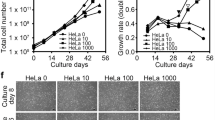
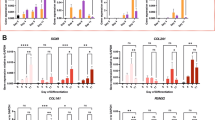
Abstract
Cartilage regeneration is a priority in medicine for the treatment of osteoarthritis and isolated cartilage defects. Several molecules with potential for cartilage regeneration are under investigation. Unfortunately, in vitro chondrogenesis assays do not always predict the stability of the newly formed cartilage in vivo. Therefore, there is a need for a stringent, quantifiable assay to assess in vivo the capacity of molecules to promote the stable formation of cartilage that is resistant to calcification and endochondral bone formation. We developed an ectopic cartilage formation assay (ECFA) that enables one to assess the capacity of bioactive molecules to support cartilage formation in vivo using cartilage organoids. The ECFA predicted good clinical outcomes when used as a quality control for efficacy of chondrocyte preparations before implantation in patients with cartilage defects. In this assay, articular chondrocytes from human donors or animals are injected either intramuscularly or subcutaneously in nude mice. As early as 2 weeks later, cartilage organoids can be retrieved. The size of the implants and their degree of differentiation can be assessed by histomorphometry, immunostainings of molecular markers and real-time PCR. Mineralization can be assessed by micro-computed tomography or by staining. The effects of molecules on cartilage formation can be tested following the systemic administration of the molecule in mice previously injected with chondrocytes, or after co-injection of chondrocytes with cell lines overexpressing and secreting the protein of interest. Here we describe the ECFA procedure, including steps for harvesting human and bovine articular cartilage, isolating primary chondrocytes, preparing overexpression cell lines, injecting the cells intramuscularly and retrieving the implants. This assay can be performed by technicians and researchers with appropriate animal training within 3 weeks.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
We are sorry, but there is no personal subscription option available for your country.
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
Data availability
The data that support the findings of this study are available from the corresponding authors upon request.
References
Hiligsmann, M. et al. Health economics in the field of osteoarthritis: an expert’s consensus paper from the European Society for Clinical and Economic Aspects of Osteoporosis and Osteoarthritis (ESCEO). Semin. Arthritis Rheum. 43, 303–313 (2013).
Decker, R. S., Koyama, E. & Pacifici, M. Genesis and morphogenesis of limb synovial joints and articular cartilage. Matrix Biol. 39, 5–10 (2014).
Sherwood, J. C., Bertrand, J., Eldridge, S. E. & Dell’Accio, F. Cellular and molecular mechanisms of cartilage damage and repair. Drug Discov. Today 19, 1172–1177 (2014).
Saito, T. et al. Transcriptional regulation of endochondral ossification by HIF-2α during skeletal growth and osteoarthritis development. Nat. Med. 16, 678–686 (2010).
Fuerst, M. et al. Calcification of articular cartilage in human osteoarthritis. Arthritis Rheum. 60, 2694–2703 (2009).
Kim, H. A. & Blanco, F. J. Cell death and apoptosis in osteoarthritic cartilage. Curr. Drug Targets 8, 333–345 (2007).
Goldring, S. R. & Goldring, M. B. Clinical aspects, pathology and pathophysiology of osteoarthritis. J. Musculoskelet. Neuronal Interact. 6, 376–378 (2006).
Messner, K. & Maletius, W. The long-term prognosis for severe damage to weight-bearing cartilage in the knee: a 14-year clinical and radiographic follow-up in 28 young athletes. Acta Orthop. Scand. 67, 165–168 (1996).
Shelbourne, K. D., Jari, S. & Gray, T. Outcome of untreated traumatic articular cartilage defects of the knee: a natural history study. J. Bone Joint Surg. 85-A(Suppl), 8–16 (2003).
Steadman, J. R., Rodkey, W. G. & Rodrigo, J. J. Microfracture: surgical technique and rehabilitation to treat chondral defects. Clin. Orthop. Relat. Res. 391(Suppl), S362–S369 (2001).
Brittberg, M. et al. Treatment of deep cartilage defects in the knee with autologous chondrocyte transplantation. N. Engl. J. Med. 331, 889–895 (1994).
Knutsen, G. et al. Autologous chondrocyte implantation compared with microfracture in the knee. A randomized trial. J. Bone Joint Surg. 86-A, 455–464 (2004).
Saris, D. B. F. et al. Characterized chondrocyte implantation results in better structural repair when treating symptomatic cartilage defects of the knee in a randomized controlled trial versus microfracture. Am. J. Sports Med. 36, 235–246 (2008).
Eldridge, S. et al. Agrin mediates chondrocyte homeostasis and requires both LRP4 and α-dystroglycan to enhance cartilage formation in vitro and in vivo. Ann. Rheum. Dis. 75, 1228–1235 (2016).
Dell’Accio, F., De Bari, C. & Luyten, F. P. Molecular markers predictive of the capacity of expanded human articular chondrocytes to form stable cartilage in vivo. Arthritis Rheum 44, 1608–1619 (2001).
De Bari, C., Dell’Accio, F. & Luyten, F. P. Failure of in vitro-differentiated mesenchymal stem cells from the synovial membrane to form ectopic stable cartilage in vivo. Arthritis Rheum. 50, 142–150 (2004).
Dell’Accio, F., Bari, C., De & Luyten, F. P. Microenvironment and phenotypic stability specify tissue formation by human articular cartilage-derived cells in vivo. Exp. Cell Res. 287, 16–27 (2003).
Nalesso, G. et al. WNT-3A modulates articular chondrocyte phenotype by activating both canonical and noncanonical pathways. J. Cell Biol. 193, 551–564 (2011).
Dell’Accio, F. et al. Expanded phenotypically stable chondrocytes persist in the repair tissue and contribute to cartilage matrix formation and structural integration in a goat model of autologous chondrocyte implantation. J. Orthop. Res. 21, 123–131 (2003).
Saris, D. B. F. et al. Treatment of symptomatic cartilage defects of the knee: characterized chondrocyte implantation results in better clinical outcome at 36 months in a randomized trial compared to microfracture. Am. J. Sports Med. 37(Suppl 1), 10S–19S (2009).
Crombleholme, T. M. Adenoviral-mediated gene transfer in wound healing. Wound Repair Regen. 8, 460–472 (2000).
Davidson, E. N. B. et al. Inducible chondrocyte-specific overexpression of BMP2 in young mice results in severe aggravation of osteophyte formation in experimental OA without altering cartilage damage. Ann. Rheum. Dis. 74, 1257–1264 (2014).
van der Kraan, P. M., Blaney Davidson, E. N. & van den Berg, W. B. Bone morphogenetic proteins and articular cartilage: to serve and protect or a wolf in sheep clothing’s? Osteoarthritis Cartilage 18, 735–741 (2010).
Greco, K. V. et al. Analyses on the mechanisms that underlie the chondroprotective properties of calcitonin. Biochem. Pharmacol. 91, 348–358 (2014).
Heywood, H. K., Nalesso, G., Lee, D. A. & Dell’accio, F. Culture expansion in low-glucose conditions preserves chondrocyte differentiation and enhances their subsequent capacity to form cartilage tissue in three-dimensional culture. Biores. Open Access 3, 9–18 (2014).
Hiramatsu, K. et al. Generation of hyaline cartilaginous tissue from mouse adult dermal fibroblast culture by defined factors. J. Clin. Invest. 121, 640–657 (2011).
Yasuhara, R. et al. Roles of beta-catenin signaling in phenotypic expression and proliferation of articular cartilage superficial zone cells. Lab Invest. 91, 1739–1752 (2011).
Dell’Accio, F. et al. Identification of the molecular response of articular cartilage to injury, by microarray screening: Wnt-16 expression and signaling after injury and in osteoarthritis 1. Arthritis Rheum 58, 1410–1421 (2008).
De Bari, C., Dell’Accio, F. & Luyten, F. P. Human periosteum-derived cells maintain phenotypic stability and chondrogenic potential throughout expansion regardless of donor age. Arthritis Rheum 44, 85–95 (2001).
Gosset, M., Berenbaum, F., Thirion, S. & Jacques, C. Primary culture and phenotyping of murine chondrocytes. Nat. Protoc. 3, 1253–1260 (2008).
Willert, K. et al. Wnt proteins are lipid-modified and can act as stem cell growth factors. Nature 423, 448–452 (2003).
Wozney, J. M. & Seeherman, H. J. Protein-based tissue engineering in bone and cartilage repair. Curr. Opin. Biotechnol. 15, 392–398 (2004).
Yin, W., Park, J.-I. & Loeser, R. F. Oxidative stress inhibits insulin-like growth factor-I induction of chondrocyte proteoglycan synthesis through differential regulation of phosphatidylinositol 3-Kinase–Akt and MEK–ERK MAPK signaling pathways. J. Biol. Chem. 284, 31972–31981 (2009).
Serafini, M. et al. Establishment of bone marrow and hematopoietic niches in vivo by reversion of chondrocyte differentiation of human bone marrow stromal cells. Stem Cell Res. 12, 659–672 (2014).
Hamada, D., Sampson, E. R., Maynard, R. D. & Zuscik, M. J. Surgical induction of posttraumatic osteoarthritis in the mouse. Methods Mol. Biol. 1130, 61–72 (2014).
Glasson, S. S., Blanchet, T. J. & Morris, E. A. The surgical destabilization of the medial meniscus (DMM) model of osteoarthritis in the 129/SvEv mouse. Osteoarthritis Cartilage 15, 1061–1069 (2007).
van der Kraan, P. M., Vitters, E. L., van Beuningen, H. M., van de Putte, L. B. & van den Berg, W. B. Degenerative knee joint lesions in mice after a single intra-articular collagenase injection. A new model of osteoarthritis. J. Exp. Pathol. 71, 19–31 (1990).
Ivanavicius, S. P. et al. Structural pathology in a rodent model of osteoarthritis is associated with neuropathic pain: increased expression of ATF-3 and pharmacological characterisation. Pain 128, 272–282 (2007).
Stoop, R. et al. Type II collagen degradation in spontaneous osteoarthritis in C57Bl/6 and BALB/c mice. Arthritis Rheum. 42, 2381–2389 (1999).
Griffin, T. M., Huebner, J. L., Kraus, V. B., Yan, Z. & Guilak, F. Induction of osteoarthritis and metabolic inflammation by a very high-fat diet in mice: effects of short-term exercise. Arthritis Rheum. 64, 443–453 (2012).
Asquith, D. L., Miller, A. M., McInnes, I. B. & Liew, F. Y. Animal models of rheumatoid arthritis. Eur. J. Immunol. 39, 2040–2044 (2009).
Eltawil, N., De, M., Bari, C., Achan, P., Pitzalis, C. & Dell’accio, F. A novel in vivo murine model of cartilage regeneration. Age and strain-dependent outcome after joint surface injury. Osteoarthritis Cartilage 17, 695–704 (2009).
Glasson, S. S., Chambers, M. G., Van Den Berg, W. B. & Little, C. B. The OARSI histopathology initiative—recommendations for histological assessments of osteoarthritis in the mouse. Osteoarthritis Cartilage 18, S17–S23 (2010).
Vanlauwe, J. et al. Five-year outcome of characterized chondrocyte implantation versus microfracture for symptomatic cartilage defects of the knee: early treatment matters. Am. J. Sports Med. 39, 2566–2574 (2011).
Thorup, A. S. et al. ROR2 blockade as a therapy for osteoarthritis. Sci. Transl. Med. 12, 3063 (2020).
Thomas, B. L. et al. WNT3A-loaded exosomes enable cartilage repair. J. Extracell. Vesicles 10, e12088 (2021).
Barbero, A. et al. Age related changes in human articular chondrocyte yield, proliferation and post-expansion chondrogenic capacity. Osteoarthritis Cartilage 12, 476–484 (2004).
Outerbridge, R. E. The etiology of chondromalacia patellae. J. Bone Joint Surg. Br. 43-B, 752–757 (1961).
Acknowledgements
This work was supported by grants from FOREUM, grant/award number 1016807; Versus Arthritis, grant/award number 21515 and 22628; Rosetrees Trust, grant/award numbers A589 and A2575; the MRC grant/award numbers MR/L022893/1 and MR/K013076/1; the St Bartholomew’s Hospital Medical College Trust (James Paget Studentship Award) and Versus Arthritis Centre for Osteoarthritis Pathogenesis grant number 20205. We also thank P. Achan and M. Ramachandran (Barts and the London National Health Service Trust, London, England, UK) for the supply of human knee samples, and C. Mole (School of Engineering and Materials Science, QMUL) for helping with the provision of the hooves.
Author information
Authors and Affiliations
Contributions
A-S.T., S.C., G.N. and S.E.E. designed and performed the co-injection experiments, analyzed the data and wrote the manuscript; B.L.T. performed experiments and contributed to writing the manuscript; Y.S. performed experiments; F.P.L. designed the ECFA and contributed to writing the manuscript; F.D.A. designed and developed the ECFA, supervised the co-injection experiments and contributed to writing the manuscript. F.D.A. and S.E.E. share senior authorship.
Corresponding authors
Ethics declarations
Competing interests
F.P.L and F.D.A. are co-inventors on patent WO2008061804 (A2): ‘In vivo assay and molecular markers for testing the phenotypic stability of cell populations, and selecting cell populations for autologous transplantation’. The other authors declare no competing interests.
Peer review
Peer review information
Lab Animal thanks the anonymous reviewers for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Thorup, AS., Caxaria, S., Thomas, B.L. et al. In vivo potency assay for the screening of bioactive molecules on cartilage formation. Lab Anim 51, 103–120 (2022). https://doi.org/10.1038/s41684-022-00943-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41684-022-00943-y
This article is cited by
-
From cells to organs: progress and potential in cartilaginous organoids research
Journal of Translational Medicine (2023)