Abstract
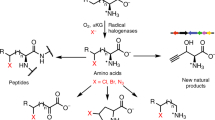
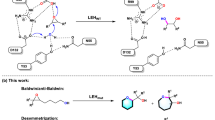
Lactam rings are found in many biologically active natural products and pharmaceuticals, including important classes of antibiotics. Methods for the asymmetric synthesis of these molecules are therefore highly desirable, particularly through the selective functionalization of unreactive aliphatic C–H bonds. Here we show the development of a strategy for the asymmetric synthesis of β-, γ- and δ-lactams via the haemoprotein-catalysed intramolecular C–H amidation of readily accessible dioxazolone reagents. Engineered myoglobin variants serve as excellent biocatalysts for this transformation, yielding the desired lactam products in high yields with high enantioselectivity and on a preparative scale. Mechanistic and computational studies were conducted to elucidate the nature of the C–H amidation and enantiodetermining steps and provide insights into the protein-mediated control of the regioselectivity and stereoselectivity. Additionally, an alkaloid natural product and a drug molecule were synthesized chemoenzymatically in fewer steps (7–8 versus 11–12) than previously reported, further demonstrating the power of biosynthetic strategies for the preparation of complex bioactive molecules.

This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
Data availability
The crystallographic data of the small molecules have been deposited at the Cambridge Crystallographic Data Centre (CCDC) under deposition numbers CCDC 1893087 (2a), 2157011 (2m), 2157007 (4j) and 2157010 (6c). The data can be obtained free of charge via https://www.ccdc.cam.ac.uk/structures/.
References
Guillemard, L., Kaplaneris, N., Ackermann, L. & Johansson, M. J. Late-stage C–H functionalization offers new opportunities in drug discovery. Nat. Rev. Chem. 5, 522–545 (2021).
Hong, B. K., Luo, T. P. & Lei, X. G. Late-stage diversification of natural products. ACS Cent. Sci. 6, 622–635 (2020).
Gutekunst, W. R. & Baran, P. S. C–H functionalization logic in total synthesis. Chem. Soc. Rev. 40, 1976–1991 (2011).
Wencel-Delord, J. & Glorius, F. C–H bond activation enables the rapid construction and late-stage diversification of functional molecules. Nat. Chem. 5, 369–375 (2013).
Upp, D. M. & Lewis, J. C. Selective C–H bond functionalization using repurposed or artificial metalloenzymes. Curr. Opin. Chem. Biol. 37, 48–55 (2017).
Ren, X. K. & Fasan, R. Engineered and artificial metalloenzymes for selective C–H functionalization. Curr. Opin. Green Sustain. Chem. 31, 100494 (2021).
Chakrabarty, S., Wang, Y., Perkins, J. C. & Narayan, A. R. H. Scalable biocatalytic C–H oxyfunctionalization reactions. Chem. Soc. Rev. 49, 8137–8155 (2020).
Roizen, J. L., Harvey, M. E. & Du Bois, J. Metal-catalyzed nitrogen-atom transfer methods for the oxidation of aliphatic C–H bonds. Acc. Chem. Res. 45, 911–922 (2012).
Dequirez, G., Pons, V. & Dauban, P. Nitrene chemistry in organic synthesis: still in its infancy? Angew. Chem. Int. Ed. 51, 7384–7395 (2012).
Park, Y., Kim, Y. & Chang, S. Transition metal-catalyzed C–H amination: scope, mechanism, and applications. Chem. Rev. 117, 9247–9301 (2017).
Royer, J. Chiral Amine Synthesis. Methods, Developments and Applications (Wiley-VCH, 2010).
Tahlan, K. & Jensen, S. E. Origins of the β-lactam rings in natural products. J. Antibiot. 66, 401–410 (2013).
Hong, S. Y. et al. Selective formation of γ-lactams via C–H amidation enabled by tailored iridium catalysts. Science 359, 1016–1021 (2018).
Park, Y. & Chang, S. Asymmetric formation of γ-lactams via C–H amidation enabled by chiral hydrogen-bond-donor catalysts. Nat. Catal. 2, 219–227 (2019).
Zhou, Z. J. et al. Non-C2-symmetric chiral-at-ruthenium catalyst for highly efficient enantioselective intramolecular C(sp3)–H amidation. J. Am. Chem. Soc. 141, 19048–19057 (2019).
Breslow, R. & Gellman, S. H. Tosylamidation of cyclohexane by a cytochrome P450 model. J. Chem. Soc. Chem. Commun. 1400–1401 (1982).
McIntosh, J. A. et al. Enantioselective intramolecular C–H amination catalyzed by engineered cytochrome P450 enzymes in vitro and in vivo. Angew. Chem. Int. Ed. 52, 9309–9312 (2013).
Singh, R., Bordeaux, M. & Fasan, R. P450-catalyzed intramolecular sp3 C–H amination with arylsulfonyl azide substrates. ACS Catal. 4, 546–552 (2014).
Hyster, T. K., Farwell, C. C., Buller, A. R., McIntosh, J. A. & Arnold, F. H. Enzyme-controlled nitrogen-atom transfer enables regiodivergent C–H amination. J. Am. Chem. Soc. 136, 15505–15508 (2014).
Singh, R., Kolev, J. N., Sutera, P. A. & Fasan, R. Enzymatic C(sp3)–H amination: P450-catalyzed conversion of carbonazidates into oxazolidinones. ACS Catal. 5, 1685–1691 (2015).
Steck, V., Kolev, J. N., Ren, X. K. & Fasan, R. Mechanism-guided design and discovery of efficient cytochrome P450-derived C–H amination biocatalysts. J. Am. Chem. Soc. 142, 10343–10357 (2020).
Yang, Y., Cho, I., Qi, X. T., Liu, P. & Arnold, F. H. An enzymatic platform for the asymmetric amination of primary, secondary and tertiary C(sp3)–H bonds. Nat. Chem. 11, 987–993 (2019).
Prier, C. K., Zhang, R. J. K., Buller, A. R., Brinkmann-Chen, S. & Arnold, F. H. Enantioselective, intermolecular benzylic C–H amination catalysed by an engineered iron-haem enzyme. Nat. Chem. 9, 629–634 (2017).
Athavale, S. V. et al. Biocatalytic, intermolecular C–H bond functionalization for the synthesis of enantioenriched amides. Angew. Chem. Int. Ed. 60, 24864–24869 (2021).
Liu, Z. et al. An enzymatic platform for primary amination of 1-aryl-2-alkyl alkynes. J. Am. Chem. Soc. 144, 80–85 (2022).
Dydio, P., Key, H. M., Hayashi, H., Clark, D. S. & Hartwig, J. F. Chemoselective, enzymatic C–H bond amination catalyzed by a cytochrome P450 containing an Ir(Me)-PIX cofactor. J. Am. Chem. Soc. 139, 1750–1753 (2017).
Goldberg, N. W., Knight, A. M., Zhang, R. J. K. & Arnold, F. H. Nitrene transfer catalyzed by a non-heme iron enzyme and enhanced by non-native small-molecule ligands. J. Am. Chem. Soc. 141, 19585–19588 (2019).
Vila, M. A., Steck, V., Giordano, S. R., Carrera, I. & Fasan, R. C–H amination via nitrene transfer catalyzed by mononuclear non-heme iron-dependent enzymes. ChemBioChem 21, 1981–1987 (2020).
van Vliet, K. M. & de Bruin, B. Dioxazolones: stable substrates for the catalytic transfer of acyl nitrenes. ACS Catal. 10, 4751–4769 (2020).
Biegasiewicz, K. F. et al. Photoexcitation of flavoenzymes enables a stereoselective radical cyclization. Science 364, 1166–1169 (2019).
Black, M. J. et al. Asymmetric redox-neutral radical cyclization catalysed by flavin-dependent ‘ene’-reductases. Nat. Chem. 12, 71–75 (2020).
Zhou, Q., Chin, M., Fu, Y., Liu, P. & Yang, Y. Stereodivergent atom-transfer radical cyclization by engineered cytochromes P450. Science 374, 1612–1616 (2021).
Rumo, C. et al. An artificial metalloenzyme based on a copper heteroscorpionate enables sp3 C–H functionalization via intramolecular carbene insertion. J. Am. Chem. Soc. 144, 11676–11684 (2022).
Kweon, J. & Chang, S. Highly robust iron catalyst system for intramolecular C(sp3)–H amidation leading to γ-lactams. Angew. Chem. Int. Ed. 60, 2909–2914 (2021).
Bordeaux, M., Tyagi, V. & Fasan, R. Highly diastereoselective and enantioselective olefin cyclopropanation using engineered myoglobin-based catalysts. Angew. Chem. Int. Ed. 54, 1744–1748 (2015).
Bordeaux, M., Singh, R. & Fasan, R. Intramolecular C(sp3)–H amination of arylsulfonyl azides with engineered and artificial myoglobin-based catalysts. Bioorg. Med. Chem. 22, 5697–5704 (2014).
Pineda-Knauseder, A. J., Vargas, D. A. & Fasan, R. Organic solvent stability and long-term storage of myoglobin-based carbene transfer biocatalysts. Biotechnol. Appl. Biochem. 67, 516–526 (2020).
Deng, T. et al. Rh2(II)-catalyzed intermolecular N-aryl aziridination of olefins using nonactivated N atom precursors. J. Am. Chem. Soc. 143, 19149–19159 (2021).
Sangster, J. J., Marshall, J. R., Turner, N. J. & Mangas-Sanchez, J. New trends and future opportunities in the enzymatic formation of C–C, C–N, and C–O bonds. ChemBioChem 23, e202100464 (2022).
Mai, B. K., Neris, N. M., Yang, Y. & Liu, P. C–N bond forming radical rebound is the enantioselectivity-determining step in P411-catalyzed enantioselective C(sp3)–H amination: a combined computational and experimental investigation. J. Am. Chem. Soc. 144, 11215–11225 (2022).
Lang, K., Hu, Y., Lee, W.-C. L. & Zhang, X. P. Combined radical and ionic approach for the enantioselective synthesis of β-functionalized amines from alcohols. Nat. Synth. 1, 548–557 (2022).
Tinoco, A. et al. Origin of high stereocontrol in olefin cyclopropanation catalyzed by an engineered carbene transferase. ACS Catal. 9, 1514–1524 (2019).
Crombie, L., Haigh, D., Jones, R. C. F. & Matzin, A. R. Synthesis of the alkaloid homaline in (±) and natural (S,S)-(−) forms, using amination and transamidative ring expansion in liquid-ammonia. J. Chem. Soc. Perkin Trans. 1 2047–2054 (1993).
Chincholkar, P. M., Kale, A. S., Gumaste, V. K. & Deshmukh, A. R. A. S. An efficient formal synthesis of (S)-dapoxetine from enantiopure 3-hydroxy azetidin-2-one. Tetrahedron 65, 2605–2609 (2009).
Wang, J. P., Gao, H., Yang, L. J. & Gao, Y. Q. Role of engineered iron-haem enzyme in reactivity and stereoselectivity of intermolecular benzylic C–H bond amination. ACS Catal. 10, 5318–5327 (2020).
Kalita, S., Shaik, S. & Dubey, K. D. MD simulations and QM/MM calculations reveal the key mechanistic elements which are responsible for the efficient C–H amination reaction performed by a bioengineered P450 enzyme. Chem. Sci. 12, 14507–14518 (2021).
Bajaj, P., Sreenilayam, G., Tyagi, V. & Fasan, R. Gram-scale synthesis of chiral cyclopropane-containing drugs and drug precursors with engineered myoglobin catalysts featuring complementary stereoselectivity. Angew. Chem. Int. Ed. 55, 16110–16114 (2016).
Ren, X. K., Chandgude, A. L. & Fasan, R. Highly stereoselective synthesis of fused cyclopropane-γ-lactams via biocatalytic iron-catalyzed intramolecular cyclopropanation. ACS Catal. 10, 2308–2313 (2020).
Chandgude, A. L., Ren, X. & Fasan, R. Stereodivergent intramolecular cyclopropanation enabled by engineered carbene transferases. J. Am. Chem. Soc. 141, 9145–9150 (2019).
Acknowledgements
This work was supported by the US National Institute of Health (grant no. GM098628, to R.F.) and Cancer Prevention and Research Institute of Texas (CPRIT, RR230018 to R.F.). R.F. acknowledges endowed professorship support from the Robert A. Welch Foundation. D.A.V. acknowledges support from the National Science Foundation Graduate Fellowship Program. K.N.H. acknowledges support from the US National Science Foundation (grant no. CHE-1764328), and the Natural Science Foundation of China (grant no. 22103060) provided the computational resources used in the QM analyses. We are grateful to W. Brennessel (University of Rochester) for assistance with the crystallographic analyses. MS and X-ray instrumentation at the University of Rochester are supported by US National Science Foundation (grant nos. CHE-0946653 and CHE-1725028) and the US National Institute of Health (grant no. S10OD030302).
Author information
Authors and Affiliations
Contributions
D.A.V., S.R. and R.F. conceived the project and designed the experiments. S.R. and D.A.V. performed the experiments with guidance from R.F. K.N.H. mentored P.M., A.S. and L.Z. in the molecular dynamics and quantum mechanics calculations and contributed to the writing of the mechanistic parts of the paper. D.A.V., S.R., A.S.and R.F wrote the paper. All authors discussed the results and contributed to the final paper.
Corresponding authors
Ethics declarations
Competing interests
The authors have no competing interests.
Peer review
Peer review information
Nature Catalysis thanks Sabine Flitsch and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
Supplementary methods, Computational analyses, Crystal data collection, NMR spectra, tables, figures and references.
Supplementary Data
1Initial and final configurations of MD trajectories.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Roy, S., Vargas, D.A., Ma, P. et al. Stereoselective construction of β-, γ- and δ-lactam rings via enzymatic C–H amidation. Nat Catal 7, 65–76 (2024). https://doi.org/10.1038/s41929-023-01068-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41929-023-01068-2