Abstract
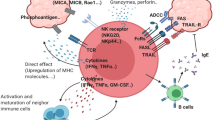
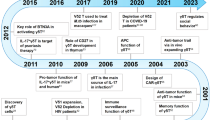
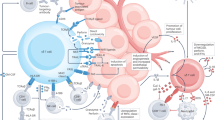
While the effector cells that mediate anti-tumor immunity have historically been attributed to αβ T cells and natural killer cells, γδ T cells are now being recognized as a complementary mechanism mediating tumor rejection. γδ T cells possess a host of functions ranging from antigen presentation to regulatory function and, importantly, have critical roles in eliciting anti-tumor responses where other immune effectors may be rendered ineffective. Recent discoveries have elucidated how these differing functions are mediated by γδ T cells with specific T cell receptors and spatial distribution. Their relative resistance to mechanisms of dysfunction like T cell exhaustion has spurred the development of therapeutic approaches exploiting γδ T cells, and an improved understanding of these cells should enable more effective immunotherapies.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Saito, H. et al. Complete primary structure of a heterodimeric T-cell receptor deduced from eDNA sequences. Nature 309, 757–762 (1984).
Brenner, M. B. et al. Identification of a putative second T-cell receptor. Nature 322, 145–149 (1986).
Hayday, A. C. γδ cells: a right time and a right place for a conserved third way of protection. Annu. Rev. Immunol. 18, 975–1026 (2000).
Girardi, M. et al. Regulation of cutaneous malignancy by γδ T cells. Science 294, 605–609 (2001).
Gao, Y. et al. γδ T cells provide an early source of interferon γ in tumor immunity. J. Exp. Med. 198, 433–442 (2003).
Liu, Z. et al. Protective immunosurveillance and therapeutic antitumor activity of γδ T cells demonstrated in a mouse model of prostate cancer. J. Immunol. 180, 6044–6053 (2008).
Gentles, A. J. et al. The prognostic landscape of genes and infiltrating immune cells across human cancers. Nat. Med. 21, 938–945 (2015).
Hayday, A. C. γδ T cells and the lymphoid stress-surveillance response. Immunity 31, 184–196 (2009).
Vantourout, P. & Hayday, A. Six-of-the-best: unique contributions of γδ T cells to immunology. Nat. Rev. Immunol. 13, 88–100 (2013).
Bonneville, M., O’Brien, R. L. & Born, W. K. γδ T cell effector functions: a blend of innate programming and acquired plasticity. Nat. Rev. Immunol. 10, 467–478 (2010).
Silva-Santos, B., Mensurado, S. & Coffelt, S. B. γδ T cells: pleiotropic immune effectors with therapeutic potential in cancer. Nat. Rev. Cancer 19, 392–404 (2019).
Pellicci, D. G., Koay, H.-F. & Berzins, S. P. Thymic development of unconventional T cells: how NKT cells, MAIT cells and γδ T cells emerge. Nat. Rev. Immunol. 20, 756–770 (2020).
Hu, Y. et al. T cells: origin and fate, subsets, diseases and immunotherapy. Signal Transduct. Target. Ther. 8, 434 (2023).
Carding, S. R. & Egan, P. J. γδ T cells: functional plasticity and heterogeneity. Nat. Rev. Immunol. 2, 336–345 (2002).
Hunter, S. et al. Human liver infiltrating γδ T cells are composed of clonally expanded circulating and tissue-resident populations. J. Hepatol. 69, 654–665 (2018).
Davey, M. S. et al. Clonal selection in the human Vδ1 T cell repertoire indicates γδ TCR-dependent adaptive immune surveillance. Nat. Commun. 8, 14760 (2017).
Khairallah, C. et al. γδ T cells confer protection against murine cytomegalovirus (MCMV). PLoS Pathog. 11, e1004702 (2015).
Ravens, S. et al. Human γδ T cells are quickly reconstituted after stem-cell transplantation and show adaptive clonal expansion in response to viral infection. Nat. Immunol. 18, 393–401 (2017).
Khairallah, C., Déchanet-Merville, J. & Capone, M. γδ T cell-mediated immunity to cytomegalovirus infection. Front. Immunol. 8, 105 (2017).
Street, S. E. A. et al. Innate immune surveillance of spontaneous B cell lymphomas by natural killer cells and γδ T cells. J. Exp. Med. 199, 879–884 (2004).
Wu, D., Wu, P., Qiu, F., Wei, Q. & Huang, J. Human γδT-cell subsets and their involvement in tumor immunity. Cell. Mol. Immunol. 14, 245–253 (2017).
Willcox, C. R., Davey, M. S. & Willcox, B. E. Development and selection of the human Vγ9Vδ2+ T-cell repertoire. Front. Immunol. 9, 1501 (2018).
Wrobel, P. et al. Lysis of a broad range of epithelial tumour cells by human γδ T cells: involvement of NKG2D ligands and T-cell receptor- versus NKG2D-dependent recognition. Scand. J. Immunol. 66, 320–328 (2007).
Rincon-Orozco, B. et al. Activation of Vγ9Vδ2 T cells by NKG2D. J. Immunol. 175, 2144–2151 (2005).
Tokuyama, H. et al. Vγ9Vδ2 T cell cytotoxicity against tumor cells is enhanced by monoclonal antibody drugs—rituximab and trastuzumab. Int. J. Cancer 122, 2526–2534 (2008).
Brandes, M., Willimann, K. & Moser, B. Professional antigen-presentation function by human γδ T cells. Science 309, 264–268 (2005).
Brandes, M. et al. Cross-presenting human γδ T cells induce robust CD8+ αβ T cell responses. Proc. Natl Acad. Sci. USA 106, 2307–2312 (2009).
Krangel, M. S., Yssel, H., Brocklehurst, C. & Spits, H. A distinct wave of human T cell receptor γ/δ lymphocytes in the early fetal thymus: evidence for controlled gene rearrangement and cytokine production. J. Exp. Med. 172, 847–859 (1990).
Di Lorenzo, B., Ravens, S. & Silva-Santos, B. High-throughput analysis of the human thymic Vδ1+ T cell receptor repertoire. Sci. Data 6, 115 (2019).
Sanz, M. et al. Deep characterization of human γδ T cell subsets defines shared and lineage-specific traits. Front. Immunol. 14, 1148988 (2023).
Poggi, A. et al. Vδ1 T lymphocytes from B-CLL patients recognize ULBP3 expressed on leukemic B cells and up-regulated by trans-retinoic acid. Cancer Res. 64, 9172–9179 (2004).
Knight, A., Mackinnon, S. & Lowdell, M. W. Human Vδ1 γ-δ T cells exert potent specific cytotoxicity against primary multiple myeloma cells. Cytotherapy 14, 1110–1118 (2012).
Mikulak, J. et al. NKp46-expressing human gut-resident intraepithelial Vδ1 T cell subpopulation exhibits high antitumor activity against colorectal cancer. JCI Insight 4, e125884 (2019).
Correia, D. V. et al. Differentiation of human peripheral blood Vδ1+ T cells expressing the natural cytotoxicity receptor NKp30 for recognition of lymphoid leukemia cells. Blood 118, 992–1001 (2011).
Pitard, V. et al. Long-term expansion of effector/memory Vδ2− γδ T cells is a specific blood signature of CMV infection. Blood 112, 1317–1324 (2008).
Farnault, L. et al. Clinical evidence implicating γ-δ T cells in EBV control following cord blood transplantation. Bone Marrow Transplant. 48, 1478–1479 (2013).
Fujishima, N. et al. Skewed T cell receptor repertoire of Vδ1+ γδ T lymphocytes after human allogeneic haematopoietic stem cell transplantation and the potential role for Epstein–Barr virus-infected B cells in clonal restriction. Clin. Exp. Immunol. 149, 70–79 (2007).
Kenna, T. et al. Distinct subpopulations of γδ T cells are present in normal and tumor-bearing human liver. Clin. Immunol. 113, 56–63 (2004).
Mangan, B. A. et al. Cutting edge: CD1d restriction and TH1/TH2/TH17 cytokine secretion by human Vδ3 T cells. J. Immunol. 191, 30–34 (2013).
Dunne, M. R. et al. Persistent changes in circulating and intestinal γδ T cell subsets, invariant natural killer T cells and mucosal-associated invariant T cells in children and adults with coeliac disease. PLoS ONE 8, e76008 (2013).
Willcox, C. R. et al. Cytomegalovirus and tumor stress surveillance by binding of a human γδ T cell antigen receptor to endothelial protein C receptor. Nat. Immunol. 13, 872–879 (2012).
Willcox, B. E. & Willcox, C. R. γδ TCR ligands: the quest to solve a 500-million-year-old mystery. Nat. Immunol. 20, 121–128 (2019).
Jacques, J. & Bonneville, M. Stimulation of γδ T cells by phosphoantigens. Res. Immunol. 147, 338–347 (1996).
Dieli, F. et al. Characterization of lung γδ T cells following intranasal infection with Mycobacterium bovis bacillus Calmette–Guérin. J. Immunol. 170, 463–469 (2003).
Lang, J. M. et al. Pilot trial of interleukin-2 and zoledronic acid to augment γδ T cells as treatment for patients with refractory renal cell carcinoma. Cancer Immunol. Immunother. 60, 1447–1460 (2011).
Fisher, J. P. H. et al. Neuroblastoma killing properties of Vδ2 and Vδ2-negative γδT cells following expansion by artificial antigen-presenting cells. Clin. Cancer Res. 20, 5720–5732 (2014).
Rigau, M. et al. Butyrophilin 2A1 is essential for phosphoantigen reactivity by γδ T cells. Science 367, eaay5516 (2020).
Karunakaran, M. M. et al. A distinct topology of BTN3A IgV and B30.2 domains controlled by juxtamembrane regions favors optimal human γδ T cell phosphoantigen sensing. Nat. Commun. 14, 7617 (2023).
Mensurado, S., Blanco-Domínguez, R. & Silva-Santos, B. The emerging roles of γδ T cells in cancer immunotherapy. Nat. Rev. Clin. Oncol. 20, 178–191 (2023).
Kabelitz, D., Serrano, R., Kouakanou, L., Peters, C. & Kalyan, S. Cancer immunotherapy with γδ T cells: many paths ahead of us. Cell. Mol. Immunol. 17, 925–939 (2020).
Legut, M., Cole, D. K. & Sewell, A. K. The promise of γδ T cells and the γδ T cell receptor for cancer immunotherapy. Cell. Mol. Immunol. 12, 656–668 (2015).
De Vries, N. L. et al. γδ T cells are effectors of immunotherapy in cancers with HLA class I defects. Nature 613, 743–750 (2023).
Rancan, C. et al. Exhausted intratumoral Vδ2− γδ T cells in human kidney cancer retain effector function. Nat. Immunol. 24, 612–624 (2023).
Wu, Y. et al. An innate-like Vδ1+ γδ T cell compartment in the human breast is associated with remission in triple-negative breast cancer. Sci. Transl. Med. 11, eaax9364 (2019).
Wu, Y. et al. A local human Vδ1 T cell population is associated with survival in nonsmall-cell lung cancer. Nat. Cancer 3, 696–709 (2022).
Zakeri, N. et al. Characterisation and induction of tissue-resident γ δ T-cells to target hepatocellular carcinoma. Nat. Commun. 13, 1372 (2022).
Paul, S. & Lal, G. Regulatory and effector functions of gamma-delta (γδ) T cells and their therapeutic potential in adoptive cellular therapy for cancer. Int. J. Cancer 139, 976–985 (2016).
Wu, P. et al. γδT17 cells promote the accumulation and expansion of myeloid-derived suppressor cells in human colorectal cancer. Immunity 40, 785–800 (2014).
Sacchi, A. et al. Myeloid-derived suppressor cells specifically suppress IFN-γ production and antitumor cytotoxic activity of Vδ2 T cells. Front. Immunol. 9, 1271 (2018).
Peng, G. et al. Tumor-infiltrating γδ T cells suppress T and dendritic cell function via mechanisms controlled by a unique Toll-like receptor signaling pathway. Immunity 27, 334–348 (2007).
Moser, B. & Brandes, M. γδ T cells: an alternative type of professional APC. Trends Immunol. 27, 112–118 (2006).
Rampoldi, F., Ullrich, L. & Prinz, I. Revisiting the interaction of γδ T-cells and B-cells. Cells 9, 743 (2020).
Mensurado, S. et al. Tumor-associated neutrophils suppress pro-tumoral IL-17+ γδ T cells through induction of oxidative stress. PLoS Biol. 16, e2004990 (2018).
Harmon, C. et al. γδ T cell dichotomy with opposing cytotoxic and wound healing functions in human solid tumors. Nat. Cancer 4, 1122–1137 (2023).
Ye, J. et al. Tumor-derived γδ regulatory T cells suppress innate and adaptive immunity through the induction of immunosenescence. J. Immunol. 190, 2403–2414 (2013).
Ma, C. et al. Tumor-infiltrating γδ T lymphocytes predict clinical outcome in human breast cancer. J. Immunol. 189, 5029–5036 (2012).
Daley, D. et al. γδ T cells support pancreatic oncogenesis by restraining αβ T cell activation. Cell 166, 1485–1499 (2016).
Hu, G. et al. Tumor-infiltrating CD39+ γδ Tregs are novel immunosuppressive T cells in human colorectal cancer. Oncoimmunology 6, e1277305 (2017).
Castella, B. et al. Anergic bone marrow Vγ9Vδ2 T cells as early and long-lasting markers of PD-1-targetable microenvironment-induced immune suppression in human myeloma. Oncoimmunology 4, e1047580 (2015).
Mao, Y. et al. A new effect of IL-4 on human γδ T cells: promoting regulatory Vδ1 T cells via IL-10 production and inhibiting function of Vδ2 T cells. Cell. Mol. Immunol. 13, 217–228 (2016).
Viey, E. et al. Phosphostim-activated γδ T cells kill autologous metastatic renal cell carcinoma. J. Immunol. 174, 1338–1347 (2005).
Inman, B. A. et al. Questionable relevance of γδ T lymphocytes in renal cell carcinoma. J. Immunol. 180, 3578–3584 (2008).
Roy, S. et al. Molecular analysis of lipid-reactive Vδ1 γδ T cells identified by CD1c tetramers. J. Immunol. 196, 1933–1942 (2016).
Luoma, A. M. et al. Crystal structure of Vδ1 T cell receptor in complex with CD1d-sulfatide shows MHC-like recognition of a self-lipid by human γδ T cells. Immunity 39, 1032–1042 (2013).
Barbee, S. D. et al. Skint-1 is a highly specific, unique selecting component for epidermal T cells. Proc. Natl Acad. Sci. USA 108, 3330–3335 (2011).
Harly, C. et al. Key implication of CD277/butyrophilin-3 (BTN3A) in cellular stress sensing by a major human γδ T-cell subset. Blood 120, 2269–2279 (2012).
Marlin, R. et al. Sensing of cell stress by human γδ TCR-dependent recognition of annexin A2. Proc. Natl Acad. Sci. USA 114, 3163–3168 (2017).
Groh, V., Steinle, A., Bauer, S. & Spies, T. Recognition of stress-induced MHC molecules by intestinal epithelial γδ T cells. Science 279, 1737–1740 (1998).
Wu, J., Groh, V. & Spies, T. T. Cell antigen receptor engagement and specificity in the recognition of stress-inducible MHC class I-related chains by human epithelial γδ T cells. J. Immunol. 169, 1236–1240 (2002).
Dai, Y., Chen, H., Mo, C., Cui, L. & He, W. Ectopically expressed human tumor biomarker MutS homologue 2 is a novel endogenous ligand that is recognized by human γδ T cells to induce innate anti-tumor/virus immunity. J. Biol. Chem. 287, 16812–16819 (2012).
Le Nours, J. et al. A class of γδ T cell receptors recognize the underside of the antigen-presenting molecule MR1. Science 366, 1522–1527 (2019).
Rice, M. T. et al. Recognition of the antigen-presenting molecule MR1 by a Vδ3+ γδ T cell receptor. Proc. Natl Acad. Sci. USA 118, e2110288118 (2021).
Silva‐Santos, B., Schamel, W. W. A., Fisch, P. & Eberl, M. γδ T‐cell conference 2012: close encounters for the fifth time. Eur. J. Immunol. 42, 3101–3105 (2012).
Kong, Y. et al. The NKG2D ligand ULBP4 binds to TCRγ9/δ2 and induces cytotoxicity to tumor cells through both TCRγδ and NKG2D. Blood 114, 310–317 (2009).
Scotet, E. et al. Tumor recognition following Vγ9Vδ2 T cell receptor interactions with a surface F1-ATPase-related structure and apolipoprotein A-I. Immunity 22, 71–80 (2005).
Bauer, S. et al. Activation of NK cells and T cells by NKG2D, a receptor for stress-inducible MICA. Science 285, 727–729 (1999).
McGraw, J. M. & Witherden, D. A. γδ T cell costimulatory ligands in antitumor immunity. Explor. Immunol. 2, 79–97 (2022).
Toutirais, O. et al. DNAX accessory molecule‐1 (CD226) promotes human hepatocellular carcinoma cell lysis by Vγ9Vδ2 T cells. Eur. J. Immunol. 39, 1361–1368 (2009).
Herold, N. et al. Integrin activation enables rapid detection of functional Vδ1+ and Vδ2+ γδ T cells. Eur. J. Immunol. 52, 730–736 (2022).
Gober, H.-J. et al. Human T cell receptor γδ cells recognize endogenous mevalonate metabolites in tumor cells. J. Exp. Med. 197, 163–168 (2003).
Morita, C. T., Jin, C., Sarikonda, G. & Wang, H. Nonpeptide antigens, presentation mechanisms, and immunological memory of human Vγ2Vδ2 T cells: discriminating friend from foe through the recognition of prenyl pyrophosphate antigens. Immunol. Rev. 215, 59–76 (2007).
Chen, H. et al. Identification of human T cell receptor γδ-recognized epitopes/proteins via CDR3δ peptide-based immunobiochemical strategy. J. Biol. Chem. 283, 12528–12537 (2008).
Kunzmann, V. et al. Stimulation of γδ T cells by aminobisphosphonates and induction of antiplasma cell activity in multiple myeloma. Blood 96, 384–392 (2000).
Payne, K. K. et al. BTN3A1 governs antitumor responses by coordinating αβ and γδ T cells. Science 369, 942–949 (2020).
Mamedov, M. R. et al. CRISPR screens decode cancer cell pathways that trigger γδ T cell detection. Nature 621, 188–195 (2023).
Song, Z. et al. Human γδ T cell identification from single-cell RNA sequencing datasets by modular TCR expression. J. Leukoc. Biol. 114, 630–638 (2023).
Wherry, E. J. T cell exhaustion. Nat. Immunol. 12, 492–499 (2011).
Wherry, E. J. & Kurachi, M. Molecular and cellular insights into T cell exhaustion. Nat. Rev. Immunol. 15, 486–499 (2015).
Beltra, J.-C. et al. Developmental relationships of four exhausted CD8+ T cell subsets reveals underlying transcriptional and epigenetic landscape control mechanisms. Immunity 52, 825–841 (2020).
Li, Y.-L. et al. Single-cell analysis reveals immune modulation and metabolic switch in tumor-draining lymph nodes. Oncoimmunology 9, 1830513 (2020).
Peters, C., Oberg, H.-H., Kabelitz, D. & Wesch, D. Phenotype and regulation of immunosuppressive Vδ2-expressing γδ T cells. Cell. Mol. Life Sci. 71, 1943–1960 (2014).
Dondero, A. et al. PD-L1 expression in metastatic neuroblastoma as an additional mechanism for limiting immune surveillance. Oncoimmunology 5, e1064578 (2016).
Hoeres, T., Holzmann, E., Smetak, M., Birkmann, J. & Wilhelm, M. PD-1 signaling modulates interferon-γ production by gamma delta (γδ) T-cells in response to leukemia. Oncoimmunology 8, 1550618 (2019).
Wu, K. et al. Vδ2 T cell subsets, defined by PD-1 and TIM-3 expression, present varied cytokine responses in acute myeloid leukemia patients. Int. Immunopharmacol. 80, 106122 (2020).
Weimer, P. et al. Tissue-specific expression of TIGIT, PD-1, TIM-3, and CD39 by γδ T cells in ovarian cancer. Cells 11, 964 (2022).
Wistuba-Hamprecht, K. et al. Proportions of blood-borne Vδ1+ and Vδ2+ T-cells are associated with overall survival of melanoma patients treated with ipilimumab. Eur. J. Cancer 64, 116–126 (2016).
Foord, E., Arruda, L. C. M., Gaballa, A., Klynning, C. & Uhlin, M. Characterization of ascites- and tumor-infiltrating γδ T cells reveals distinct repertoires and a beneficial role in ovarian cancer. Sci. Transl. Med. 13, eabb0192 (2021).
Davies, D. et al. PD-1 defines a distinct, functional, tissue-adapted state in Vδ1+ T cells with implications for cancer immunotherapy. Nat. Cancer 5, 420–432 (2024).
Lien, S. C. et al. Tumor reactive γδ T cells contribute to a complete response to PD-1 blockade in a Merkel cell carcinoma patient. Nat. Commun. 15, 1094 (2024).
Hoeres, T., Smetak, M., Pretscher, D. & Wilhelm, M. Improving the efficiency of Vγ9Vδ2 T-cell immunotherapy in cancer. Front. Immunol. 9, 800 (2018).
Kobayashi, H. et al. Safety profile and anti-tumor effects of adoptive immunotherapy using γ-δ T cells against advanced renal cell carcinoma: a pilot study. Cancer Immunol. Immunother. 56, 469–476 (2007).
Kobayashi, H., Tanaka, Y., Yagi, J., Minato, N. & Tanabe, K. Phase I/II study of adoptive transfer of γδ T cells in combination with zoledronic acid and IL-2 to patients with advanced renal cell carcinoma. Cancer Immunol. Immunother. 60, 1075–1084 (2011).
Bennouna, J. et al. Phase-I study of Innacell γδ™, an autologous cell-therapy product highly enriched in γ9δ2 T lymphocytes, in combination with IL-2, in patients with metastatic renal cell carcinoma. Cancer Immunol. Immunother. 57, 1599–1609 (2008).
Nakajima, J. et al. A phase I study of adoptive immunotherapy for recurrent non-small-cell lung cancer patients with autologous γδ T cells. Eur. J. Cardiothorac. Surg. 37, 1191–1197 (2010).
Meraviglia, S. et al. In vivo manipulation of Vγ9Vδ2 T cells with zoledronate and low-dose interleukin-2 for immunotherapy of advanced breast cancer patients. Clin. Exp. Immunol. 161, 290–297 (2010).
Nicol, A. J. et al. Clinical evaluation of autologous γ δ T cell-based immunotherapy for metastatic solid tumours. Br. J. Cancer 105, 778–786 (2011).
Kakimi, K. et al. Adoptive transfer of zoledronate-expanded autologous Vγ9Vδ2 T-cells in patients with treatment-refractory non-small-cell lung cancer: a multicenter, open-label, single-arm, phase 2 study. J. Immunother. Cancer 8, e001185 (2020).
Aoki, T. et al. Adjuvant combination therapy with gemcitabine and autologous γδ T-cell transfer in patients with curatively resected pancreatic cancer. Cytotherapy 19, 473–485 (2017).
Lobbous, M. et al. INB-200 phase I study of gene modified autologous gamma-delta (γδ) T cells in patients with newly diagnosed glioblastoma multiforme (GBM) receiving maintenance temozolomide (TMZ). J. Clin. Oncol. 41, 2007 (2023).
Sebestyen, Z., Prinz, I., Déchanet-Merville, J., Silva-Santos, B. & Kuball, J. Translating gammadelta (γδ) T cells and their receptors into cancer cell therapies. Nat. Rev. Drug Discov. 19, 169–184 (2020).
Vydra, J. et al. A phase I trial of allogeneic γδ T lymphocytes from haploidentical donors in patients with refractory or relapsed acute myeloid leukemia. Clin. Lymphoma Myeloma Leuk. 23, e232–e239 (2023).
Lamb, L., Rochlin, K. & Goshwani, T. Harnessing the Power of γ-δ T Cells investors.in8bio.com/static-files/f27cfe0f-d60a-4f95-afac-1741694b0d28 (2023).
McGuirk, J. P. et al. Inb-100: a pilot study of donor derived, ex-vivo expanded/activated γ-δ T cell (EAGD) infusion following haploidentical hematopoietic stem cell transplantation and post-transplant cyclophosphamide (PTCy). Blood 142, 4853 (2023).
Nishimoto, K. P. et al. Allogeneic CD20‐targeted γδ T cells exhibit innate and adaptive antitumor activities in preclinical B‐cell lymphoma models. Clin. Transl. Immunol. 11, e1373 (2022).
Neelapu, S. S. et al. A phase 1 study of ADI-001: anti-CD20 CAR-engineered allogeneic gamma delta1 (γδ) T cells in adults with B-cell malignancies. Blood 140, 4617–4619 (2022).
Bai, Lu. et al. Effects of IL-2 and IL-15 on the proliferative and antitumor capacities of allogeneic anti-CD20 CAR-engineered γδ T cells in a 3D B cell lymphoma spheroid assay. J. Immunother. Cancer 8, A1–A559 (2020).
Sasse, S. et al. AFM13 in patients with relapsed or refractory classical Hodgkin lymphoma: final results of an open-label, randomized, multicenter phase II trial. Leuk. Lymphoma 63, 1871–1878 (2022).
De Gassart, A. et al. Development of ICT01, a first-in-class, anti-BTN3A antibody for activating Vγ9Vδ2 T cell-mediated antitumor immune response. Sci. Transl. Med. 13, eabj0835 (2021).
Bono, J. D. et al. ICT01 plus low dose SC IL-2 produces a robust anti-tumor immune activation in advanced cancer patients (EVICTION-2 Study). J. Immunother. Cancer 11, A715 (2023).
Makkouk, A. et al. Off-the-shelf Vδ1 γ δ T cells engineered with glypican-3 (GPC-3)-specific chimeric antigen receptor (CAR) and soluble IL-15 display robust antitumor efficacy against hepatocellular carcinoma. J. Immunother. Cancer 9, e003441 (2021).
Majzner, R. G. & Mackall, C. L. Tumor antigen escape from CAR T-cell therapy. Cancer Discov. 8, 1219–1226 (2018).
Acknowledgements
L.F. is supported by NCI R35CA253175, Peter Michael Foundation, and the Prostate Cancer Foundation.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
L.F. reports research support to the institution from AbbVie, Bavarian Nordic, Bristol Myers Squibb, Dendreon, Janssen, Merck and Roche–Genentech and ownership interests in Actym, Atreca, BioAtla, Bolt, ImmunoGenesis, Nutcracker, RAPT, Scribe and Senti, unrelated to the work here. The other authors declare no competing interests.
Peer review information
Nature Cancer thanks the anonymous reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Arias-Badia, M., Chang, R. & Fong, L. γδ T cells as critical anti-tumor immune effectors. Nat Cancer 5, 1145–1157 (2024). https://doi.org/10.1038/s43018-024-00798-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s43018-024-00798-x