Abstract
We describe a new taxon of terrestrial bird of the genus Aphrastura (rayaditos) inhabiting the Diego Ramírez Archipelago, the southernmost point of the American continent. This archipelago is geographically isolated and lacks terrestrial mammalian predators as well as woody plants, providing a contrasted habitat to the forests inhabited by the other two Aphrastura spp. Individuals of Diego Ramírez differ morphologically from Aphrastura spinicauda, the taxonomic group they were originally attributed to, by their larger beaks, longer tarsi, shorter tails, and larger body mass. These birds move at shorter distances from ground level, and instead of nesting in cavities in trees, they breed in cavities in the ground, reflecting different life-histories. Both taxa are genetically differentiated based on mitochondrial and autosomal markers, with no evidence of current gene flow. Although further research is required to define how far divergence has proceeded along the speciation continuum, we propose A. subantarctica as a new taxonomic unit, given its unique morphological, genetic, and behavioral attributes in a non-forested habitat. The discovery of this endemic passerine highlights the need to monitor and conserve this still-pristine archipelago devoid of exotic species, which is now protected by the recently created Diego Ramírez Islands-Drake Passage Marine Park.
Similar content being viewed by others
Introduction
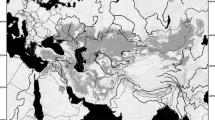
The genus Aphrastura (Passeriformes: Furnariidae) is endemic to southwestern South America and includes two allopatric species: the Thorn-tailed Rayadito (A. spinicauda) that inhabits the temperate forest biome of South America1, and the Masafuera Rayadito (A. masafuerae) whose distribution is limited to the misty tree fern forests of the oceanic Alejandro Selkirk Island2,3. This island has an area of only 85 km2 and is part of the Juan Fernández Archipelago, separated from the continent by 670 km. Unlike A. masafuerae, A. spinicauda has a broad distribution along the entire latitudinal range of the South American temperate forests’ biome4. It inhabits deciduous and evergreen forest types ranging from north-central Chile to the extreme south of Chile and Argentina5,6,7. Its northernmost population is found in the evergreen relict forest of Fray Jorge National Park (30° 30′ S), whilst its southerly population reaches the world’s southernmost forests in the Cape Horn Biosphere Reserve (CHBR) (56° S)7. However, a putative population of A. spinicauda also occurs on the Diego Ramírez Archipelago (hereafter Diego Ramírez) (56° 32′ S)8,9,10 (Fig. 1). Whether this population differs morphologically, ecologically, and genetically from its continental relatives remains unexplored.
Study areas for the morphological and genetic characterization of Aphrastura spinicauda and A. subantarctica. The distribution range of the nominal species is shown in light green, and the new taxonomic group from the Diego Ramírez Archipelago in light blue. (A) Sampling sites for morphology. (B) Sampling sites for mtDNA. (C) Sampling sites for microsatellite markers. The numbers correspond to the sample size. The names of the colored sites follow the methods description, and Tables 1 and 2. Bird illustrations by Mauricio Alvarez Abel.
The Diego Ramírez is the southernmost point of the South American continent. While emerging only ca. 100 km southwest of Cape Horn on the margin of the continental shelf, it is separated from it by one of the world’s roughest seas in the northern section of the Drake Passage, with harsh climatic conditions and difficult access from the continent11. The archipelago of small islets, rocks, and reefs provides a total land area of only 79 hectares12. The strong barrier to dispersal and the small size likely severely limit effective population migration to Diego Ramírez, thereby promoting the emergence of a fauna with zoogeographic novelties8. Their terrestrial avifauna is mainly limited to breeding seabirds, but despite the complete absence of woody plants (Fig. 2), it also includes passerines9, such as a resident population of Aphrastura spinicauda4. This population is currently considered to be conspecific with populations from the mainland (i.e., hereafter, we refer to them as Aphrastura populations from either the continent or Diego Ramírez). The geographic isolation and entirely herbaceous vegetation of Diego Ramírez raises the question whether its Aphrastura population constitutes a different taxonomic unit. This possibility is supported by the fact that two other populations of A. spinicauda that occur on islands have already been recognized as subspecies, based on their morphological distinctiveness, in terms of color and size, namely A. s. fulva from Chiloé Island (42° S) and the Chonos Archipelago (44° S), and A. s. bullocki from Mocha Island (38° S)4,13 (Supplementary Information Appendix 1). However, in contrast to the population inhabiting Diego Ramírez, both subspecies are geographically closer to the continental population and inhabit similar forest habitats.
Habitat characteristics and individual appearance of two populations of Aphrastura. (A) Forest habitat on Navarino Island. (B) An individual of the thorn-tailed rayadito (Aphrastura spinicauda) from Navarino Island. (C) Tussock (Poa flabellata) habitat on Gonzalo Island, Diego Ramírez Archipelago, with a mist net. (D,E) Individuals of the proposed new species A. subantarctica from Gonzalo Island. Images by Omar Barroso.
The two currently recognized species of Aphrastura (A. spinicauda and A. masafuerae) are restricted to forest ecosystems, including forest edges and surrounding woody vegetation14,15,16,17. The association with such habitats is striking in the northern range of A. spinicauda, where it is confined to forest patches and birds rarely venture into the arid shrubland18,19. Individuals usually forage for adult insects and larvae in the bark of trunks and branches, but they also glean them from the canopy and understory vegetation20. A. spinicauda typically occurs in Nothofagus-dominated forests, where they are one of the most abundant bird species, nesting in narrow cavities of old-growth trees that provide protection from rain, strong wind, and predators7. Given their strong association to forests, it is noticeable to find a population of Aphrastura on Diego Ramírez, whose vegetation is completely devoid of trees and other woody plants21,22,23.
Gene flow between the Diego Ramírez and the continental populations of Aphrastura is absent10, but whether this led to phenotypic divergence remains unknown. Here, we present an integrative taxonomic study combining morphological, ecological, and genetic data to clarify the taxonomic status of the Diego Ramírez population. We propose that this population is sufficiently distinct from continental populations to warrant taxonomic recognition, as the endemic Aphrastura subantarctica.
Methods
Study area
The Diego Ramírez Archipelago (56° S) is subject to an oceanic climate, characterized by an even rainfall throughout the year, low annual temperature range, and strong western winds21,22. The average annual rainfall is 1500 mm, and mean annual temperature is 5.6 °C, with August being the coldest month (3.3 °C on average) and March the warmest month (7.5 °C)24. The vegetation lacks trees and shrubs, and is dominated by tall (> 1 m) tussocks (Poa flabellata), a herbaceous plant that does not offer cavities for nesting8,22 (Fig. 2). Yet, dense aggregations of tussocks provide shelter from the strong and constant winds, such that the wind velocity drops to near zero at ground level. Also, inside the tussocks, temperatures can be up to 5 °C higher than above the plants23, creating a protected microhabitat that is used by the Aphrastura birds. Gonzalo Island is the second largest island of Diego Ramírez (38 ha), and is also an important nesting site for the Black-browed (Thalassarche melanophrys) and Grey-headed albatross (T. chrysostoma) (> 11,000 and > 5000 pairs, respectively25), which nest in openings of the tussock grassland.
To quantify morphological differences, we surveyed several sites on the north-western arm of the Beagle Channel on Tierra del Fuego Island and on Navarino Island (Fig. 1)26,27. These sites represent the southern limit of continental Aphrastura populations and are the closest forested habitat to Diego Ramírez. These areas are dominated by evergreen forests of Magellan's beech (Nothofagus betuloides) and Winter's bark (Drimys winteri)27,28. The influence of the ocean moderates thermal fluctuations, allowing the growth of a lush broadleaf evergreen forest. At the eastern end of the Beagle Channel, Navarino Island has an oceanic climate with a reduced annual rainfall of 460 mm, and snowfall mostly from fall to spring28. On this island, the high deciduous beech (N. pumilio) co-dominates with Magellan's beech, generating mixed forest of evergreen and deciduous trees. Nothofagus trees reach heights of 20 m and their straight trunks often have natural cavities that are used by the Aphrastura population for nesting7. Samples for the morphological analysis included individuals captured on the sites above mentioned (Fig. 1). For the genetic analyses, additional samples were obtained from across the distributional range of A. spinicauda (see below).
Bird capture and measurements
We obtained measurements of Aphrastura from Diego Ramírez and two areas of the CHBR, i.e., the northwest arm of the Beagle Channel and Navarino Island (Fig. 1). On Diego Ramírez, we sampled the population on Gonzalo Island (56° 32′ S, 68° 42′ W) on three expeditions: (1) November 29–December 2, 2016; (2) July 20–22, 2017; and (3) March 28—April 1, 2018. In the Beagle Channel, we sampled at four sites: (1) Pia sound (54° 47′ S, 69° 35′ W), (2) German glacier (54° 53′ S, 69° 24′ W), and (3) Olla Cove (54° 56′ S, 69° 09′ W), and (4) Contreras Cove (54° 50′ S, 68° 05′ W). The first three sites were visited between January 20 and 30 in 2015, and Contreras Cove from April 1 to 8, 2017. For Navarino Island, we used data from the Long-Term Ornithological Study Program7 at two sites: (5) the Omora Park (54° 06′ S, 67° 39′ W) collected from December 2015 to January 2016, and on April 1, 2018 (to compare with the 2018 Diego Ramírez expedition), and at (6) Guerrico (54° 54′ S, 67° 51′ W) in March 2015 and 2016. To avoid seasonal morphological variability, we restricted the morphological analysis to data obtained during the austral summer (November to April).
In all sites, birds were captured using 2–6 mist nets of mesh size 30 mm and 2.4 × 12 m long each. We operated the nets for 6-h periods after sunrise. Given the difficulties to operate the nets on Diego Ramírez (strong winds and dense herbaceous vegetation), we cleared a transect in the tussock to set the mist nets (Fig. 2). The nets were checked every 15–20 min. Birds were aged, banded, measured, and released following a protocol designed for the Ornithological Studies Program of the Omora Park7. We measured wing length, tarsus length, tail length, beak length and beak width using a caliper (0.01 mm precision), and we determined body mass using a digital scale (0.1 g precision). The same person (OB) performed all the measurements. Blood samples were obtained by puncturing the brachial vein and stored on FTA® cards for subsequent genetic analyses.
Observations of habitat use on Diego Ramírez
To compare the well-known habitat used by other Aphrastura populations, we recorded the habitat use of the Diego Ramírez Aphrastura individuals. In each austral spring and summer between 2010 and 2021, one observer (CGS) recorded all individuals along two transects (1.02 km length each) starting from the shoreline to the highest ridge of Gonzalo Island (129 m above sea level, masl). These transects included locations with different tussock density (50–100% coverage) and plant heights (0.4–2.0 m), as well as other bare sites with rocky or muddy substratum. These habitats are used as nesting sites by black-browed and grey-headed albatrosses. The transects also covered areas used by other surface-nesting seabirds, like the rockhopper (Eudyptes chrysocome), and macaroni penguin (E. chrysolophus), as well as burrowing seabird species, such as the Magellanic penguin (Spheniscus magellanicus), blue petrel (Halobaena caerulea) and the common diving petrel (Pelecanoides urinatrix). Nesting colonies of these sea birds modify the habitat in a way that may be relevant for the Diego Ramírez Aphrastura population. Based on the transects, we calculated the relative number of Aphrastura (birds/km) on each island. Based on all observations, we qualitatively described the habitat type used by Aphrastura (i.e., tussock coverage (%) and height (m), their nesting sites, and their behavior (foraging, territory defense, and their association with other bird species)). In addition, one observer (OB) carried out opportunistic observations during mist-netting, observing birds when walking in different directions to cover an area of approximately 0.16 km2 (~ 42% of the surface of Gonzalo Island).
Data analysis
Morphological analysis
To avoid age-related morphological differences, we only used measurements from adult individuals for this analysis. We compared trait values across the sites using a Kruskal–Wallis rank sum test and Conover's non-parametric all-pairs comparison test for a posteriori analysis (α = 0.05). To examine overall morphological differences among populations, we performed a Principal Components Analysis (PCA) including z scores for all measured variables. All analyses were performed using R29.
Molecular analysis and sequencing
We used blood samples from populations that covered the entire distributional range of A. spinicauda, from Fray Jorge National Park in north-central Chile to Navarino and Horn islands, and including nine individuals from Diego Ramírez (Fig. 1; Table 1). Total DNA was isolated from 100 µg of blood using the DNeasy Blood and Tissue kit from Qiagen (Germany) following the manufacturer’s protocol. PCR amplifications and bidirectional sequencing (Macrogen Inc., South Korea) of the mitochondrial cytochrome b gene (cytb) were carried out followed Gonzalez and Wink30. Consensus sequences of forward and reverse sequences were aligned using Pro-Seq 2.9131 against reference sequences30, and were visually checked.
Phylogeographic analysis
We used Arlequin 3.5.1.232 to estimate the number of haplotypes and polymorphic sites, gene diversity, differences between pairs of sequences (π) and nucleotide diversity (π per nucleotide site). For comparison of the four geographic groups (Fig. 1; Table 1), we used a rarefaction analysis with PAST33 to adjust for unequal sample sizes. We generated a haplotype network using the median-joining approach method34 implemented in Network 4.6. Using Arlequin 3.5.1.232, we estimated indices of genetic differentiation among geographic units based on both allele frequencies and pairwise nucleotide differences (pairwise FST and ФST, respectively). We tested for phylogeographic structure by comparing GST and NST coefficients using permutation tests implemented in PERMUT35. To infer the spatial genetic structure of Aphrastura populations, we used an analysis of molecular variance (AMOVA)36, defining groups of populations that are geographically homogeneous and maximally differentiated from each other.
Population genetic analysis
We performed further genetic analyses based on nuclear microsatellite markers to check the conclusions from the mitochondrial phylogeographic approach. We analyzed an additional sub-sample of 153 individuals from Central Chile, Southern Chile and Argentina, and Diego Ramírez (Fig. 1; Table 2). These individuals were originally genotyped for another study led by EB-D10, using 12 autosomal polymorphic loci37. The marker panel consisted of seven species-specific markers and five cross-species amplifying markers (see10,38). None of the loci showed evidence for deviations from Hardy–Weinberg equilibrium (all p > 0.1) and all had null allele frequencies < 0.05. We calculated measures of genetic diversity per population using the hierfstat package39 in R. To summarize overall genetic variability among individuals and to preliminarily assess genetic structure, we used a PCA as implemented in the R package adegenet40. To estimate the optimal (most likely) number of genetic clusters that were present in our sample, we used the ‘snapclust’ method in adegenet. This method relies on maximizing the likelihood of a number of panmictic populations by combining model-based and geometric approaches41. The best-supported number of groups was determined using the Akaike Information Criterion (AIC).
The resulting genetic clusters were subsequently used for population structure analyses. First, we used GenAlex 6.542 to calculate pairwise G-statistics for the genetic clusters identified, implementing both the Nei’s standardized index (G’ST(Nei)) and the Hedrick’s standardized index corrected for small samples (G″ST). Second, we calculated the posterior membership probabilities of all sampled individuals for the newly identified genetic clusters, using the probability of assignment obtained from the ‘snapclust’ method (see41). Finally, we performed a complementary analysis for inferring individual membership to each genetic group using GeneClass243 to identify first-generation migrants. Detection of migrants was carried out using Paetkau et al.’s44 criterion for likelihood computation (Lh) and Paetkau et al.’s45 resampling method for probability calculation, setting the default frequency for missing alleles at 0.01 and using 10 replicates with 100 simulated individuals each (α = 0.01).
Permits and ethical statement
The sampling methodology was approved by the Ethics Committee of the Sciences Faculty of the University of Chile, following guidelines from the Biosecurity Manual of CONICYT (version 2008) and Chilean law N°20380 about animal protection. Full permissions for sampling and animal ethics approval were granted by the Servicio Agrícola y Ganadero (SAG; permits N° 5158/2016, 8195/2016, 4209/2017, and 2667/2018), the Corporación Nacional Forestal (CONAF; Resolution N° 711/2014), Chile; and the Administración de Parques Nacionales (APN-DRPN research permit N° 1405), Argentina. This manuscript complies with the Animal Research: Reporting of In Vivo Experiments (ARRIVE) guidelines46.
Results
Comparison of morphology
We captured and measured 117 adult individuals of Aphrastura in the NW Beagle Channel (n = 50), Navarino Island (n = 54), and Diego Ramírez (n = 13). Morphological differences among populations were statistically supported for weight, tail length, tarsus length, beak length, and beak width (all p < 0.05, Fig. 3A–E), but not for wing length (p > 0.05, Fig. 3F). Birds from the Diego Ramírez population were significantly heavier and larger (with a longer and wider bill and longer tarsi), but they had a significantly shorter tail than birds from the other two populations (Fig. 3A–E). The first two axes of the PCA explained 63.2% of the variance in morphological traits (PC1 = 43.2%, PC2 = 20.0%). The PCA analysis shows that the Aphrastura populations of the Beagle Channel and Navarino Island overlap in body dimensions, whereas the individuals of the Diego Ramírez population form a clearly separate cluster (Fig. 4).
Comparison of body weight (A), tail length (B), beak length (C), beak width (D), tarsus length (E), and wing length (F) among Aphrastura populations. Metrics (means ± SD) of birds from the northwest arm of the Beagle Channel (NW Beagle, n = 50), Navarino Island (n = 54), and the population of the proposed new species A. subantarctica on Gonzalo Island, Diego Ramírez (n = 13). Lowercase letters indicate statistically significant differences at an alpha of 0.05.
Principal component analysis of morphological variation of populations of thorn-tailed Rayadito (Aphrastura spinicauda) in the northwest arm of the Beagle Channel (blue) and Navarino Island (green), and for the population of the proposed new species A. subantarctica on Gonzalo Island, Diego Ramírez (red). Plotted are the first two components from a principal component analysis based on 117 measured individuals. WL wing length, TsL tarsus length, TL tail length, BL beak length, BW beak width, Weight body mass.
Behavior and habitat use on Diego Ramírez
We estimated the relative abundance on Gonzalo Island as 47.1 birds/km (~ 48 birds per transect). Birds of Diego Ramírez typically performed short and low flights, rarely moving above the cover of the tussock grass. On Gonzalo Island, individuals always remained in the wind-protected habitat provided by the tall grass (< 2 m), except for brief horizontal movements to and from more open foraging areas.
We observed 10 nesting sites of Aphrastura on Gonzalo Island. Two of them were found in the areas with a higher concentration of breeding seabirds, which were close to the coast (11 masl), characterized by 50% coverage of small tussocks (~ 0.4 m high) and a rocky substrate. Three nesting sites were found in the higher and wind-exposed areas of the island (> 100 masl), but sheltered by 100% tussock coverage. Yet, most nesting sites (n = 5) were observed in the lower and medium altitude zones, particularly in the presence of protected creeks and areas with tussocks at least 1 m high. These five nesting sites were composed of burrows with single entrances. Four of these burrows were generated and abandoned by underground nesting seabirds, and one was a natural soil cavity. One Aphrastura nest was located below an active nest of an incubating Grey-headed albatross (for details see Supplementary Information Appendix 2).
On Gonzalo Island, Aphrastura typically foraged about 10–30 m away from the nesting site, capturing invertebrates. Foraging areas were covered by dense tussock or were located in the periphery of the tussock-matrix. We recorded pairs or groups of up to four individuals foraging together. Birds responded to a human intruder (the observer) by active flapping displays, vertical movements between the ground and the tussock canopy, and mobbing calls that started from 25 to 15 m away from the observer and involved pairs or groups of up to five individuals.
Phylogeographic patterns
We obtained 471 bp cytb sequences from 120 individuals across the distributional range of A. spinicauda (see sample sizes in Fig. 1). Overall mitochondrial genetic diversity was low, revealing a short genealogy of the cytb gene. Genetic diversity was larger in populations from southern Chile between 42 and 53° S (from Chiloé to Punta Arenas), compared to that of populations in the center and the north of the distribution, and also of the populations in the extreme southern part of the distribution (Navarino and Horn islands; Table 1). Noticeably, based on pairwise FST and ФST values, the Diego Ramírez population is strongly and significantly separated from all other populations, which exhibit little structure across the distribution range (from Fray Jorge in the north to Horn Island in the south; Table 3). The AMOVA showed that 69% of the total genetic variance was explained by clustering Diego Ramírez apart from all the other geographic groups (Table 4). We also found a strong phylogeographic structure between Diego Ramírez and the other groups, supported by NST coefficients (0.537 ± 0.136) being higher than GST values (0.320 ± 0.141; p < 0.001).
Individuals sampled from Diego Ramírez shared the exact same haplotype, which differed by one mutation from the most dominant haplotype found in A. spinicauda. The Diego Ramírez haplotype is also present on Horn Island (1 out of 3 individuals) and in low frequency in Navarino Island (1 out of 20 individuals) (Fig. 5).
Mitochondrial haplotype network of Aphrastura geographic groups. Each circle in the network corresponds to a different haplotype, the size of the circles corresponds to haplotype frequencies, and the colors correspond to the different geographical groups. CNS Central, north and south, PATAG Patagonia (Chiloé to Tierra del Fuego), NAVHOR Navarino and Horn islands, DIERAM Diego Ramírez (representing the proposed new species A. subantarctica).
Population genetic structure
Based on the genotypes of 153 individuals at 12 polymorphic microsatellite loci (see sample sizes in Fig. 1), genetic diversity was relatively high in all sampled populations, except the one from Diego Ramírez (Table 2). The latter exhibited the lowest heterozygosity, the highest within-population inbreeding coefficient, and the lowest allelic richness, both before and after rarefaction (Table 2).
For the PCA analysis of the five sampled populations, we retained the first 43 dimensions, which explained 85% of the total genetic variation. A plot with the first three components (~ 26% of variation explained) showed that Navarino Island and the southern continental populations (Bariloche and Tierra del Fuego) formed a homogeneous group (Fig. 6). The northernmost continental population (Manquehue, central Chile) was slightly separated, but relatively well mixed with the southern continental group (Fig. 6). In contrast, the Diego Ramírez population appeared well isolated from all other sampled localities, regardless of the combination of principal components being examined (Fig. 6).
Genetic variation among five populations of Aphrastura, based on 153 individuals genotyped at 12 polymorphic microsatellite loci. (A) Scatterplot from the first three principal components that explained 25.5% of the genetic variance. 95% CI ellipses are shown. (B) Individual assignment to genetic clusters for the five sampled populations. Bars represent individual posterior membership probabilities to each of the three genetic clusters found using the ‘snapclust’ method. MA Manquehue, BA Bariloche, TF Tierra del Fuego, NI Navarino Island, DR Diego Ramírez (representing the proposed new species A. subantarctica) .
According to the AIC values obtained from the ‘snapclust’ method, the optimal number of genetic clusters within the group of sampled individuals was three (Supplementary Information Appendix 3). This estimate is congruent with the number of clusters emerging from the PCA analysis: (1) a central cluster, comprising the Manquehue population in the center of the species’ distributional range; (2) a southern cluster, composed of Bariloche, Tierra del Fuego Island, and Navarino Island populations; and (3) Diego Ramírez. The pairwise G-statistics indicated that differentiation between the central and southern clusters was low to moderate (Table 5), whereas Diego Ramírez showed strong differentiation from the latter two, regardless of the GST index being considered (Table 5). The posterior membership probabilities calculated from the ‘snapclust’ showed that some individuals were assigned to a population different from the one from which they were captured. This was true for some birds in the central and southern clusters, but not for any individual from Diego Ramírez (100% correct assignment; Fig. 6). These results are in line with the analysis using GeneClass2, in which no first-generation migrants were detected in Diego Ramírez for any of the 10 replicates performed, whereas the southern cluster harbored four potential immigrants from the central cluster (Supplementary Information Appendix 4).
Characterization of the new species
Based on the strong genetic differentiation, as well as marked morphological and behavioral differences described above for the Diego Ramírez Aphrastura population, as compared to the continental Aphrastura population, we propose the following new bird species.
Order: Passeriformes (Linnaeus, 1758)47.
Family: Furnariidae (Gray, 1840)48.
Genus: Aphrastura (Oberholser, 1899)49.
Scientific name: Aphrastura subantarctica, sp. nov. R Rozzi, CS Quilodrán, E Botero-Delgadillo, RD Crego, C Napolitano, O Barroso, JC Torres-Mura & RA Vásquez.
Common name (English): Subantarctic Rayadito.
Common name (Spanish): Rayadito Subantártico.
Diagnosis: Morphology.—Typical Aphrastura structure with rounded wings, and an idiosyncratic tail morphology. Aphrastura’s distal third of the inner web of the rectrices is abruptly and deeply excised, giving the tips of the feathers a thornlike appearance. No other genus in the family has a similar tail structure. Aphrastura differs in these morphological characters from the phylogenetically closest related genera in the subfamily Synallaxinae present in southwestern South America: Leptasthenura and Sylviorthorhynchus50,51. In contrast to Aphrastura, Leptasthenura’s tail is not abruptly and deeply excised at the distal portion of the inner web of the rectrices; in Sylviorthorhynchus, the rectrices are denuded of barbs1. A. subantarctica differs from A. spinicauda, in having on average a larger and heavier body (~ 25%), a larger beak (~ 15%), a larger tarsus (~ 5%), and a shorter tail (~ 16%) (Fig. 3; Supplementary Information Appendix 5 and 6). The primaries and secondaries are greyish on the ventral side with whitish edges; the central rectrices are dark grey on the ventral side, but do not differ between the two species. Color terms using Munsell Color’s52 notation are shown in Appendix 7 (Supplementary Information).
Holotype.—Adult male, Museo Nacional de Historia Natural, in Santiago, Chile (MNHNCL/AVE Nº 5439), collected on 22 July 2017 by OB and JCTM on Gonzalo Island, Diego Ramírez Archipelago, Magallanes and Antarctic region, Chile, at 56.52141°S 68.71647°W.
Description of holotype.—Color descriptions follow Vaurie1. Crown, hind neck and face sooty black, divided by a broad and conspicuous dull orange superciliary streak that extends to the back and merges with the brown color of the back. Other upperparts are fulvous brown, with dusky dark edges becoming rufous and uniform on the rump. Underparts whitish on the throat, breast, and abdomen becoming fulvous on the flanks and undertail coverts. No brood patch; no cloacal protuberance, little fat (1), no evidence of molt on head, body, wing, or tail.
Measurements of holotype.—Total length 145 mm; head and bill length 32.4 mm; exposed culmen 12.1 mm; bill depth 3.4 mm; bill width 3.4 mm; wing chord 59 mm; tarsus length 21.8 mm; tail length along central rectrices 66 mm; mass 15.1 g.
Paratypes (two).—(1) Juvenile female, MNHNCL/AVE 5440, collected 22 July 2017 at Gonzalo Island, Diego Ramírez Archipelago by OB and JCTM; same coloration as the adult, tarsus and gape yellowish; white tips in primaries and secondaries. No brood patch; no cloacal protuberance, no fat (0), no head, body, wing, or tail molt. Total length 150 mm; head and bill length 33.6 mm; exposed culmen 12.6 mm; bill depth 3.6 mm; bill width 3.2 mm; wing chord 63 mm; tarsus length 23 mm; tail length along central rectrices 69 mm; mass 15.9 g.—(2) Juvenile male, MNHNCL/AVE 5441, collected 22 July 2017 on Gonzalo Island, Diego Ramírez Archipelago by OB and JCTM; same coloration as the adult, gape and tarsus yellowish; white tips in primaries and secondaries. No brood patch; no cloacal protuberance, no fat (0), no head, body, wing, or tail molt. Total length 150 mm; head and bill length 33.6 mm; exposed culmen 12.9 mm; bill depth 3.5 mm; bill width 3.3 mm; wing chord 59 mm; tarsus length 23 mm; tail length along central rectrices 72 mm; mass 15.9 g.
Etymology. The specific epithet refers to the distribution restricted to the remote sub-Antarctic Magellanic ecoregion of Chile. By naming the new species after this region, one of the last pristine regions in the world53, the authors encourage the adoption of this small bird as a symbol for the conservation of this unique environment.
Discussion
Oceans constitute a strong dispersal barrier for terrestrial animals but also for birds54. For small birds, such as Aphrastura spp., an island separated from the closest island (Horn Island) by 111 km in one of the world’s windiest oceans12, can limit exchange and hence favor population differentiation. One of the best-studied examples on islands separated by short distances is the radiation of the Darwin’s finches (Geospiza spp.) on the Galapagos Islands in the Pacific Ocean55. We studied several populations of Aphrastura spinicauda, a year-round resident of forested habitats that rarely engages in long-distance flights56,57,58,59,60. While several populations are located on southern islands, our results suggest they are not reproductively isolated, except for the population on Diego Ramírez. This population is genetically differentiated from populations inhabiting the closest islands on Tierra del Fuego and the mainland, with no evidence of recent gene flow. This may explain the morphological divergence of the population in terms of body weight, beak shape, and tarsus and tail length. In addition, the distinct environmental conditions on Diego Ramírez, in particular the lack of trees or any other woody plants, strong winds, and the presence of dense tussock used by burrow-nesting seabirds, might have triggered behavioral divergence related to living in a herbaceous environment, such as nesting in cavities below or close to ground level. This markedly contrasts with the tree creeping behavior and cavity nesting displayed by their continental relatives inhabiting forest ecosystems.
Morphological and behavioral differentiation
Individuals of Aphrastura on Diego Ramírez are on average heavier than their continental counterparts, and their tails are shorter. These morphological shifts might represent an adaptation to shorter and lower flights in dense tall grass habitats and windy conditions. These birds tend to fly mostly short distances among the Poa flabellata tussocks, which protect them from the strong winds. These movement patterns, shaped by the habitat conditions, conspicuously contrast with those of individuals from continental populations that move within the understory (< 1 m) to the top of the canopy (> 15 m), and often fly into open spaces while crossing gaps between tree patches7. On Diego Ramírez, restricted movement within a more sheltered habitat, close to the ground level, may have driven selection for shorter tails that may facilitate moving among dense tussock. Shorter tails could also have arisen through genetic drift after a small number of birds reached the archipelago or because other selective pressures for longer tails might have been relaxed in the environmental conditions of Diego Ramírez. A larger body size is typically found in birds that have colonized islands, and is a well-documented pattern of convergent evolution61. This has been described as the “island syndrome”, in which small birds (< 1 kg) are predicted to increase their body size in response to reduced predation pressure and food limitation under the new insular conditions62. Selection on fast escape from predators may be relaxed on the archipelago, as terrestrial predators are absent63. In addition, a larger body size may have been favored on Diego Ramírez because it increases thermoregulation efficiency64. While a larger body size has also been described in the subspecies (Aphrastura s. bullocki)13 and species (Aphrastura masafuerae)2,65,66 of Aphrastura inhabiting islands, the behavior related to grass habitat use and nesting at ground level seems to be unique to Aphrastura on Diego Ramírez. Another subspecies inhabiting Patagonian islands is Aphrastura s. fulva, but because the main differences from continental Aphrastura spinicauda are related to color patterns13, it is also expected to be morphologically and behaviorally different from Aphrastura on Diego Ramírez. The findings of our study are thus consistent with the suggestion made by François Vuilleumier thirty years ago67, who characterized Aphrastura spinicauda as a geographically variable species. He also pointed out that the individuals observed on Diego Ramírez were morphologically and behaviorally different from northern populations, and more similar to wrens (Troglodytidae) than to species ecologically equivalent to A. spinicauda, such as titmice (Paridae) or treecreepers (Certhiidae).
The population on Diego Ramírez uses a distinct habitat that lacks woody plants, in ways that are unusual when compared to the continental populations. The lack of trees with nesting cavities has led to an association with breeding seabirds, which nest on Diego Ramírez during the austral spring and summer. Aphrastura individuals use burrows of different seabird species, sometimes associating with larger seabirds (e.g., Grey-headed albatross), which may provide protection from weather and potential aerial predators (e.g., Striated caracara Phalcoboenus australis and the Chilean skua Stercorarius chilensis). Across the entire range of Aphrastura spinicauda, individuals respond to intruders near their nest sites with alarm calls. In the continental populations, mobbing calls have minimum and maximum frequencies of 2.82 and 13.01 kHz, respectively, with at least six notes per second in central and southern Chile68. However, based on a previous record in 2001, it seems that minimum and maximum frequencies of mobbing calls of Aphrastura subantarctica are lower, with the same number of notes (see69). While these preliminary observations have to be confirmed by future studies, the low call frequencies of Aphrastura on Diego Ramírez could be related to the high ambient noise, as well as their larger body size70.
Genetic differentiation
The genetic structure resulting from both mitochondrial and autosomal data suggests that the Aphrastura population from Diego Ramírez represents an isolated entity. The autosomal differentiation is substantial even when considering values of the G′ST(Nei) index, which is a more conservative statistic than Hedrick’s index71. Despite sharing a mitochondrial haplotype with populations from Navarino and the Horn islands, the Aphrastura population of Diego Ramírez is also differentiated at the mitochondrial level, as shown by the significant genetic distance between them and individuals in Navarino and the Horn islands. Furthermore, Diego Ramírez appeared as a genetically separated cluster in our analyses, indicating a complete absence of subsequent, or at least recent, migration to or from the archipelago. When excluding the Diego Ramírez individuals, the other populations of A. spinicauda have no clear genetic structure at the autosomal and mitochondrial level across their distribution, which is consistent with moderate to high levels of gene flow, as documented by other authors30. Our results support the recent observation of the absence of current gene flow between the Diego Ramírez population and their relatives inhabiting the continent and islands closer to it10. This population could thus be considered a separate evolutionary unit.
The sub-Antarctic Magellanic ecoregion experienced variations in sea level and volume of ice sheet during the Last Glacial Maximum (LGM, 18,000–28,000 years BP)72,73, which has resulted in a heterogeneous environment with high level of endemism across various biological groups74. The population on Diego Ramírez was probably established from a unique colonization event of a small group of individuals that originated from the southern Patagonian islands, which are geographically nearest to Diego Ramírez, such as Navarino or Horn islands, with one shared mitochondrial haplotype among them suggesting this historical event. In addition, the presence of a single cytb haplotype on Diego Ramírez suggests a strong founder effect, while the absence of new private haplotypes suggests a scenario of recent, post-LGM colonization of Diego Ramírez. The noticeably low levels of heterozygosity, allelic richness, and number of private alleles in the microsatellite data further supports this hypothesis. In evolutionary terms, the relatively short time that has passed since a post-LGM colonization event might have been sufficient for the Diego Ramírez population to genetically differentiate from A. spinicauda. The absence of the main mitochondrial haplotype present in all A. spinicauda populations, from the northernmost to the southernmost ones, suggests that the population on Diego Ramírez is undergoing a peripatric divergence from A. spinicauda.
A new taxonomic unit
Our analyses indicate that the Aphrastura population from Diego Ramírez compose a demographically independent population that should be considered as a new conservation and taxonomic unit. This population has remained isolated and encompasses specific morphological, genetic, and behavioral features, for which reason it is now proposed as a new species75. In his influential paper, de Queiroz76 reconciled the concept of species and stated that the only nonnegotiable condition for a species to be considered as such is to separately evolve from another lineage, which according to our data has taken place for the Aphrastura population in Diego Ramírez. Whether there is complete lineage sorting and intrinsic reproductive isolation among the Aphrastura populations or not will only show how far the diverging process has gone through the speciation continuum (e.g.,77).
We have presented multiple pieces of evidence suggesting the existence of a new species by combining morphological, ecological and genetic data, and we argue that this integrative framework is crucial for defining new taxonomic units. Acoustic analysis, a powerful tool for supporting species limits in suboscine passerines, including Furnariidae51, may provide more evidence of differentiation of the Diego Ramírez population. Further research may include samples from A. masafuerae and A. s. bullocki, as well as full genomes for evaluating the genomic landscape of divergence during the speciation process of the genus Aphrastura. Several other bird taxa with small genetic differences have been reclassified and subsequently considered as valid species, such as the common swift (Apus apus) and pallid swift (A. pallidus)78, as well as the greater spotted eagle (Clanga clanga) and lesser spotted eagle (C. pomarine)79, among others (see80).
Concluding remarks
We propose A. subantartica as a new species. The genetic, morphological, and ecological divergence of this population, which may have resulted from isolation on an island with a distinct habitat, is probably an ongoing evolutionary process. Because of the small size of the Diego Ramírez islands and the potential arrival of exotic mammal predators, it is pressing to protect this new endemic species from extinction. The Diego Ramírez Archipelago encompasses the southernmost extreme islands of the American continent and is free of invasive alien species. Measures should be put into place to keep exotic mammals, such as rats (Rattus rattus), domestic cats (Felis catus), and American minks (Neovison vison)–which are all present on other islands of the Cape Horn Biosphere Reserve24,81–off the Diego Ramírez islands. In particular, the rapid expansion of the American mink has impacted bird populations on other subantarctic islands that have evolved in the absence of terrestrial mammal predators82,83. Collaboration between the scientific community and other institutions, in particular the Chilean Navy, which has been regularly present on this archipelago since the establishment of the lighthouse on Gonzalo island in 1951, is critical for the success of scientific long-term monitoring and conservation programs9. In 2019, the Diego Ramírez-Drake Passage Marine Park was created by the Chilean government to protect one of the few archipelagos that is still free from the arrival of exotic species worldwide12. The description of A. subantarctica is also an appeal to the community to protect not only a population of a new species, but also to protect a remote natural laboratory that represents an opportunity to preserve the diversity of nature and its ecological and evolutionary processes.
Data availability
The dataset used in the analysis is publicly available on zenodo: https://doi.org/10.5281/zenodo.6983420.
References
Vaurie, C. Taxonomy and geographical distribution of the Furnariidae (Aves, Passeriformes). Bulletin of the AMNH; v. 166, article 1. Bull. Am. Museum Nat. Hist. 166, 1–357 (1980).
Hahn, I. & Römer, U. New observations of the Masafuera Rayadito Aphrastura masafuerae. Cotinga 6, 17–19 (1996).
Hahn, I. & Römer, U. Threatened avifauna of the Juan Fernández Archipelago, Chile: The impact of introduced mammals and conservation priorities. Cotinga 17, 66–72 (2002).
Remsen, J. V. Family Furnariidae (Ovenbirds) Vol 8 162–348 (Lynx Edicions, 2003).
Moreno, J., Merino, S., Lobato, E., Rodríguez-Gironés, M. A. & Vásquez, R. A. Sexual dimorphism and parental roles in the Thorn-tailed Rayadito (Furnariidae). Condor 109, 312–320 (2007).
Moreno, J., Merino, S., Vásquez, R. A. & Armesto, J. J. Breeding biology of the Thorn-tailed Rayadito (Furnariidae) in south-temperate rainforests of Chile. Condor 107, 69–77 (2005).
Rozzi, R. & Jiménez, J. Sub-Antarctic Magellanic Ornithology: The First Decade of Long-term Bird Studies at the Omora Ethnobotanical Park, Cape Horn Biosphere Reserve, Chile (Universidad de Magallanes, Chile-University of North Texas Press, 2014).
Schlatter, R. & Riveros, G. Historia natural del Archipiélago Diego Ramírez, Chile. Ser. Cie. Ina. 47, 87–112 (1997).
Barroso, O. et al. Scientific collaboration with the Chilean Navy for long-term ornithological studies in the Diego Ramírez Archipelago: First year-round monitoring of Gonzalo Island’s bird assemblage. Anal. Inst. Patagonia 48, 149–168 (2020).
Botero-Delgadillo, E. et al. Range-wide genetic structure in the thorn-tailed rayadito suggests limited gene flow towards peripheral populations. Sci. Rep. 10, 1–14 (2020).
Aguirre, F. et al. Gradientes climáticos y su influyente rol sobre los ecosistemas terrestres de la Reserva de Biosfera Cabo de Hornos, Chile. Anal. Inst. Patagonia (In press).
Rozzi, R. et al. Parque Marino Cabo de Hornos-Diego Ramírez, Informe Técnico para la Propuesta de Creación (Universidad de Magallanes, 2017).
Johnson, A. W. & Goodall, J. The Birds of Chile and Adjacent Regions of Argentina, Bolivia and Peru (Platt Establecimientos Graficos, 1967).
Tomasevic, J. A., Hodum, P. J. & Estades, C. F. On the ecology and conservation of the critically endangered Masafuera Rayadito (Aphrastura masafuerae). Ornitol. Neotrop. 21, 535–543 (2010).
Ippi, S., Anderson, C. B., Rozzi, R. & Elphick, C. S. Annual variation of abundance and composition in forest bird assemblages on Navarino Island, Cape Horn Biosphere Reserve, Chile. Ornitol. Neotrop. 20, 231–245 (2009).
Rozzi, R., Martínez, D., Willson, M. F. & Sabag, C. In Ecología de los Bosques Nativos de Chile (eds Armesto, J. J. et al.) 135–152 (Editorial Universitaria, 1996).
Hahn, I., Römer, U. & Schlatter, R. Distribution, habitat use, and abundance patterns of land bird communities on the Juan Fernández Islands, Chile. Ornitol. Neotrop. 16, 371–385 (2005).
Vergara, P. M. & Marquet, P. A. On the seasonal effect of landscape structure on a bird species: The thorn-tailed rayadito in a relict forest in northern Chile. Landsc. Ecol. 22, 1059–1071 (2007).
Kelt, D. A. et al. The avifauna of Bosque Fray Jorge National Park and Chile’s Norte Chico. J. Arid Environ. 126, 23–36 (2016).
Espíndola-Hernández, P., Castaño-Villa, G. J., Vásquez, R. A. & Quirici, V. Sex-specific provisioning of nutritious food items in relation to brood sex ratios in a non-dimorphic bird. Behav. Ecol. Sociobiol. 71, 65 (2017).
Pisano Valdés, E. & Schlatter, R. P. Vegetación y flora de las islas Diego Ramírez (Chile). 1. Características y relaciones de la flora Vascular. Anal. Inst. Patagonia 12, 183–194 (1981).
Pisano Valdés, E. & Schlatter, R. P. Vegetación y flora de las islas Diego Ramírez (Chile). 2. Comunidades vegetales vasculares. Anal. Inst. Patagonia 12, 195–204 (1981).
Mackenzie, R. et al. Vascular flora and vegetational types at the long-term socio-ecological studies site, Gonzalo Island, Diego Ramírez Archipelago (56°31’S), Chile. Anal. Inst. Patagonia 48, 139–148 (2020).
Rozzi, R. et al. Un centinela para el monitoreo del cambio climático y su impacto sobre la biodiversidad en la cumbre austral de América: La nueva red de estudios a largo Plazo Cabo de Hornos. Anal. Inst. Patagonia 48, 45–81 (2020).
Robertson, G. et al. Continued increase in the number of black-browed albatrosses (Thalassarche melanophris) at Diego Ramírez, Chile. Polar Biol. 40, 1035–1042 (2017).
Arroniz-Crespo, M. et al. Bryophyte-cyanobacteria associations during primary succession in recently deglaciated areas of Tierra del Fuego (Chile). PLoS One 9, e96081 (2014).
Rozzi, R. et al. Cape Horn Biosphere Reserve: A challenge for biodiversity conservation, and implementation of sustainable development in southernmost South America. Anal. Inst. Patagonia 36, 55–70 (2007).
Rozzi, R. et al. principles for biocultural conservation at the southern tip of the Americas: The approach of the Omora Ethnobotanical Park. Ecol. Soc. 11, 25 (2006).
R: A Language and Environment for Statistical Computing (R Foundation for Statistical Computing, 2019).
Gonzalez, J. & Wink, M. Genetic differentiation of the Thorn-tailed Rayadito Aphrastura spinicauda (Furnariidae: Passeriformes) revealed by ISSR profiles suggests multiple palaeorefugia and high recurrent gene flow. Ibis 152, 761–774 (2010).
Filatov, D. A. ProSeq: A software for preparation and evolutionary analysis of DNA sequence data sets. Mol. Ecol. Notes 2, 621–624 (2002).
Excoffier, L. & Lischer, H. E. Arlequin suite ver 3.5: A new series of programs to perform population genetics analyses under Linux and Windows. Mol. Ecol. Resour. 10, 564–567 (2010).
Hammer, Ø., Harper, D. A. & Ryan, P. D. PAST: Paleontological statistics software package for education and data analysis. Palaeontol. Electron. 4, 9 (2001).
Bandelt, H.-J., Forster, P. & Röhl, A. Median-joining networks for inferring intraspecific phylogenies. Mol. Biol. Evol. 16, 37–48 (1999).
Pons, O. & Petit, R. Measwring and testing genetic differentiation with ordered versus unordered alleles. Genetics 144, 1237–1245 (1996).
Excoffier, L., Smouse, P. E. & Quattro, J. M. Analysis of molecular variance inferred from metric distances among DNA haplotypes: Application to human mitochondrial DNA restriction data. Genetics 131, 479–491 (1992).
Botero-Delgadillo, E. et al. Variation in fine-scale genetic structure and local dispersal patterns between peripheral populations of a South American passerine bird. Ecol. Evol. 7, 8363–8378 (2017).
Botero-Delgadillo, E., Quirici, V., Vásquez, R. A. & Kempenaers, B. Heterozygosity-fitness correlations in a continental island population of Thorn-tailed Rayadito. J. Hered. 111, 628–639 (2020).
Goudet, J. & Jombart, T. hierfstat: Estimation and tests of hierarchical F-statistics. R package version 0.04-22. https://CRAN.R-project.org/package=hierfstat (2015).
Jombart, T. adegenet: A R package for the multivariate analysis of genetic markers. Bioinformatics 24, 1403–1405 (2008).
Beugin, M. P., Gayet, T., Pontier, D., Devillard, S. & Jombart, T. A fast likelihood solution to the genetic clustering problem. Methods Ecol. Evol. 9, 1006–1016 (2018).
Peakall, R. & Smouse, P. E. GENALEX 6: Genetic analysis in Excel. Population genetic software for teaching and research. Mol. Ecol. Notes 6, 288–295 (2006).
Piry, S. et al. GENECLASS2: A software for genetic assignment and first-generation migrant detection. J. Hered. 95, 536–539 (2004).
Paetkau, D., Calvert, W., Stirling, I. & Strobeck, C. Microsatellite analysis of population structure in Canadian polar bears. Mol. Ecol. 4, 347–354 (1995).
Paetkau, D., Slade, R., Burden, M. & Estoup, A. Genetic assignment methods for the direct, real-time estimation of migration rate: A simulation-based exploration of accuracy and power. Mol. Ecol. 13, 55–65 (2004).
Du Sert, N. P. et al. Reporting animal research: Explanation and elaboration for the ARRIVE guidelines 20. PLoS Biol. 18, e3000411 (2020).
Linnaeus, C. Systema Naturae per regna tria naturae. Secundum classes, ordines, genera, species, cum characteribus, differentiis, synonymis, locis. Vol. 1 (Impensis Direct Laurentii Salvii, 1758).
Gray, G. R. A List of the Genera of Birds, with an Indication of the Typical Species of Each Genus, Compiled from Various Sources (Richard and John E. Taylor, 1940).
Oberholser, H. C. Some untenable names in ornithology. Proc. Acad. Nat. Sci. Philadelphia 20, 201–216 (1899).
Derryberry, E. P. et al. Lineage diversification and morphological evolution in a large-scale continental radiation: The Neotropical ovenbirds and woodcreepers (Aves: Furnariidae). Evol. Int. J. Organ. Evol. 65, 2973–2986 (2011).
Fjeldsa, J., Christidis, L. & Ericson, P. G. The Largest Avian Radiation: The Evolution of Perching Birds, or the Order Passeriformes (Lynx Edicions, 2020).
Munsell Color Charts. Munsell Soil Color Charts (Munsell Color Company, 2000).
Mittermeier, R. A. et al. Wilderness and biodiversity conservation. Proc. Natl. Acad. Sci. 100, 10309–10313 (2003).
Lomolino, M., Riddle, B. & Whittaker, R. (Oxford University Press, 2016).
Whittaker, R. J. Island Biogeography: Ecology, Evolution, and Conservation (Oxford University Press, 1998).
Botero-Delgadillo, E. et al. Ecological and social correlates of natal dispersal in female and male Thorn-tailed Rayadito (Aphrastura spinicauda) in a naturally isolated and fragmented habitat. Auk Ornithol. Adv. 136, ukz016 (2019).
Botero-Delgadillo, E., Serrano, D., Orellana, N., Poblete, Y. & Vásquez, R. A. Effects of temperature and time constraints on the seasonal variation in nest morphology of the Thorn-tailed Rayadito (Aphrastura spinicauda). Emu-Austral Ornithol. 117, 181–187 (2017).
Cornelius, C. Spatial variation in nest-site selection by a secondary cavity-nesting bird in a human-altered landscape. Condor 110, 615–626 (2008).
Quilodrán, C. S., Estades, C. F. & Vásquez, R. A. Conspecific effect on habitat selection of a territorial cavity-nesting bird. Wilson J. Ornithol. 126, 534–543 (2014).
Quilodrán, C. S., Vásquez, R. A. & Estades, C. F. Nesting of the Thorn-tailed Rayadito (Aphrastura spinicauda) in a pine plantation in southcentral Chile. Wilson J. Ornithol. 124, 737–742 (2012).
Wright, N. A., Steadman, D. W. & Witt, C. C. Predictable evolution toward flightlessness in volant island birds. Proc. Natl. Acad. Sci. 113, 4765–4770 (2016).
Sandvig, E. M., Coulson, T. & Clegg, S. M. The effect of insularity on avian growth rates and implications for insular body size evolution. Proc. R. Soc. B 286, 20181967 (2019).
Reznick, D. N. et al. Eco-evolutionary feedbacks predict the time course of rapid life-history evolution. Am. Nat. 194, 671–692 (2019).
Clavel, J. & Morlon, H. Accelerated body size evolution during cold climatic periods in the Cenozoic. Proc. Natl. Acad. Sci. 114, 4183–4188 (2017).
Philippi, R. & Landbeck, L. Beitrage zur Fauna Chiles. Arch Naturgesch 32, 121–132 (1866).
Vaurie, C. Taxonomy and geographical distribution of the Furnariidae (Aves, Passeriformes). Bull. AMNH 166, 1 (1980).
Vuilleumier, F. A quantitative survey of speciation phenomena in Patagonian birds. Ornitol. Neotrop. 2, 5–28 (1991).
Ippi, S., Vasquez, R. A., van Dongen, W. F. & Lazzoni, I. Geographical variation in the vocalizations of the suboscine Thorn-tailed Rayadito Aphrastura spinicauda. Ibis 153, 789–805 (2011).
Imberti, S. Internet Bird Collection: Thorn-tailed Rayadito (Aphrastura spinicauda). https://macaulaylibrary.org/asset/204019791 (2001).
Mikula, P. et al. A global analysis of song frequency in passerines provides no support for the acoustic adaptation hypothesis but suggests a role for sexual selection. Ecol. Lett. 24, 477–486 (2021).
Meirmans, P. G. & Hedrick, P. W. Assessing population structure: FST and related measures. Mol. Ecol. Resour. 11, 5–18 (2011).
Davies, B. J. et al. The evolution of the Patagonian Ice Sheet from 35 ka to the present day (PATICE). Earth Sci. Rev. 204, 103152 (2020).
Lamy, F. et al. Glacial reduction and millennial-scale variations in Drake Passage throughflow. Proc. Natl. Acad. Sci. 112, 13496–13501 (2015).
Rozzi, R. et al. Integrating ecology and environmental ethics: Earth stewardship in the southern end of the Americas. Bioscience 62, 226–236 (2012).
Collins, R. & Cruickshank, R. H. The seven deadly sins of DNA barcoding. Mol. Ecol. Resour. 13, 969–975 (2013).
De Queiroz, K. Species concepts and species delimitation. Syst. Biol. 56, 879–886 (2007).
Sendell-Price, A. T. et al. The genomic landscape of divergence across the speciation continuum in island-colonising silvereyes (Zosterops lateralis). G3 Genes Genomes Genet. 10, 3147–3163 (2020).
Päckert, M., Martens, J., Wink, M., Feigl, A. & Tietze, D. T. Molecular phylogeny of Old World swifts (Aves: Apodiformes, Apodidae, Apus and Tachymarptis) based on mitochondrial and nuclear markers. Mol. Phylogenet. Evol. 63, 606–616 (2012).
Lerner, H. et al. Phylogeny and new taxonomy of the booted eagles (Accipitriformes: Aquilinae). Zootaxa 4216, 301–320 (2017).
De Silva, T. N., Peterson, A. T., Bates, J. M., Fernando, S. W. & Girard, M. G. Phylogenetic relationships of weaverbirds (Aves: Ploceidae): A first robust phylogeny based on mitochondrial and nuclear markers. Mol. Phylogenet. Evol. 109, 21–32 (2017).
Schüttler, E. et al. New records of invasive mammals from the sub-Antarctic Cape Horn Archipelago. Polar Biol. 42, 1093–1105 (2019).
Martin, A. & Richardson, M. Rodent eradication scaled up: Clearing rats and mice from South Georgia. Oryx 53, 27–35 (2019).
Schüttler, E., Klenke, R., McGehee, S., Rozzi, R. & Jax, K. Vulnerability of ground-nesting waterbirds to predation by invasive American mink in the Cape Horn Biosphere Reserve, Chile. Biol. Conserv. 142, 1450–1460 (2009).
Acknowledgements
This study was funded with Grants from the Sub-Antarctic Biocultural Conservation Program of the University of North Texas, University of Magallanes, the Cape Horn International Center (ANID CHIC-FB210018), the Institute of Ecology and Biodiversity of Chile (CONICYT PFB-23), and the Patagonia Mar y Tierra Working Group (The Pew Charitable Trust - Chile). Dr. Jaime E. Jiménez and Dr. Francisca Massardo provided comments on an early draft. We thank the support of the Omora Foundation, and FONDECYT 1140548 to RAV. C.N. thanks support from ANID PAI 77190064, and ANID/BASAL FB210006. CSQ acknowledges support from the Swiss National Science Foundation (N° P400PB_183930 and P5R5PB_203169). We are grateful to Sylvia Kuhn and Alexander Girg from the Max Planck Institute for Ornithology for help in the laboratory, and to Jaime A. Cursach and Maximiliano Daigre during fieldwork and ornithological records in Gonzalo Island. Fieldwork in protected areas was possible thanks to people from Parque Nacional Bosque Fray Jorge, Minera Los Pelambres, Estación Biológica Senda Darwin, Parque Nacional Nahuel Huapi, and Parque Natural Karukinka. We also express our gratitude for early communications by Professor Roberto P. Schlatter during his work in Diego Ramírez in the 1980s and for all the logistical and personnel support from the 3rd Naval Zone of the Chilean Navy. We finally thank Mauricio Alvarez Abel for making the color painting illustrations of Aphrastura subantarctica and A. spinicauda.
Author information
Authors and Affiliations
Contributions
R.R., R.A.V., E.B.-D. conceived the original idea. R.R., O.B., J.C.T.M., R.D.C., B.G., R.M., C.S. conducted fieldwork. C.S.Q., E.B.-D., R.D.C., C.N., C.B., S.I., V.Q., J.R.A., B.G., E.P., R.A.V., R.R., participated in data analysis and interpretation. R.R., C.S.Q., E.B.-D., R.D.C., B.G., R.A.V. prepared the draft version of the manuscript. All authors contributed in the form of discussion and critical comments.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Rozzi, R., Quilodrán, C.S., Botero-Delgadillo, E. et al. The Subantarctic Rayadito (Aphrastura subantarctica), a new bird species on the southernmost islands of the Americas. Sci Rep 12, 13957 (2022). https://doi.org/10.1038/s41598-022-17985-4
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-022-17985-4
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.