow, N. D. Neurobiology of addiction: a neurocircuitry analysis. Lancet Psychiatry 3, 760–773 (2016).
Zhao, Y. et al. Anterior cingulate cortex in addiction: new insights for neuromodulation. Neuromodulation 24, 187–196 (2021).
Uddin, L. Q., Nomi, J. S., Hebert-Seropian, B., Ghaziri, J. & Boucher, O. Structure and function of the human insula. J. Clin. Neurophysiol. 34, 300–306 (2017).
Huang, A. S., Mitchell, J. A., Haber, S. N., Alia-Klein, N. & Goldstein, R. Z. The thalamus in drug addiction: from rodents to humans. Philos. Trans. R. Soc. Lond. B Biol. Sci. 373, 20170028 (2018).
Steele, J. D., Christmas, D., Eljamel, M. S. & Matthews, K. Anterior cingulotomy for major depression: clinical outcome and relationship to lesion characteristics. Biol. Psychiatry 63, 670–677 (2008).
Sharim, J. & Pouratian, N. Anterior cingulotomy for the treatment of chronic intractable pain: a systematic review. Pain Physician 19, 537–550 (2016).
Leong, S. L. et al. Anterior cingulate cortex implants for alcohol addiction: a feasibility study. Neurother. J. Am. Soc. Exp. Neurother. 17, 1287–1299 (2020).
Patel, S. R., Aronson, J. P., Sheth, S. A. & Eskandar, E. N. Lesion procedures in psychiatric neurosurgery. World Neurosurg. 80, S31.e9–16 (2013).
Naqvi, N. H., Rudrauf, D., Damasio, H. & Bechara, A. Damage to the insula disrupts addiction to cigarette smoking. Science 315, 531–534 (2007).
Jiang, J., Ferguson, M. A., Grafman, J., Cohen, A. L. & Fox, M. D. A lesion-derived brain network for emotion regulation. Biol. Psychiatry https://doi.org/10.1016/j.biopsych.2023.02.007 (2023).
Lees, B. et al. Promising vulnerability markers of substance use and misuse: a review of human neurobehavioral studies. Neuropharmacology 187, 108500 (2021).
Fox, M. D. et al. Resting-state networks link invasive and noninvasive brain stimulation across diverse psychiatric and neurological diseases. Proc. Natl Acad. Sci. USA 111, E4367–E4375 (2014).
Boes, A. D. et al. Network localization of neurological symptoms from focal brain lesions. Brain J. Neurol. 138, 3061–3075 (2015).
Horn, A. et al. Connectivity predicts deep brain stimulation outcome in Parkinson disease. Ann. Neurol. 82, 67–78 (2017).
Lancaster, J. L. et al. Bias between MNI and Talairach coordinates analyzed using the ICBM-152 brain template. Hum. Brain Mapp. 28, 1194–1205 (2007).
Cohen, A., Soussand, L., McManus, P. & Fox, M. GSP1000 Preprocessed Connectome (Harvard Dataverse, 2020); https://doi.org/10.7910/DVN/ILXIKS
Winkler, A. M., Ridgway, G. R., Webster, M. A., Smith, S. M. & Nichols, T. E. Permutation inference for the general linear model. NeuroImage 92, 381–397 (2014).
Bachi, K., Sierra, S., Volkow, N. D., Goldstein, R. Z. & Alia-Klein, N. Is biological aging accelerated in drug addiction? Curr. Opin. Behav. Sci. 13, 34–39 (2017).
Büttner, A. Neuropathology of Drug Abuse (Springer, 2021).
Byars, J. A., Beglinger, L. J., Moser, D. J., Gonzalez-Alegre, P. & Nopoulos, P. Substance abuse may be a risk factor for earlier onset of Huntington disease. J. Neurol. 259, 1824–1831 (2012).
Livingston, G. et al. Dementia prevention, intervention and care: 2020 report of the Lancet Commission. Lancet 396, 413–446 (2020).
R Core Team. R: A Language and Environment for Statistical Computing (R Foundation for Statistical Computing, 2021).
Siddiqi, S. H. et al. Distinct symptom-specific treatment targets for circuit-based neuromodulation. Am. J. Psychiatry 177, 435–446 (2020).
Padmanabhan, J. L. et al. A human depression circuit derived from focal brain lesions. Biol. Psychiatry 86, 749–758 (2019).
Joutsa, J. Smoking Lesion Datasets (2022); https://doi.org/10.7910/DVN/8BHHRS
Horn, A. et al. Lead-DBS v2: towards a comprehensive pipeline for deep brain stimulation imaging. NeuroImage 184, 293–316 (2019).
Edlow, B. L. et al. 7 Tesla MRI of the ex vivo human brain at 100 micron resolution. Sci. Data 6, 244 (2019).
Acknowledgements
We thank Drs. A. Boes, J. Bruss and E. van Wijngaarden for sharing the lesion and clinical data included in the lesion validation analysis and Fig. 5, and we thank Dr. J. Joutsa for sharing the lesion case study data included in Supplementary Fig. 5. J.L.S. was supported by a Canadian Institutes of Health Research Vanier Scholarship and a University of British Columbia Friedman Award for Scholars in Health. J.J.T. was supported by the National Institute of Mental Health (K23MH129829), the Brain and Behavior Research Foundation, Sidney R. Baer Foundation, the Baszucki Brain Research Fund and Harvard Medical School. F.L.W.V.J.S. was supported by the NIH (R01NS127892). A.C. was supported by the NIH (K23MH120510), the Child Neurology Foundation and the Simons Foundation. Data from the Rochester cohort was supported in part by the National Heart, Lung and Blood Institute Preventive Cardiology Training (grant no. T32 HL007937) and by the Clinical and Translational Science Institute (grant no. UL1 RR024160) from the National Institutes of Health. W.G.H. was supported by the Jack Bell Chair in Schizophrenia. M.D.F. was supported by grants from the NIH (R01MH113929, R21MH126271, R56AG069086, R21NS123813 and R01NS127892), the Kaye Family Research Endowment, the Ellison/Baszucki Family Foundation and the Manley Family.
Author information
Authors and Affiliations
Contributions
J.L.S., J.J.T. and M.D.F. designed the study. J.L.S. and W.D. processed the data. J.L.S., J.J.T., S.H.S., F.L.W.V.J.S., A.L.C., W.D. and M.D.F. conducted and interpreted analyses. J.L.S., J.J.T., S.H.S., F.L.W.V.J.S., A.L.C., W.D., C.A.H., A.A., H.Z.W., W.G.H., W.J.P. and M.D.F. contributed to the interpretation of the results and writing of the manuscript.
Corresponding author
Ethics declarations
Competing interests
C.H. is employed by BrainsWay and has financial interest in the company. S.H.S. is a scientific consultant for Magnus Medical, and a clinical consultant for Acacia Mental Health, Kaizen Brain Center and Boston Precision Neurotherapeutics. S.H.S. has received investigator-initiated research funding from Neuronetics and BrainsWay. S.H.S. has served as a speaker for BrainsWay (branded) and PsychU.org (unbranded, sponsored by Otsuka). S.H.S. owns stock in BrainsWay (publicly traded) and Magnus Medical (not publicly traded). S.H.S. owns intellectual property involving the use of functional connectivity to target TMS. M.D.F. is a consultant for Magnus Medical, Solaris and Boston Scientific, and has intellectual property using connectivity imaging to guide brain stimulation. The remaining authors declare no competing interests.
Peer review
Peer review information
Nature Mental Health thanks Tianye Jia and the other, anonymous, reviewers for their contributions to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
Supplementary Information
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Stubbs, J.L., Taylor, J.J., Siddiqi, S.H. et al. Heterogeneous neuroimaging findings across substance use disorders localize to a common brain network. Nat. Mental Health 1, 772–781 (2023). https://doi.org/10.1038/s44220-023-00128-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s44220-023-00128-7
This article is cited by
-
Adolescent substance use initiation and long-term neurobiological outcomes: insights, challenges and opportunities
Molecular Psychiatry (2024)
-
Investigating the impact of lumping heterogenous conduct problems: aggression and rule-breaking rely on distinct spontaneous brain activity
European Child & Adolescent Psychiatry (2024)