Abstract
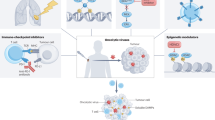
Local administration of oncolytic viruses to tumours can promote anticancer immune responses that lead to the abscopal regression of distant metastases, especially in patients receiving systemic immune-checkpoint inhibitors. Growing preclinical evidence indicates that non-virally induced oncolysis, defined as chemical or physical treatment administered locally to destroy malignant lesions, can promote a similar effect owing to the release of danger-associated molecular patterns that lead to the recruitment of immune cells, thus inducing a systemic response against tumour antigens that protects against local disease relapse and also mediates distant antineoplastic effects. An accumulating body of preclinical evidence supports the implementation of therapies that combine oncolysis with local or systemic immunotherapies. In this Review, we summarize the available data on innovative non-viral oncolysis strategies, including intratumorally applied cytotoxicants, photodynamic therapy, laser therapy, microwave, radiofrequency or photothermal ablation, high-intensity focused ultrasonography and cryotherapy for the local treatment of patients with solid tumours.
Key points
-
Oncolytic viruses are beginning to enter clinical use in patients with cancer despite practical considerations precluding their widespread use, highlighting the need for non-viral methods of oncolysis.
-
Multiple distinct physical and/or chemical methods of non-viral oncolysis are available. Each type of oncolysis causes a distinct type of cell stress and tissue destruction with variable immunological outcomes.
-
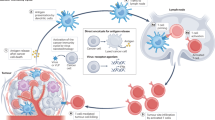
Immunogenic oncolysis initiates a local antitumour immune response through the destruction of immunosuppressive cells and the release of immunoactivatory danger-associated molecular patterns, leading to the recruitment of immune effectors.
-
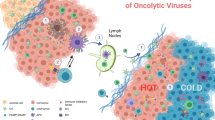
Various strategies for increasing the immune-mediated abscopal effects of oncolysis, including local or systemic immunotherapies, are being developed. The optimal schedule of such combination regimens must be carefully evaluated in future trials.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Galluzzi, L., Buque, A., Kepp, O., Zitvogel, L. & Kroemer, G. Immunological effects of conventional chemotherapy and targeted anticancer agents. Cancer Cell 28, 690–714 (2015).
Dadi, S. et al. Cancer immunosurveillance by tissue-resident innate lymphoid cells and innate-like T cells. Cell 164, 365–377 (2016).
Fridman, W. H., Zitvogel, L., Sautes-Fridman, C. & Kroemer, G. The immune contexture in cancer prognosis and treatment. Nat. Rev. Clin. Oncol. 14, 717–734 (2017).
Fucikova, J. et al. Calreticulin expression in human non-small cell lung cancers correlates with increased accumulation of antitumor immune cells and favorable prognosis. Cancer Res. 76, 1746–1756 (2016).
Pages, F. et al. International validation of the consensus immunoscore for the classification of colon cancer: a prognostic and accuracy study. Lancet 391, 2128–2139 (2018).
Weichselbaum, R. R., Liang, H., Deng, L. & Fu, Y. X. Radiotherapy and immunotherapy: a beneficial liaison? Nat. Rev. Clin. Oncol. 14, 365–379 (2017).
Postow, M. A. et al. Immunologic correlates of the abscopal effect in a patient with melanoma. N. Engl. J. Med. 366, 925–931 (2012).
De Meerleer, G. et al. Radiotherapy for renal-cell carcinoma. Lancet Oncol. 15, e170–e177 (2014).
Mothersill, C., Rusin, A., Fernandez-Palomo, C. & Seymour, C. History of bystander effects research 1905-present; what is in a name? Int. J. Radiat. Biol. 94, 696–707 (2018).
Ngwa, W. et al. Using immunotherapy to boost the abscopal effect. Nat. Rev. Cancer 18, 313–322 (2018).
Rodriguez-Ruiz, M. E., Vanpouille-Box, C., Melero, I., Formenti, S. C. & Demaria, S. Immunological mechanisms responsible for radiation-induced abscopal effect. Trends Immunol. 39, 644–655 (2018).
Lin, A. J., Roach, M., Bradley, J. & Robinson, C. Combining stereotactic body radiation therapy with immunotherapy: current data and future directions. Transl Lung Cancer Res. 8, 107–115 (2019).
Vanpouille-Box, C. et al. DNA exonuclease Trex1 regulates radiotherapy-induced tumour immunogenicity. Nat. Commun. 8, 15618 (2017).
Formenti, S. C. et al. Radiotherapy induces responses of lung cancer to CTLA-4 blockade. Nat. Med. 24, 1845–1851 (2018).
Kaufman, H. L., Kohlhapp, F. J. & Zloza, A. Oncolytic viruses: a new class of immunotherapy drugs. Nat. Rev. Drug Discov. 14, 642–662 (2015).
Achard, C. et al. Lighting a fire in the tumor microenvironment using oncolytic immunotherapy. EBioMedicine 31, 17–24 (2018).
Greig, S. L. Talimogene laherparepvec: first global approval. Drugs 76, 147–154 (2016).
Ribas, A. et al. Oncolytic virotherapy promotes intratumoral T cell infiltration and improves anti-PD-1 immunotherapy. Cell 170, 1109–1119.e10 (2017).
Chesney, J. et al. Randomized, open-label phase II study evaluating the efficacy and safety of talimogene laherparepvec in combination with ipilimumab versus ipilimumab alone in patients with advanced, unresectable melanoma. J. Clin. Oncol. 36, 1658–1667 (2018).
Twumasi-Boateng, K., Pettigrew, J. L., Kwok, Y. Y. E., Bell, J. C. & Nelson, B. H. Oncolytic viruses as engineering platforms for combination immunotherapy. Nat. Rev. Cancer 18, 419–432 (2018).
Bommareddy, P. K., Shettigar, M. & Kaufman, H. L. Integrating oncolytic viruses in combination cancer immunotherapy. Nat. Rev. Immunol. 18, 498–513 (2018).
G Pol, J. et al. Trial watch: oncolytic viro-immunotherapy of hematologic and solid tumors. Oncoimmunology 7, e1503032 (2018).
Harrington, K. J. et al. A practical guide to the handling and administration of talimogene laherparepvec in Europe. Onco Targets Ther. 10, 3867–3880 (2017).
Martin, C. Oncolytic viruses: treatment and implications for patients with gliomas. Clin. J. Oncol. Nurs. 21, 60–64 (2017).
Adkins, I., Fucikova, J., Garg, A. D., Agostinis, P. & Spisek, R. Physical modalities inducing immunogenic tumor cell death for cancer immunotherapy. Oncoimmunology 3, e968434 (2015).
Chu, K. F. & Dupuy, D. E. Thermal ablation of tumours: biological mechanisms and advances in therapy. Nat. Rev. Cancer 14, 199–208 (2014).
Slovak, R., Ludwig, J. M., Gettinger, S. N., Herbst, R. S. & Kim, H. S. Immuno-thermal ablations - boosting the anticancer immune response. J. Immunother. Cancer 5, 78 (2017).
Schneider, T. et al. Immune response after radiofrequency ablation and surgical resection in nonsmall cell lung cancer. Semin. Thorac. Cardiovasc. Surg. 28, 585–592 (2016).
Chen, Q. et al. Photothermal therapy promotes tumor infiltration and antitumor activity of CAR T cells. Adv. Mater. 31, e1900192 (2019).
den Brok, M. H. et al. Efficient loading of dendritic cells following cryo and radiofrequency ablation in combination with immune modulation induces anti-tumour immunity. Br. J. Cancer 95, 896–905 (2006).
Jansen, M. C. et al. Cryoablation induces greater inflammatory and coagulative responses than radiofrequency ablation or laser induced thermotherapy in a rat liver model. Surgery 147, 686–695 (2010).
Sabel, M. S. Cryo-immunology: a review of the literature and proposed mechanisms for stimulatory versus suppressive immune responses. Cryobiology 58, 1–11 (2009).
Kato, T. et al. Characterization of the cryoablation-induced immune response in kidney cancer patients. Oncoimmunology 6, e1326441 (2017).
Zhao, J. et al. Irreversible electroporation reverses resistance to immune checkpoint blockade in pancreatic cancer. Nat. Commun. 10, 899 (2019).
Pandit, H. et al. Evaluating the regulatory immunomodulation effect of irreversible electroporation (IRE) in pancreatic adenocarcinoma. Ann. Surg. Oncol. 26, 800–806 (2019).
White, S. B., Zhang, Z., Chen, J., Gogineni, V. R. & Larson, A. C. Early immunologic response of irreversible electroporation versus cryoablation in a rodent model of pancreatic cancer. J. Vasc. Interv. Radiol. 29, 1764–1769 (2018).
Calvet, C. Y., Famin, D., Andre, F. M. & Mir, L. M. Electrochemotherapy with bleomycin induces hallmarks of immunogenic cell death in murine colon cancer cells. Oncoimmunology 3, e28131 (2014).
Bugaut, H. et al. Bleomycin exerts ambivalent antitumor immune effect by triggering both immunogenic cell death and proliferation of regulatory T cells. PLOS ONE 8, e65181 (2013).
Casares, N. et al. Caspase-dependent immunogenicity of doxorubicin-induced tumor cell death. J. Exp. Med. 202, 1691–1701 (2005).
Tremble, L. F., O’Brien, M. A., Soden, D. M. & Forde, P. F. Electrochemotherapy with cisplatin increases survival and induces immunogenic responses in murine models of lung cancer and colorectal cancer. Cancer Lett. 442, 475–482 (2019).
Garg, A. D. et al. A novel pathway combining calreticulin exposure and ATP secretion in immunogenic cancer cell death. EMBO J. 31, 1062–1079 (2012).
Gomes-da-Silva, L. C. et al. Photodynamic therapy with redaporfin targets the endoplasmic reticulum and Golgi apparatus. EMBO J. 37, e98354 (2018).
Preise, D. et al. Systemic antitumor protection by vascular-targeted photodynamic therapy involves cellular and humoral immunity. Cancer Immunol. Immunother. 58, 71–84 (2009).
Kleinovink, J. W., Fransen, M. F., Lowik, C. W. & Ossendorp, F. Photodynamic-immune checkpoint therapy eradicates local and distant tumors by CD8+ T cells. Cancer Immunol. Res. 5, 832–838 (2017).
Cho, A., Jantschitsch, C. & Knobler, R. Extracorporeal photopheresis-an overview. Front. Med. 5, 236 (2018).
Edelson, R. et al. Treatment of cutaneous T-cell lymphoma by extracorporeal photochemotherapy. Preliminary results. N. Engl. J. Med. 316, 297–303 (1987).
Ventura, A. et al. Extracorporeal photochemotherapy drives monocyte-to-dendritic cell maturation to induce anticancer immunity. Cancer Res. 78, 4045–4058 (2018).
Habib, A. et al. Transarterial approaches to primary and secondary hepatic malignancies. Nat. Rev. Clin. Oncol. 12, 481–489 (2015).
Lee, H. L. et al. Inflammatory cytokines and change of Th1/Th2 balance as prognostic indicators for hepatocellular carcinoma in patients treated with transarterial chemoembolization. Sci. Rep. 9, 3260 (2019).
Obeid, M. et al. Calreticulin exposure is required for the immunogenicity of gamma-irradiation and UVC light-induced apoptosis. Cell Death Differ. 14, 1848–1850 (2007).
Chew, V. et al. Immune activation underlies a sustained clinical response to Yttrium-90 radioembolisation in hepatocellular carcinoma. Gut 68, 335–346 (2019).
Pasquereau-Kotula, E., Habault, J., Kroemer, G. & Poyet, J. L. The anticancer peptide RT53 induces immunogenic cell death. PLOS ONE 13, e0201220 (2018).
Zhou, H. et al. The oncolytic peptide LTX-315 triggers immunogenic cell death. Cell Death Dis. 7, e2134 (2016).
Zhou, H. et al. The oncolytic compound LTX-401 targets the Golgi apparatus. Cell Death Differ. 23, 2031–2041 (2016).
Zhou, H. et al. Oncolysis with DTT-205 and DTT-304 generates immunological memory in cured animals. Cell Death Dis. 9, 1086 (2018).
Nestvold, J. et al. Oncolytic peptide LTX-315 induces an immune-mediated abscopal effect in a rat sarcoma model. Oncoimmunology 6, e1338236 (2017).
Camilio, K. A. et al. Combining the oncolytic peptide LTX-315 with doxorubicin demonstrates therapeutic potential in a triple-negative breast cancer model. Breast Cancer Res. 21, 9 (2019).
Zitvogel, L., Tesniere, A. & Kroemer, G. Cancer despite immunosurveillance: immunoselection and immunosubversion. Nat. Rev. Immunol. 6, 715–727 (2006).
Schreiber, R. D., Old, L. J. & Smyth, M. J. Cancer immunoediting: integrating immunity’s roles in cancer suppression and promotion. Science 331, 1565–1570 (2011).
Palucka, A. K. & Coussens, L. M. The basis of oncoimmunology. Cell 164, 1233–1247 (2016).
Chaurasiya, S., Chen, N. G. & Fong, Y. Oncolytic viruses and immunity. Curr. Opin. Immunol. 51, 83–90 (2018).
Dvorak, H. F. Tumors: wounds that do not heal. Similarities between tumor stroma generation and wound healing. N. Engl. J. Med. 315, 1650–1659 (1986).
Bloy, N. et al. Immunogenic stress and death of cancer cells: contribution of antigenicity vs adjuvanticity to immunosurveillance. Immunol. Rev. 280, 165–174 (2017).
Obeid, M. et al. Calreticulin exposure dictates the immunogenicity of cancer cell death. Nat. Med. 13, 54–61 (2007).
Michaud, M. et al. Autophagy-dependent anticancer immune responses induced by chemotherapeutic agents in mice. Science 334, 1573–1577 (2011).
Martins, I. et al. Molecular mechanisms of ATP secretion during immunogenic cell death. Cell Death Differ. 21, 79–91 (2014).
Green, D. R., Ferguson, T., Zitvogel, L. & Kroemer, G. Immunogenic and tolerogenic cell death. Nat. Rev. Immunol. 9, 353–363 (2009).
Blander, J. M. & Medzhitov, R. Toll-dependent selection of microbial antigens for presentation by dendritic cells. Nature 440, 808–812 (2006).
Ali, M. Y. et al. Activation of dendritic cells by local ablation of hepatocellular carcinoma. J. Hepatol. 43, 817–822 (2005).
Fietta, A. M. et al. Systemic inflammatory response and downmodulation of peripheral CD25+Foxp3+T-regulatory cells in patients undergoing radiofrequency thermal ablation for lung cancer. Hum. Immunol. 70, 477–486 (2009).
Chen, T., Guo, J., Han, C., Yang, M. & Cao, X. Heat shock protein 70, released from heat-stressed tumor cells, initiates antitumor immunity by inducing tumor cell chemokine production and activating dendritic cells via TLR4 pathway. J. Immunol. 182, 1449–1459 (2009).
Sabel, M. S. et al. Immunologic response to cryoablation of breast cancer. Breast Cancer Res. Treat 90, 97–104 (2005).
Zitvogel, L., Galluzzi, L., Kepp, O., Smyth, M. J. & Kroemer, G. Type I interferons in anticancer immunity. Nat. Rev. Immunol. 15, 405–414 (2015).
Galluzzi, L., Buque, A., Kepp, O., Zitvogel, L. & Kroemer, G. Immunogenic cell death in cancer and infectious disease. Nat. Rev. Immunol. 17, 97–111 (2017).
Masopust, D. & Soerens, A. G. Tissue-resident T cells and other resident leukocytes. Annu. Rev. Immunol. 37, 521–546 (2019).
Tang, J. et al. Trial watch: the clinical trial landscape for PD1/PDL1 immune checkpoint inhibitors. Nat. Rev. Drug Discov. 17, 854–855 (2018).
Schmid, P. et al. Atezolizumab and nab-paclitaxel in advanced triple-negative breast cancer. N. Engl. J. Med. 379, 2108–2121 (2018).
Socinski, M. A. et al. Atezolizumab for first-line treatment of metastatic nonsquamous NSCLC. N. Engl. J. Med. 378, 2288–2301 (2018).
Pfirschke, C. et al. Immunogenic chemotherapy sensitizes tumors to checkpoint blockade therapy. Immunity 44, 343–354 (2016).
Liu, P. et al. Crizotinib-induced immunogenic cell death in non-small cell lung cancer. Nat. Commun. 10, 1486 (2019).
Rios-Doria, J. et al. Antibody-drug conjugates bearing pyrrolobenzodiazepine or tubulysin payloads are immunomodulatory and synergize with multiple immunotherapies. Cancer Res. 77, 2686–2698 (2017).
Yamazaki, T. et al. The oncolytic peptide LTX-315 overcomes resistance of cancers to immunotherapy with CTLA4 checkpoint blockade. Cell Death Differ. 23, 1004–1015 (2016).
Zhu, J. et al. Enhanced antitumor efficacy through microwave ablation in combination with immune checkpoints blockade in breast cancer: a pre-clinical study in a murine model. Diagn. Interv. Imaging 99, 135–142 (2018).
Shi, L. et al. PD-1 blockade boosts radiofrequency ablation-elicited adaptive immune responses against tumor. Clin. Cancer Res. 22, 1173–1184 (2016).
O’Shaughnessy, M. J. et al. Systemic antitumor immunity by PD-1/PD-L1 inhibition is potentiated by vascular-targeted photodynamic therapy of primary tumors. Clin. Cancer Res. 24, 592–599 (2018).
Adashek, J. J., Salgia, M., Dizman, N., Kessler, J. & Pal, S. K. Concomitant radioembolization and immune checkpoint inhibition in metastatic renal cell carcinoma. Case Rep. Oncol. 11, 276–280 (2018).
Wehrenberg-Klee, E., Goyal, L., Dugan, M., Zhu, A. X. & Ganguli, S. Y-90 radioembolization combined with a PD-1 inhibitor for advanced hepatocellular carcinoma. Cardiovasc. Intervent. Radiol. 41, 1799–1802 (2018).
Zheng, J., Irani, Z., Lawrence, D., Flaherty, K. & Arellano, R. S. Combined effects of yttrium-90 transarterial radioembolization around immunotherapy for hepatic metastases from uveal melanoma: a preliminary retrospective case series. J. Vasc. Interv. Radiol. 29, 1369–1375 (2018).
Waitz, R. et al. Potent induction of tumor immunity by combining tumor cryoablation with anti-CTLA-4 therapy. Cancer Res. 72, 430–439 (2012).
Mozzillo, N. et al. Assessing a novel immuno-oncology-based combination therapy: Ipilimumab plus electrochemotherapy. Oncoimmunology 4, e1008842 (2015).
Theurich, S. et al. Local tumor treatment in combination with systemic ipilimumab immunotherapy prolongs overall survival in patients with advanced malignant melanoma. Cancer Immunol. Res. 4, 744–754 (2016).
Xie, C. et al. Tremelimumab in combination with microwave ablation in patients with refractory biliary tract cancer. Hepatology 69, 2048–2060 (2019).
Behm, B. et al. Additive antitumour response to the rabbit VX2 hepatoma by combined radio frequency ablation and toll like receptor 9 stimulation. Gut 65, 134–143 (2016).
Lin, M. et al. An important discovery on combination of irreversible electroporation and allogeneic natural killer cell immunotherapy for unresectable pancreatic cancer. Oncotarget 8, 101795–101807 (2017).
Yang, Y. et al. Safety and short-term efficacy of irreversible electroporation and allogenic natural killer cell immunotherapy combination in the treatment of patients with unresectable primary liver cancer. Cardiovasc. Intervent. Radiol. 42, 48–59 (2019).
Bear, A. S. et al. Elimination of metastatic melanoma using gold nanoshell-enabled photothermal therapy and adoptive T cell transfer. PLOS ONE 8, e69073 (2013).
Marabelle, A. et al. Depleting tumor-specific Tregs at a single site eradicates disseminated tumors. J. Clin. Invest. 123, 2447–2463 (2013).
Marabelle, A., Tselikas, L., de Baere, T. & Houot, R. Intratumoral immunotherapy: using the tumor as the remedy. Ann. Oncol. 28, xii33–xii43 (2017).
Frank, M. J. et al. In situ vaccination with a TLR9 agonist and local low-dose radiation induces systemic responses in untreated indolent lymphoma. Cancer Discov. 8, 1258–1269 (2018).
Sagiv-Barfi, I. et al. Eradication of spontaneous malignancy by local immunotherapy. Sci. Transl Med. 10, eaan4488 (2018).
Hammerich, L. et al. Systemic clinical tumor regressions and potentiation of PD1 blockade with in situ vaccination. Nat. Med. 25, 814–824 (2019).
Li, L. et al. Microwave ablation combined with OK-432 induces Th1-type response and specific antitumor immunity in a murine model of breast cancer. J. Transl Med. 15, 23 (2017).
Lemdani, K. et al. Local immunomodulation combined to radiofrequency ablation results in a complete cure of local and distant colorectal carcinoma. Oncoimmunology 8, 1550342 (2019).
Veenstra, J. J. et al. Cryotherapy with concurrent CpG oligonucleotide treatment controls local tumor recurrence and modulates HER2/neu immunity. Cancer Res. 74, 5409–5420 (2014).
Machlenkin, A. et al. Combined dendritic cell cryotherapy of tumor induces systemic antimetastatic immunity. Clin. Cancer Res. 11, 4955–4961 (2005).
Alteber, Z. et al. Cryoimmunotherapy with local co-administration of ex vivo generated dendritic cells and CpG-ODN immune adjuvant, elicits a specific antitumor immunity. Cancer Immunol. Immunother. 63, 369–380 (2014).
Chua, C. Y. X. et al. Nanofluidic drug-eluting seed for sustained intratumoral immunotherapy in triple negative breast cancer. J. Control. Release 285, 23–34 (2018).
Luo, L. et al. Sustained release of anti-PD-1 peptide for perdurable immunotherapy together with photothermal ablation against primary and distant tumors. J. Control. Release 278, 87–99 (2018).
Chen, Q. et al. Photothermal therapy with immune-adjuvant nanoparticles together with checkpoint blockade for effective cancer immunotherapy. Nat. Commun. 7, 13193 (2016).
Andtbacka, R. H. et al. Patterns of clinical response with talimogene laherparepvec (T-VEC) in patients with melanoma treated in the optim phase III clinical trial. Ann. Surg. Oncol. 23, 4169–4177 (2016).
Chao, Y. et al. Combined local immunostimulatory radioisotope therapy and systemic immune checkpoint blockade imparts potent antitumour responses. Nat. Biomed. Eng. 2, 611–621 (2018).
Chen, Q. et al. In situ sprayed bioresponsive immunotherapeutic gel for post-surgical cancer treatment. Nat. Nanotechnol. 14, 89–97 (2019).
Abdo, J., Cornell, D. L., Mittal, S. K. & Agrawal, D. K. Immunotherapy plus cryotherapy: potential augmented abscopal effect for advanced cancers. Front. Oncol. 8, 85 (2018).
Fu, J. et al. STING agonist formulated cancer vaccines can cure established tumors resistant to PD-1 blockade. Sci. Transl Med. 7, 283ra52 (2015).
Zamarin, D. et al. Localized oncolytic virotherapy overcomes systemic tumor resistance to immune checkpoint blockade immunotherapy. Sci. Transl Med. 6, 226ra32 (2014).
Postel-Vinay, S. et al. Challenges of phase 1 clinical trials evaluating immune checkpoint-targeted antibodies. Ann. Oncol. 27, 214–224 (2016).
Skolnik, J. M., Barrett, J. S., Jayaraman, B., Patel, D. & Adamson, P. C. Shortening the timeline of pediatric phase I trials: the rolling six design. J. Clin. Oncol. 26, 190–195 (2008).
Turajlic, S., Sottoriva, A., Graham, T. & Swanton, C. Resolving genetic heterogeneity in cancer. Nat. Rev. Genet. 20, 404–416 (2019).
Angelova, M. et al. Evolution of metastases in space and time under immune selection. Cell 175, 751–765.e16 (2018).
Rosenthal, R. et al. Neoantigen-directed immune escape in lung cancer evolution. Nature 567, 479–485 (2019).
Galluzzi, L., Chan, T. A., Kroemer, G., Wolchok, J. D. & Lopez-Soto, A. The hallmarks of successful anticancer immunotherapy. Sci. Transl Med. 10, eaat7807 (2018).
Engelhard, V. H. et al. Immune cell infiltration and tertiary lymphoid structures as determinants of antitumor immunity. J. Immunol. 200, 432–442 (2018).
Lavin, Y. et al. Tissue-resident macrophage enhancer landscapes are shaped by the local microenvironment. Cell 159, 1312–1326 (2014).
Lavin, Y. et al. Innate immune landscape in early lung adenocarcinoma by paired single-cell analyses. Cell 169, 750–765.e17 (2017).
Lewis, K. L. et al. Notch2 receptor signaling controls functional differentiation of dendritic cells in the spleen and intestine. Immunity 35, 780–791 (2011).
Zitvogel, L., Ma, Y., Raoult, D., Kroemer, G. & Gajewski, T. F. The microbiome in cancer immunotherapy: diagnostic tools and therapeutic strategies. Science 359, 1366–1370 (2018).
Fend, L. et al. Immune checkpoint blockade, immunogenic chemotherapy or IFN-α blockade boost the local and abscopal effects of oncolytic virotherapy. Cancer Res. 77, 4146–4157 (2017).
Seremet, T. et al. Successful treatment with intralesional talimogene laherparepvec in two patients with immune checkpoint inhibitor-refractory, advanced-stage melanoma. Melanoma Res. 29, 85–88 (2019).
Woodland, D. L. Jump-starting the immune system: prime-boosting comes of age. Trends Immunol. 25, 98–104 (2004).
Vrankar, M. & Unk, M. Immune RECIST criteria and symptomatic pseudoprogression in non-small cell lung cancer patients treated with immunotherapy. Radiol. Oncol. 52, 365–369 (2018).
Andtbacka, R. H. et al. Talimogene laherparepvec improves durable response rate in patients with advanced melanoma. J. Clin. Oncol. 33, 2780–2788 (2015).
Marabelle, A. et al. Starting the fight in the tumor: expert recommendations for the development of human intratumoral immunotherapy (HIT-IT). Ann. Oncol. 29, 2163–2174 (2018).
Gujar, S., Pol, J. G., Kim, Y., Lee, P. W. & Kroemer, G. Antitumor benefits of antiviral immunity: an underappreciated aspect of oncolytic virotherapies. Trends Immunol. 39, 209–221 (2018).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/ct2/results?cond=&term=Cryosurgery%2C+Fuda+Cancer+Hospital%2C+Guangzhou.&cntry=CN&state=&city=&dist= (2019)
Acknowledgements
We thank E. Deutsch (GRCC) for critical reading. A.M. gratefully acknowledges financial support from BMS, Fondation MSD Avenir, INSERM CIC 1428 Biotherapies, the Institut National du Cancer (INCa) and the SIRIC Stratified Oncology Cell DNA Repair and Tumour Immune Elimination (SOCRATE). G.K. gratefully acknowledges financial support from the Agence National de la Recherche (ANR) – Projets blancs, ANR under the frame of E-Rare-2, the ERA-Net for Research on Rare Diseases, the Association pour la recherche sur le cancer (ARC), Cancéropôle Ile-de-France, Chancellerie des universités de Paris (Legs Poix), a donation from Elior, the European Research Area Network on Cardiovascular Diseases (ERA-CVD, MINOTAUR), the European Union Horizon 2020 Project Oncobiome, Fondation Carrefour; High-end Foreign Expert Program in China (GDW20171100085 and GDW20181100051), Fondation pour la Recherche Médicale (FRM), Gustave Roussy Odyssea, Inserm (HTE), the Institut National du Cancer (INCa), the Institut Universitaire de France, LabEx Immuno-Oncology, the LeDucq Foundation, the Ligue contre le Cancer (équipe labellisée), the RHU Torino Lumière, the Seerave Foundation, the SIRIC Cancer Research and Personalized Medicine (CARPEM), and the SIRIC Stratified Oncology Cell DNA Repair and Tumour Immune Elimination (SOCRATE).
Author information
Authors and Affiliations
Contributions
O.K. and G.K. researched data for the manuscript, all authors made a substantial contribution to discussions of content and wrote the manuscript, and O.K., L.Z. and G.K. reviewed and/or edited the manuscript prior to submission.
Corresponding author
Ethics declarations
Competing interests
G.K. and L.Z. have received research funding (contracts) from Lytix Ltd. A.M. has received funding from BMS, Roche and Transgene for academic sponsored clinical trials involving intratumoural immunotherapies (NCT02857569, NCT02977156 and NCT03892525) and has received honoraria and/or consulting fees from Amgen, AstraZeneca/Medimmune, Bioncotech, Boehringer Ingelheim, EISAI, Lytix pharma, MSD, Pillar Partners and Sanofi.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Glossary
- Oncolysis
-
The lysis of malignant cells using locally applied physical or chemical methods.
- Oncolytic virus
-
A virus designed to infect and kill cancer cells.
- Dendritic cells
-
(DCs). A specialized subset of myeloid cells with the capacity to process and present antigens to cells that mediate the adaptive immune response. Such cells engulf proportions of tumour cells, digest their proteins and present the resulting peptides in the context of MHC class I molecules to CD8+ T lymphocytes.
- Necrosis
-
An unregulated form of cell death accompanied by early rupture of the plasma membrane.
- Coagulation
-
Denaturation of proteins that clot together. This effect is usually induced by heat.
- Apoptosis
-
The best characterized form of regulated cell death, involving activation of caspases, shrinkage of cells and the generation of apoptotic bodies.
- CD8:FOXP3 ratio
-
The ratio of CD8 (mostly expressed by cytotoxic T lymphocytes) to FOXP3 (mostly expressed by regulatory T cells) expression, which indicates the local immune tonus. A high ratio suggests the presence of an active immune response, whereas a low ratio is suggestive of immunosuppression.
- Immunogenic cell death
-
(ICD). A form of cell death occurring in virally infected or cancer cells that stimulates an immune response against antigens released by dead or dying cells.
- Immunogenicity
-
The capacity to elicit a protective immune response.
- Necroptosis
-
A regulated form of cell death that is distinctly different to apoptosis and involves the activation of specific effector molecules such as RIPK3 and MLKL.
- Antigenicity
-
The presence of antigenic determinants that are absent in non-malignant cells and tissues and can be recognized by specific B and T lymphocytes.
- Adjuvanticity
-
The presence of danger-associated molecules that activate the effector cells of the innate immune system.
- Penumbra
-
The area of the tumour surrounding the region of central necrosis.
- Tumour-associated antigens
-
(TAAs). Proteins that are either structurally different (often owing to mutations) to those present in non-malignant cells or expressed by malignant but not by non-malignant cells.
- Toll-like receptor
-
(TLR). A pattern-recognition receptor that recognizes bacterial or viral motifs located within or on the surface of cells.
Rights and permissions
About this article
Cite this article
Kepp, O., Marabelle, A., Zitvogel, L. et al. Oncolysis without viruses — inducing systemic anticancer immune responses with local therapies. Nat Rev Clin Oncol 17, 49–64 (2020). https://doi.org/10.1038/s41571-019-0272-7
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41571-019-0272-7