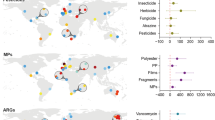
Abstract
Healthy, uncontaminated soils and clean water support all life on Earth and are essential for human health. Chemical pollution of soil, water, air and food is a major environmental threat, leading to an estimated 9 million premature deaths worldwide. The Global Burden of Disease study estimated that pollution was responsible for 5.5 million deaths related to cardiovascular disease (CVD) in 2019. Robust evidence has linked multiple pollutants, including heavy metals, pesticides, dioxins and toxic synthetic chemicals, with increased risk of CVD, and some reports suggest an association between microplastic and nanoplastic particles and CVD. Pollutants in soil diminish its capacity to produce food, leading to crop impurities, malnutrition and disease, and they can seep into rivers, worsening water pollution. Deforestation, wildfires and climate change exacerbate pollution by triggering soil erosion and releasing sequestered pollutants into the air and water. Despite their varied chemical makeup, pollutants induce CVD through common pathophysiological mechanisms involving oxidative stress and inflammation. In this Review, we provide an overview of the relationship between soil and water pollution and human health and pathology, and discuss the prevalence of soil and water pollutants and how they contribute to adverse health effects, focusing on CVD.
Key points
-
Illnesses related to chemical pollution of the soil, water and air are responsible for an estimated 9 million premature deaths annually, which equates to 16% of all global deaths; half of these deaths are of cardiovascular origin.
-
Degradation of the soil threatens the health of at least 3.2 billion people (40% of the global population), whereas >2 billion people live in water-stressed countries (25% of the global population).
-
Eco-disruptive causes of soil and water pollution include deforestation, climate change, airborne dust, over-fertilization and unhealthy city designs.
-
Pollution by heavy metals, pesticides, and microplastics and nanoplastics causes cardiovascular damage by interacting with protein-bound thiols, inducing oxidative stress and inflammation, and impairing circadian rhythms.
-
Exposure to chemicals (such as heavy metals, solvents, dioxins and pesticides) at workplaces, through consumer products or indirectly via environmental contamination contributes to endothelial dysfunction and cardiovascular disease.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
References
Fuller, R. et al. Pollution and health: a progress update. Lancet Planet Health 6, e535–e547 (2022).
Landrigan, P. J. et al. The Lancet Commission on Pollution and Health. Lancet 391, 462–512 (2018).
Munzel, T., Hahad, O., Daiber, A. & Landrigan, P. J. Soil and water pollution and human health: what should cardiologists worry about? Cardiovasc. Res. 119, 440–449 (2023).
GBD 2015 DALYs and HALE Collaborators Global, regional, and national disability-adjusted life-years (DALYs) for 315 diseases and injuries and healthy life expectancy (HALE), 1990-2015: a systematic analysis for the Global Burden of Disease Study 2015. Lancet 388, 1603–1658 (2016).
Institute for Health Metrics and Evaluation. Global health data exchange tool. Global Health Data Exchange http://ghdx.healthdata.org/gbd-results-tool (2023).
Lelieveld, J. et al. Cardiovascular disease burden from ambient air pollution in Europe reassessed using novel hazard ratio functions. Eur. Heart J. 40, 1590–1596 (2019).
Lelieveld, J. et al. Air pollution deaths attributable to fossil fuels: observational and modelling study. BMJ 383, e077784 (2023).
World Health Organization. The public health impact of chemicals: knowns and unknowns – data addendum for 2019. WHO https://www.who.int/publications/i/item/WHO-HEP-ECH-EHD-21.01 (2021).
World Health Organization. Global action plan for the prevention and control of non-communicable diseases 2013–2020. WHO https://www.who.int/publications/i/item/9789241506236 (2013).
United Nations Environment Program. Global assessment of soil pollution. UNEP https://www.unep.org/resources/report/global-assessment-soil-pollution (2021).
United Nations Environment Program. Soil pollution a risk to our health and food security. UNEP https://www.unep.org/news-and-stories/story/soil-pollution-risk-our-health-and-food-security (2020).
Mielke, H. W. & Reagan, P. L. Soil is an important pathway of human lead exposure. Environ. Health Perspect. 106, 217–229 (1998).
European Envrionment Agency. Water Pollution and Health. EEA https://www.eea.europa.eu/publications/zero-pollution/health/water-pollution (2022).
United Nations. Water. summary progress update 2021: SDG 6 – Water and sanitation for all. UN-Water http://www.unwater.org/sites/default/files/app/uploads/2021/12/SDG-6-Summary-Progress-Update-2021_Version-July-2021a.pdf (2021).
Global Allliance on Health and Pollution. Pollution and health metrics. GAHP https://gahp.net/wp-content/uploads/2019/12/PollutionandHealthMetrics-final-12_18_2019.pdf (2019).
World Health Organization. The public health impact of chemicals: knowns and unknowns. WHO http://www.who.int/publications/i/item/WHO-FWC-PHE-EPE-16.01-eng (2016).
World Health Organization. Food safety. WHO http://www.who.int/news-room/fact-sheets/detail/food-safety (2022).
European Commission. The European Human Biomonitoring Initiative. HBM4EU https://www.hbm4eu.eu/ (2016).
Centers for Disease Control and Prevention. National biomonitoring program. CDC https://www.cdc.gov/biomonitoring/index.html (2018).
Persson, L. et al. Outside the safe operating space of the planetary boundary for novel entities. Environ. Sci. Technol. 56, 1510–1521 (2022).
United Nations Environment Program. Global chemicals outlook II – from legacies to innovative solutions: Implementing the 2030 Agenda for Sustainable Development (Part I). UNEP https://www.unep.org/topics/chemicals-and-pollution-action/chemicals-management/global-chemicals-outlook (2019).
Maertens, A., Golden, E. & Hartung, T. Avoiding regrettable substitutions: green toxicology for sustainable chemistry. ACS Sustain. Chem. Eng. 9, 7749–7758 (2021).
Peters, A., Nawrot, T. S. & Baccarelli, A. A. Hallmarks of environmental insults. Cell 184, 1455–1468 (2021).
La Merrill, M. A. et al. Consensus on the key characteristics of endocrine-disrupting chemicals as a basis for hazard identification. Nat. Rev. Endocrinol. 16, 45–57 (2020).
Cosselman, K. E., Navas-Acien, A. & Kaufman, J. D. Environmental factors in cardiovascular disease. Nat. Rev. Cardiol. 12, 627–642 (2015).
Liu, Z., Lu, Y., Zhong, K., Wang, C. & Xu, X. The associations between endocrine disrupting chemicals and markers of inflammation and immune responses: a systematic review and meta-analysis. Ecotoxicol. Environ. Saf. 234, 113382 (2022).
Chen, J. C. et al. Associations of per- and polyfluoroalkyl substances, polychlorinated biphenyls, organochlorine pesticides, and polybrominated diphenyl ethers with oxidative stress markers: a systematic review and meta-analysis. Environ. Res. 239, 117308 (2023).
Daiber, A. et al. Targeting vascular (endothelial) dysfunction. Br. J. Pharmacol. 174, 1591–1619 (2017).
Wenzel, P., Kossmann, S., Munzel, T. & Daiber, A. Redox regulation of cardiovascular inflammation – immunomodulatory function of mitochondrial and Nox-derived reactive oxygen and nitrogen species. Free Radic. Biol. Med. 109, 48–60 (2017).
Vogel, C. F. A., Van Winkle, L. S., Esser, C. & Haarmann-Stemmann, T. The aryl hydrocarbon receptor as a target of environmental stressors – implications for pollution mediated stress and inflammatory responses. Redox Biol. 34, 101530 (2020).
Martinez-Pinna, J. et al. Endocrine disruptors in plastics alter β-cell physiology and increase the risk of diabetes mellitus. Am. J. Physiol. Endocrinol. Metab. 324, E488–E505 (2023).
Sule, R. O., Condon, L. & Gomes, A. V. A common feature of pesticides: oxidative stress – the role of oxidative stress in pesticide-induced toxicity. Oxid. Med. Cell Longev. 2022, 5563759 (2022).
Rasin, P. & Sreekanth, A. Cadmium exposure and cardiovascular diseases. Chem. Res. Toxicol. 36, 1441–1443 (2023).
Singh, N., Kumar, A., Gupta, V. K. & Sharma, B. Biochemical and molecular bases of lead-induced toxicity in mammalian systems and possible mitigations. Chem. Res. Toxicol. 31, 1009–1021 (2018).
Hu, H., Chen, Y., Jing, L., Zhai, C. & Shen, L. The link between ferroptosis and cardiovascular diseases: a novel target for treatment. Front. Cardiovasc. Med. 8, 710963 (2021).
Farina, M., Aschner, M. & Rocha, J. B. Oxidative stress in MeHg-induced neurotoxicity. Toxicol. Appl. Pharmacol. 256, 405–417 (2011).
Kempuraj, D. et al. Mercury induces inflammatory mediator release from human mast cells. J. Neuroinflammation 7, 20 (2010).
Ellinsworth, D. C. Arsenic, reactive oxygen, and endothelial dysfunction. J. Pharmacol. Exp. Ther. 353, 458–464 (2015).
Jomova, K. & Valko, M. Advances in metal-induced oxidative stress and human disease. Toxicology 283, 65–87 (2011).
Gaetke, L. M. & Chow, C. K. Copper toxicity, oxidative stress, and antioxidant nutrients. Toxicology 189, 147–163 (2003).
Kang, Y. J. Copper and homocysteine in cardiovascular diseases. Pharmacol. Ther. 129, 321–331 (2011).
Yang, S. et al. Copper homeostasis and cuproptosis in atherosclerosis: metabolism, mechanisms and potential therapeutic strategies. Cell Death Discov. 10, 25 (2024).
Dai, H., Wang, L., Li, L., Huang, Z. & Ye, L. Metallothionein 1: a new spotlight on inflammatory diseases. Front. Immunol. 12, 739918 (2021).
Paniagua, L., Diaz-Cueto, L., Huerta-Reyes, M. & Arechavaleta-Velasco, F. Cadmium exposure induces interleukin-6 production via ROS-dependent activation of the ERK1/2 but independent of JNK signaling pathway in human placental JEG-3 trophoblast cells. Reprod. Toxicol. 89, 28–34 (2019).
Zheng, X., Zhang, K., Zhao, Y. & Fent, K. Environmental chemicals affect circadian rhythms: an underexplored effect influencing health and fitness in animals and humans. Environ. Int. 149, 106159 (2021).
Li, H. et al. Influence of mental stress and environmental toxins on circadian clocks: implications for redox regulation of the heart and cardioprotection. Br. J. Pharmacol. 177, 5393–5412 (2020).
Lim, F. L., Currie, R. A., Orphanides, G. & Moggs, J. G. Emerging evidence for the interrelationship of xenobiotic exposure and circadian rhythms: a review. Xenobiotica 36, 1140–1151 (2006).
Parmalee, N. L. & Aschner, M. Metals and circadian rhythms. Adv. Neurotoxicol. 1, 119–130 (2017).
Lafuente, A. The hypothalamic-pituitary-gonadal axis is target of cadmium toxicity. An update of recent studies and potential therapeutic approaches. Food Chem. Toxicol. 59, 395–404 (2013).
Agency for Toxic Substances and Disease Registry. Interaction profile for: benzene, toluene, ethylbenzene, and xylenes (BTEX) (US Department of Health and Human Services, 2004). https://www.atsdr.cdc.gov/interactionprofiles/ip-btex/ip05.pdf.
Drwal, E., Rak, A. & Gregoraszczuk, E. L. Review: Polycyclic aromatic hydrocarbons (PAHs) – action on placental function and health risks in future life of newborns. Toxicology 411, 133–142 (2019).
Institute for Health and Consumer Protection. Summary risk assessment report: Phenol. EUR 22522 EN/1 (Publications Office of the European Union, 2006). https://echa.europa.eu/documents/10162/3e04f30d-9953-4824-ba04-defa32a130fa.
Galbraith, D., Gross, S. A. & Paustenbach, D. Benzene and human health: a historical review and appraisal of associations with various diseases. Crit. Rev. Toxicol. 40, 1–46 (2010).
Wang, Y. L., Lee, Y. H., Chiu, I. J., Lin, Y. F. & Chiu, H. W. Potent impact of plastic nanomaterials and micromaterials on the food chain and human health. Int. J. Mol. Sci. 21, 1727 (2020).
Jamieson, A. J., Malkocs, T., Piertney, S. B., Fujii, T. & Zhang, Z. Bioaccumulation of persistent organic pollutants in the deepest ocean fauna. Nat. Ecol. Evol. 1, 51 (2017).
Landrigan, P. J. & Goldman, L. R. Children’s vulnerability to toxic chemicals: a challenge and opportunity to strengthen health and environmental policy. Health Aff. 30, 842–850 (2011).
Lind, L. & Lind, P. M. Can persistent organic pollutants and plastic-associated chemicals cause cardiovascular disease? J. Intern. Med. 271, 537–553 (2012).
Dunder, L. et al. Plasma levels of per- and polyfluoroalkyl substances (PFAS) and cardiovascular disease – results from two independent population-based cohorts and a meta-analysis. Environ. Int. 181, 108250 (2023).
Honda-Kohmo, K., Hutcheson, R., Innes, K. E. & Conway, B. N. Perfluoroalkyl substances are inversely associated with coronary heart disease in adults with diabetes. J. Diabetes Complications 33, 407–412 (2019).
Chowdhury, R. et al. Environmental toxic metal contaminants and risk of cardiovascular disease: systematic review and meta-analysis. BMJ 362, k3310 (2018).
Meneguzzi, A., Fava, C., Castelli, M. & Minuz, P. Exposure to perfluoroalkyl chemicals and cardiovascular disease: experimental and epidemiological evidence. Front. Endocrinol. 12, 706352 (2021).
Zhao, L. et al. The associations between organophosphate pesticides (OPs) and respiratory disease, diabetes mellitus, and cardiovascular disease: a review and meta-analysis of observational studies. Toxics 11, 741 (2023).
Fawell, J. & Nieuwenhuijsen, M. J. Contaminants in drinking water. Br. Med. Bull. 68, 199–208 (2003).
Jarup, L. Hazards of heavy metal contamination. Br. Med. Bull. 68, 167–182 (2003).
Chowdhury, R. et al. Reducing NCDs globally: the under-recognised role of environmental risk factors. Lancet 392, 212 (2018).
National Research Council. Toxicological Effects of Methylmercury (National Academy Press, 2000). https://nap.nationalacademies.org/catalog/9899/toxicological-effects-of-methylmercury.
Lancereaux, E. Nephrite et arthrite saturnine: coincidences de ces affections; parallele avec la nephrite et l’arthrite gouttesses. Trans. Int. Med. Congr. 2, 193–202 (1881).
Schwartz, J. Lead, blood pressure, and cardiovascular disease in men. Arch. Environ. Health 50, 31–37 (1995).
Navas-Acien, A., Guallar, E., Silbergeld, E. K. & Rothenberg, S. J. Lead exposure and cardiovascular disease – a systematic review. Environ. Health Perspect. 115, 472–482 (2007).
Schober, S. E., Mirel, L. B., Graubard, B. I., Brody, D. J. & Flegal, K. M. Blood lead levels and death from all causes, cardiovascular disease, and cancer: results from the NHANES III mortality study. Environ. Health Perspect. 114, 1538–1541 (2006).
Lanphear, B. P., Rauch, S., Auinger, P., Allen, R. W. & Hornung, R. W. Low-level lead exposure and mortality in US adults: a population-based cohort study. Lancet Public Health 3, e177–e184 (2018).
Larsen, B. & Sanchez-Triana, E. Global health burden and cost of lead exposure in children and adults: a health impact and economic modelling analysis. Lancet Planet Health 7, e831–e840 (2023).
Zhu, K. et al. Associations of exposure to lead and cadmium with risk of all-cause and cardiovascular disease mortality among patients with type 2 diabetes. Environ. Sci. Pollut. Res. Int. 29, 76805–76815 (2022).
Xing, X. et al. Association of selenium and cadmium with heart failure and mortality based on the National Health and Nutrition Examination Survey. J. Hum. Nutr. Diet. 36, 1496–1506 (2023).
Tellez-Plaza, M., Jones, M. R., Dominguez-Lucas, A., Guallar, E. & Navas-Acien, A. Cadmium exposure and clinical cardiovascular disease: a systematic review. Curr. Atheroscler. Rep. 15, 356 (2013).
Duan, W. et al. Levels of a mixture of heavy metals in blood and urine and all-cause, cardiovascular disease and cancer mortality: a population-based cohort study. Environ. Pollut. 263, 114630 (2020).
Lim, K. M. et al. Low-level mercury can enhance procoagulant activity of erythrocytes: a new contributing factor for mercury-related thrombotic disease. Environ. Health Perspect. 118, 928–935 (2010).
Morin, Y. & Daniel, P. Quebec beer-drinkers’ cardiomyopathy: etiological considerations. Can. Med. Assoc. J. 97, 926–928 (1967).
Perez, A. L. & Tang, W. H. Contribution of environmental toxins in the pathogenesis of idiopathic cardiomyopathies. Curr. Treat. Options Cardiovasc. Med. 17, 381 (2015).
Stea, F., Bianchi, F., Cori, L. & Sicari, R. Cardiovascular effects of arsenic: clinical and epidemiological findings. Environ. Sci. Pollut. Res. Int. 21, 244–251 (2014).
Moon, K., Guallar, E. & Navas-Acien, A. Arsenic exposure and cardiovascular disease: an updated systematic review. Curr. Atheroscler. Rep. 14, 542–555 (2012).
Tseng, W. P. Black foot disease in Taiwan: a 30-year follow-up study. Angiology 40, 547–558 (1989).
Kinsman, G. D., Howard, A. N., Stone, D. L. & Mullins, P. A. Studies in copper status and atherosclerosis. Biochem. Soc. Trans. 18, 1186–1188 (1990).
United Nations Environment Programme. The Stockholm Convention on Persistent Organic Pollutants, http://chm.pops.int/TheConvention/Overview/TextoftheConvention/tabid/2232/Default.aspx (2019).
Agency for Toxic Substances and Disease Registry. Toxicological profile for chlorinated dibenzo-p-dioxins (CDDs) (ATSDR, 1998).
Consonni, D. et al. Mortality in a population exposed to dioxin after the Seveso, Italy, accident in 1976: 25 years of follow-up. Am. J. Epidemiol. 167, 847–858 (2008).
Lee, D. H. et al. A strong dose-response relation between serum concentrations of persistent organic pollutants and diabetes: results from the National Health and Examination Survey 1999-2002. Diabetes Care 29, 1638–1644 (2006).
Lim, J. S., Lee, D. H. & Jacobs, D. R. Jr Association of brominated flame retardants with diabetes and metabolic syndrome in the U.S. population, 2003-2004. Diabetes Care 31, 1802–1807 (2008).
Lang, I. A. et al. Association of urinary bisphenol A concentration with medical disorders and laboratory abnormalities in adults. JAMA 300, 1303–1310 (2008).
Moon, S. et al. Effects of bisphenol A on cardiovascular disease: an epidemiological study using National Health and Nutrition Examination Survey 2003-2016 and meta-analysis. Sci. Total Environ. 763, 142941 (2021).
Wang, R. et al. The bisphenol F and bisphenol S and cardiovascular disease: results from NHANES 2013–2016. Environ. Sci. Eur. 34, 4 (2022).
Chen, Z., He, J. & Shi, W. Association between urinary environmental phenols and the prevalence of cardiovascular diseases in US adults. Environ. Sci. Pollut. Res. Int. 29, 42947–42954 (2022).
Moreno-Gómez-Toledano, R. Relationship between emergent BPA-substitutes and renal and cardiovascular diseases in adult population. Environ. Pollut. 313, 120106 (2022).
Lu, Y., Chen, S., Jin, H., Tang, L. & Xia, M. Associations of bisphenol F and S, as substitutes for bisphenol A, with cardiovascular disease in American adults. J. Appl. Toxicol. 43, 500–507 (2023).
Bar-Meir, E. et al. Guidelines for treating cardiac manifestations of organophosphates poisoning with special emphasis on long QT and Torsades De Pointes. Crit. Rev. Toxicol. 37, 279–285 (2007).
Humblet, O., Birnbaum, L., Rimm, E., Mittleman, M. A. & Hauser, R. Dioxins and cardiovascular disease mortality. Environ. Health Perspect. 116, 1443–1448 (2008).
Gustavsson, P. & Hogstedt, C. A cohort study of Swedish capacitor manufacturing workers exposed to polychlorinated biphenyls (PCBs). Am. J. Ind. Med. 32, 234–239 (1997).
OECD. Global plastics outlook. OECDiLibrary http://www.oecd-ilibrary.org/environment/global-plastics-outlook_aa1edf33-en (2022).
European Environment Agency. Microplastics from textiles: towards a circular economy for textiles in Europe. EEA http://www.eea.europa.eu/publications/microplastics-from-textiles-towards-a (2022).
Wright, S. L., Thompson, R. C. & Galloway, T. S. The physical impacts of microplastics on marine organisms: a review. Environ. Pollut. 178, 483–492 (2013).
Yong, C. Q. Y., Valiyaveettil, S. & Tang, B. L. Toxicity of microplastics and nanoplastics in mammalian systems. Int. J. Environ. Res. Public Health 17, 1509 (2020).
Oh, N. & Park, J. H. Endocytosis and exocytosis of nanoparticles in mammalian cells. Int. J. Nanomed. 9, 51–63 (2014).
Zhang, S., Gao, H. & Bao, G. Physical principles of nanoparticle cellular endocytosis. ACS Nano 9, 8655–8671 (2015).
Gong, T., Liu, L., Jiang, W. & Zhou, R. DAMP-sensing receptors in sterile inflammation and inflammatory diseases. Nat. Rev. Immunol. 20, 95–112 (2020).
Jeong, J. & Choi, J. Adverse outcome pathways potentially related to hazard identification of microplastics based on toxicity mechanisms. Chemosphere 231, 249–255 (2019).
Horstmann, H., Ng, C. P., Tang, B. L. & Hong, W. Ultrastructural characterization of endoplasmic reticulum–Golgi transport containers (EGTC). J. Cell Sci. 115, 4263–4273 (2002).
Treyer, A., Pujato, M., Pechuan, X. & Musch, A. Iterative sorting of apical and basolateral cargo in Madin-Darby canine kidney cells. Mol. Biol. Cell 27, 2259–2271 (2016).
Lim, S. L. et al. Targeted metabolomics reveals differential biological effects of nanoplastics and nanoZnO in human lung cells. Nanotoxicology 13, 1117–1132 (2019).
Cordani, M. & Somoza, A. Targeting autophagy using metallic nanoparticles: a promising strategy for cancer treatment. Cell Mol. Life Sci. 76, 1215–1242 (2019).
Besseling, E., Wang, B., Lurling, M. & Koelmans, A. A. Nanoplastic affects growth of S. obliquus and reproduction of D. magna. Environ. Sci. Technol. 48, 12336–12343 (2014).
Liu, Z. et al. Age-dependent survival, stress defense, and AMPK in Daphnia pulex after short-term exposure to a polystyrene nanoplastic. Aquat. Toxicol. 204, 1–8 (2018).
Greven, A. C. et al. Polycarbonate and polystyrene nanoplastic particles act as stressors to the innate immune system of fathead minnow (Pimephales promelas). Environ. Toxicol. Chem. 35, 3093–3100 (2016).
Foley, C. J., Feiner, Z. S., Malinich, T. D. & Hook, T. O. A meta-analysis of the effects of exposure to microplastics on fish and aquatic invertebrates. Sci. Total Environ. 631-632, 550–559 (2018).
Bucci, K., Tulio, M. & Rochman, C. M. What is known and unknown about the effects of plastic pollution: a meta-analysis and systematic review. Ecol. Appl. 30, e02044 (2019).
De-la-Torre, G. E. Microplastics: an emerging threat to food security and human health. J. Food Sci. Technol. 57, 1601–1608 (2020).
Shiwakoti, S. et al. Effects of polystyrene nanoplastics on endothelium senescence and its underlying mechanism. Environ. Int. 164, 107248 (2022).
Zhao, J. et al. Polystyrene bead ingestion promotes adiposity and cardiometabolic disease in mice. Ecotoxicol. Environ. Saf. 232, 113239 (2022).
Wei, J. et al. The impact of polystyrene microplastics on cardiomyocytes pyroptosis through NLRP3/caspase-1 signaling pathway and oxidative stress in Wistar rats. Environ. Toxicol. 36, 935–944 (2021).
Li, Z. et al. Polystyrene microplastics cause cardiac fibrosis by activating Wnt/β-catenin signaling pathway and promoting cardiomyocyte apoptosis in rats. Environ. Pollut. 265, 115025 (2020).
Wang, X. et al. Nanoplastic-induced vascular endothelial injury and coagulation dysfunction in mice. Sci. Total Environ. 865, 161271 (2023).
Zhu, X. et al. Micro- and nanoplastics: a new cardiovascular risk factor? Environ. Int. 171, 107662 (2023).
Ali, N. et al. The potential impacts of micro-and-nano plastics on various organ systems in humans. EBioMedicine 99, 104901 (2023).
Marfella, R. et al. Microplastics and nanoplastics in atheromas and cardiovascular events. N. Engl. J. Med. 390, 900–910 (2024).
Beuchle, R., Achard, F., Bourgoin, C., Vancutsem, C. Deforestation and forest degradation in the Amazon – updated status and trends for the year 2021 (Publications Office of the European Union, 2022).
Veldkamp, E., Schmidt, M., Powers, J. S. & Corre, M. D. Deforestation and reforestation impacts on soils in the tropics. Nat. Rev. Earth Environ. 1, 590–605 (2020).
United Nations. The global forest goals report 2021. UN https://www.un.org/esa/forests/wp-content/uploads/2021/08/Global-Forest-Goals-Report-2021.pdf (2021).
Johnston, F. H. et al. Estimated global mortality attributable to smoke from landscape fires. Environ. Health Perspect. 120, 695–701 (2012).
Artiola, J. F., Walworth, J. L., Musil, S. A. & Crimmins, M. A. in Environmental and Pollution Science 3rd edn (eds Brusseau, M. L., Pepper, I. L. & Gerba, C. P.) 219–235 (Academic Press, 2019).
US Environmental Protection Agency. Air quality and climate change research. EPA http://www.epa.gov/air-research/air-quality-and-climate-change-research#:~:Text=Emissions%20of%20pollutants%20into%20the,cooling%20effects%20on%20the%20climate (2024).
UN Report: Global Assessment of Soil Pollution. Chapter 4. Environmental, Health and Socio-economic Impacts of Soil Pollution. 4.4 Socio-economic impacts of soil pollution. FAO http://www.fao.org/3/cb4894en/online/src/html/chapter-04-4.html (2017).
European Environment Agency. Soil moisture deficit, EEA http://www.eea.europa.eu/en/analysis/indicators/soil-moisture-deficit (2021).
US Environmental Protection Agency. Climate change indicators: permafrost. EPA https://www.epa.gov/climate-indicators/climate-change-indicators-permafrost#:~:Text=Additionally%2C%20organic%20matter%20 (2021).
United Nations. Water – at the center of the climate crisis. UN https://www.un.org/en/climatechange/science/climate-issues/water (2021).
Liu, J. et al. Emission control priority of PM2.5-bound heavy metals in different seasons: a comprehensive analysis from health risk perspective. Sci. Total Environ. 644, 20–30 (2018).
Prospero, J. M., Ginoux, P., Torres, O., Nicholson, S. E. & Gill, T. E. Environmental characterization of global sources of atmospheric soil dust identified with the Nimbus 7 Total Ozone Mapping Spectrometer (TOMS) absorbing aerosol product. Rev. Geophys. 40, 2-1–2-31 (2002).
Chen, W., Meng, H., Song, H. & Zheng, H. Progress in dust modelling, global dust budgets, and soil organic carbon dynamics. Land 11, 176 (2022).
Kok, J. F. S. et al. Mineral dust aerosol impacts on global climate and climate change. Nat. Rev. Earth Environ. 4, 71–86 (2023).
Giannadaki, D., Pozzer, A. & Lelieveld, J. Modeled global effects of airborne desert dust on air quality and premature mortality. Atmos. Chem. Phys. 14, 957–968 (2014).
Kotsyfakis, M., Zarogiannis, S. G. & Patelarou, E. The health impact of Saharan dust exposure. Int. J. Occup. Med. Environ. Health 32, 749–760 (2019).
Gomez, A. R. et al. The projected future degradation in air quality is caused by more abundant natural aerosols in a warmer world. Commun. Earth Environ. 4, 22 (2023).
Achakulwisut, P. et al. Effects of increasing aridity on ambient dust and public health in the U.S. Southwest under climate change. Geohealth 3, 127–144 (2019).
Fussell, J. C. & Kelly, F. J. Mechanisms underlying the health effects of desert sand dust. Environ. Int. 157, 106790 (2021).
Miller, M. R. Oxidative stress and the cardiovascular effects of air pollution. Free Radic. Biol. Med. 151, 69–87 (2020).
Lwin, K. S. et al. Effects of desert dust and sandstorms on human health: a scoping review. Geohealth 7, e2022GH000728 (2023).
Yu, Z. et al. Simulating the impact of long-range-transported Asian mineral dust on the formation of sulfate and nitrate during the KORUS-AQ Campaign. ACS Earth Space Chem. 4, 1039–1049 (2020).
Karydis, V. A., Tsimpidi, A. P., Pozzer, A., Astitha, M. & Lelieveld, J. Effects of mineral dust on global atmospheric nitrate concentrations. Atmos. Chem. Phys. 16, 1491–1509 (2016).
Ho, K. F. et al. Contributions of local pollution emissions to particle bioreactivity in downwind cities in China during Asian dust periods. Environ. Pollut. 245, 675–683 (2019).
Lovett, C., Sowlat, M. H., Saliba, N. A., Shihadeh, A. L. & Sioutas, C. Oxidative potential of ambient particulate matter in Beirut during Saharan and Arabian dust events. Atmos. Environ. 188, 34–42 (2018).
Chirizzi, D. et al. Influence of Saharan dust outbreaks and carbon content on oxidative potential of water-soluble fractions of PM2.5 and PM10. Atmos. Environ. 163, 1–8 (2017).
Yang, A., Tan, Q., Rajapakshe, C., Chin, M. & Yu, H. Global premature mortality by dust and pollution PM2.5 estimated from aerosol reanalysis of the modern-era retrospective analysis for research and applications, version 2. Sec. Atmos. Clim. 10, 975755 (2022).
Kojima, S. et al. Asian dust exposure triggers acute myocardial infarction. Eur. Heart J. 38, 3202–3208 (2017).
Galloway, J. N. et al. Transformation of the nitrogen cycle: recent trends, questions, and potential solutions. Science 320, 889–892 (2008).
Fowler, D. et al. The global nitrogen cycle in the twenty-first century. Philos. Trans. R. Soc. Lond. B Biol. Sci. 368, 20130164 (2013).
Kicińska, A., Radoslaw, P. & Izquierdo-Diaz, M. Changes in soil pH and mobility of heavy metals in contaminated soils. Eur. J. Soil Sci. 73, e13203 (2022).
Zhang, X., Ward, B. B. & Sigman, D. M. Global nitrogen cycle: critical enzymes, organisms, and processes for nitrogen budgets and dynamics. Chem. Rev. 120, 5308–5351 (2020).
McDuffie, E. E. et al. Source sector and fuel contributions to ambient PM2.5 and attributable mortality across multiple spatial scales. Nat. Commun. 12, 3594 (2021).
Lelieveld, J., Evans, J. S., Fnais, M., Giannadaki, D. & Pozzer, A. The contribution of outdoor air pollution sources to premature mortality on a global scale. Nature 525, 367–371 (2015).
Lelieveld, J. et al. Effects of fossil fuel and total anthropogenic emission removal on public health and climate. Proc. Natl Acad. Sci. USA 116, 7192–7197 (2019).
Gumanova, N. G., Deev, A. D., Zhang, W., Kots, A. Y. & Shalnova, S. A. Serum nitrite and nitrate levels, NOx, can predict cardiovascular mortality in the elderly in a 3-year follow-up study. Biofactors 43, 82–89 (2017).
Gumanova, N. G., Deev, A. D., Kots, A. Y. & Shalnova, S. A. Elevated levels of serum nitrite and nitrate, NOx, are associated with increased total and cardiovascular mortality in an 8-year follow-up study. Eur. J. Clin. Invest. 49, e13061 (2019).
Bahadoran, Z., Mirmiran, P., Tahmasebinejad, Z., Azizi, F. & Ghasemi, A. Serum nitric oxide metabolites and hard clinical endpoints: a population-based prospective study. Scand. Cardiovasc. J. 53, 176–182 (2019).
Wang, M. et al. Exposure to PM2.5 and its five constituents is associated with the incidence of type 2 diabetes mellitus: a prospective cohort study in northwest China. Environ. Geochem. Health 46, 34 (2024).
United Nations. World population prospects 2019: highlights. UN http://population.un.org/wpp/Publications/Files/wpp2019_10KeyFindings.pdf (2019).
Dye, C. Health and urban living. Science 319, 766–769 (2008).
Ezzati, M. et al. Cities for global health. BMJ 363, k3794 (2018).
Bikis, A. Urban air pollution and greenness in relation to public health. J. Environ. Public Health 2023, 8516622 (2023).
European Environment Agency. Air pollution still too high across Europe. EEA http://www.eea.europa.eu/highlights/air-pollution-still-too-high (2018).
Hunter, R. F. et al. Advancing urban green and blue space contributions to public health. Lancet Public Health 8, e735–e742 (2023).
Munzel, T. et al. Heart healthy cities: genetics loads the gun but the environment pulls the trigger. Eur. Heart J. 42, 2422–2438 (2021).
Baumgartner, J., Brauer, M. & Ezzati, M. The role of cities in reducing the cardiovascular impacts of environmental pollution in low- and middle-income countries. BMC Med. 18, 39 (2020).
Guthold, R., Stevens, G. A., Riley, L. M. & Bull, F. C. Worldwide trends in insufficient physical activity from 2001 to 2016: a pooled analysis of 358 population-based surveys with 1.9 million participants. Lancet Glob. Health 6, e1077–e1086 (2018).
Munzel, T., Sorensen, M. & Daiber, A. Transportation noise pollution and cardiovascular disease. Nat. Rev. Cardiol. 18, 619–636 (2021).
Nieuwenhuijsen, M. J. New urban models for more sustainable, liveable and healthier cities post Covid19; reducing air pollution, noise and heat island effects and increasing green space and physical activity. Environ. Int. 157, 106850 (2021).
Zota, A. R., Singla, V., Adamkiewicz, G., Mitro, S. D. & Dodson, R. E. Reducing chemical exposures at home: opportunities for action. J. Epidemiol. Community Health 71, 937–940 (2017).
Hahad, O. et al. Noise and air pollution as risk factors for hypertension: part II – pathophysiologic insight. Hypertension 80, 1384–1392 (2023).
European Commission. Zero Pollution Action Plan: towards zero pollution for air, water and soil. https://environment.ec.europa.eu/strategy/zero-pollution-action-plan_en# (2021).
European Environment Agency. Soil. EEA http://www.eea.europa.eu/en/topics/in-depth/soil# (2023).
European Environment Agency. Progress in the management of contaminated sites in Europe. EEA http://www.eea.europa.eu/en/analysis/indicators/progress-in-the-management-of (2022).
Goldsborough, E.III, Gopal, M., McEvoy, J. W., Blumenthal, R. S. & Jacobsen, A. P. Pollution and cardiovascular health: a contemporary review of morbidity and implications for planetary health. Am. Heart J. 25, 100231 (2023).
Tsoi, M. F., Lo, C. W. H., Cheung, T. T. & Cheung, B. M. Y. Blood lead level and risk of hypertension in the United States National Health and Nutrition Examination Survey 1999-2016. Sci. Rep. 11, 3010 (2021).
Cook, M. K., Zhang, J. & Wei, Y. Blood lead levels and risk of deaths from cardiovascular disease. Am. J. Cardiol. 173, 132–138 (2022).
Lee, M. S., Park, S. K., Hu, H. & Lee, S. Cadmium exposure and cardiovascular disease in the 2005 Korea National Health and Nutrition Examination Survey. Environ. Res. 111, 171–176 (2011).
Tellez-Plaza, M. et al. Cadmium exposure and incident cardiovascular disease. Epidemiology 24, 421–429 (2013).
Li, Z. et al. Association between exposure to cadmium and risk of all-cause and cause-specific mortality in the general US adults: a prospective cohort study. Chemosphere 307, 136060 (2022).
Tellez-Plaza, M. et al. Cadmium exposure and all-cause and cardiovascular mortality in the U.S. general population. Environ. Health Perspect. 120, 1017–1022 (2012).
Nigra, A. E., Moon, K. A., Jones, M. R., Sanchez, T. R. & Navas-Acien, A. Urinary arsenic and heart disease mortality in NHANES 2003-2014. Environ. Res. 200, 111387 (2021).
Sun, Y. et al. Association of seafood consumption and mercury exposure with cardiovascular and all-cause mortality among US adults. JAMA Netw. Open 4, e2136367 (2021).
Isiozor, N. M. et al. Serum copper and the risk of cardiovascular disease death in Finnish men. Nutr. Metab. Cardiovasc. Dis. 33, 151–157 (2023).
Shankar, A., Teppala, S. & Sabanayagam, C. Bisphenol A and peripheral arterial disease: results from the NHANES. Environ. Health Perspect. 120, 1297–1300 (2012).
Sturgeon, S. R., Flynn, D., Kaiser, A. B. & Reeves, K. W. Urinary levels of phthalate metabolites and cardiovascular disease mortality (NHANES, 1999-2008). Int. J. Hyg. Environ. Health 219, 876–882 (2016).
Bao, W. et al. Association between bisphenol A exposure and risk of all-cause and cause-specific mortality in US adults. JAMA Netw. Open 3, e2011620 (2020).
Cai, S. et al. Relationship between urinary bisphenol A levels and cardiovascular diseases in the U.S. adult population, 2003-2014. Ecotoxicol. Environ. Saf. 192, 110300 (2020).
Chen, M. et al. Relationship between bisphenol A and the cardiovascular disease metabolic risk factors in American adults: a population-based study. Chemosphere 324, 138289 (2023).
Chen, S. et al. Association of urinary bisphenol A with cardiovascular and all-cause mortality: National Health and Nutrition Examination Survey (NHANES) 2003-2016. Environ. Sci. Pollut. Res. Int. 30, 51217–51227 (2023).
Shankar, A., Xiao, J. & Ducatman, A. Perfluorooctanoic acid and cardiovascular disease in US adults. Arch. Intern. Med. 172, 1397–1403 (2012).
Huang, M. et al. Serum polyfluoroalkyl chemicals are associated with risk of cardiovascular diseases in national US population. Environ. Int. 119, 37–46 (2018).
Simpson, C., Winquist, A., Lally, C. & Steenland, K. Relation between perfluorooctanoic acid exposure and strokes in a large cohort living near a chemical plant. Environ. Res. 127, 22–28 (2013).
Xue, Q. et al. Association between pyrethroid exposure and cardiovascular disease: a national population-based cross-sectional study in the US. Environ. Int. 153, 106545 (2021).
Bao, W., Liu, B., Simonsen, D. W. & Lehmler, H. J. Association between exposure to pyrethroid insecticides and risk of all-cause and cause-specific mortality in the general US adult population. JAMA Intern. Med. 180, 367–374 (2020).
Chang, J. W. et al. Predicting the risk of cardiovascular disease in people exposed to moderate to high levels of dioxin. J. Hazard Mater. 198, 317–322 (2011).
Ha, M. H., Lee, D. H. & Jacobs, D. R. Association between serum concentrations of persistent organic pollutants and self-reported cardiovascular disease prevalence: results from the National Health and Nutrition Examination Survey, 1999-2002. Environ. Health Perspect. 115, 1204–1209 (2007).
Lin, Y. S. et al. Environmental exposure to dioxin-like compounds and the mortality risk in the U.S. population. Int. J. Hyg. Environ. Health 215, 541–546 (2012).
Global Alliance on Health and Pollution. Pollution and Health Metrics. GAHP https://gahp.net/wp-content/uploads/2019/12/PollutionandHealthMetrics-final-12_18_2019.pdf (2019).
Tang, F. H. M., Lenzen, M., McBratney, A. & Maggi, F. Risk of pesticide pollution at the global scale. Nat. Geosci. 14, 206–210 (2021).
Amini, M. et al. Statistical modeling of global geogenic arsenic contamination in groundwater. Environ. Sci. Technol. 42, 3669–3675 (2008).
Pure Earth. Lead country statistics. https://leadpollution.org/ (2019).
Steenhuisen, F. & Wilson, S. J. Development and application of an updated geospatial distribution model for gridding 2015 global mercury emissions. Atmos. Environ. 211, 138–150 (2019).
Lebreton, L. Where mismanaged plastic waste is generated and possible paths of change. The Ocean Cleanup https://theoceancleanup.com/updates/where-mismanaged-plastic-waste-is-generated-and-possible-paths-of-change/ (2019).
Global Water Intelligence. Agencies plead for global action on water pollution. https://www.globalwaterintel.com/news/2019/34/agencies-plead-for-global-action-on-water-pollution (2019).
FAO & UNEP. Global Assessment of Soil Pollution Ch. 4. Environmental, health and socio-economic impacts of soil pollution. 4.3 Soil pollution and risk to human health. https://openknowledge.fao.org/server/api/core/bitstreams/fe5df8d6-6b19-4def-bdc6-62886d824574/content/src/html/chapter-04-3.html (2021).
Campanale, C., Massarelli, C., Savino, I., Locaputo, V. & Uricchio, V. F. A detailed review study on potential effects of microplastics and additives of concern on human health. Int. J. Environ. Res. Public Health 17, 1212 (2020).
Carré, F. et al. in Global Soil Security (eds Field, D. J., Morgan, C.L.S. & McBratney, A. B.) 275–295 (Springer, 2017).
Filippini, T., Wise, L. A. & Vinceti, M. Cadmium exposure and risk of diabetes and prediabetes: a systematic review and dose-response meta-analysis. Environ. Int. 158, 106920 (2022).
Rahimi Kakavandi, N. et al. An updated systematic review and dose-response meta-analysis on the relation between exposure to arsenic and risk of type 2 diabetes. Toxicol. Lett. 384, 115–127 (2023).
Hu, X. F., Lowe, M. & Chan, H. M. Mercury exposure, cardiovascular disease, and mortality: a systematic review and dose-response meta-analysis. Environ. Res. 193, 110538 (2021).
Guo, Y., Lv, Y., Liu, X. & Wang, G. Association between heavy metal mercury in body fluids and tissues and diabetes mellitus: a systematic review and meta-analysis. Ann. Transl. Med. 11, 114 (2023).
Vena, J. et al. Exposure to dioxin and nonneoplastic mortality in the expanded IARC international cohort study of phenoxy herbicide and chlorophenol production workers and sprayers. Environ. Health Perspect. 106, 645–653 (1998).
Song, Y. et al. Endocrine-disrupting chemicals, risk of type 2 diabetes, and diabetes-related metabolic traits: a systematic review and meta-analysis. J. Diabetes 8, 516–532 (2016).
Mattsson, K. et al. Levels of perfluoroalkyl substances and risk of coronary heart disease: findings from a population-based longitudinal study. Environ. Res. 142, 148–154 (2015).
Schillemans, T. et al. Per- and polyfluoroalkyl substances and risk of myocardial infarction and stroke: a nested case-control study in Sweden. Environ. Health Perspect. 130, 37007 (2022).
Winquist, A. & Steenland, K. Modeled PFOA exposure and coronary artery disease, hypertension, and high cholesterol in community and worker cohorts. Environ. Health Perspect. 122, 1299–1305 (2014).
Gui, S. Y. et al. Association between per- and polyfluoroalkyl substances exposure and risk of diabetes: a systematic review and meta-analysis. J. Expo. Sci. Environ. Epidemiol. 33, 40–55 (2023).
Fu, X., Xu, J., Zhang, R. & Yu, J. The association between environmental endocrine disruptors and cardiovascular diseases: a systematic review and meta-analysis. Environ. Res. 187, 109464 (2020).
Zhang, H. et al. Phthalate exposure and risk of diabetes mellitus: implications from a systematic review and meta-analysis. Environ. Res. 204, 112109 (2022).
Zeng, G., Zhang, Q., Wang, X. & Wu, K. H. Low-level plasticizer exposure and all-cause and cardiovascular disease mortality in the general population. Environ. Health 21, 32 (2022).
Su, T. C., Hwang, J. J., Sun, C. W. & Wang, S. L. Urinary phthalate metabolites, coronary heart disease, and atherothrombotic markers. Ecotoxicol. Environ. Saf. 173, 37–44 (2019).
Melzer, D. et al. Urinary bisphenol A concentration and risk of future coronary artery disease in apparently healthy men and women. Circulation 125, 1482–1490 (2012).
Hwang, S., Lim, J. E., Choi, Y. & Jee, S. H. Bisphenol A exposure and type 2 diabetes mellitus risk: a meta-analysis. BMC Endocr. Disord. 18, 81 (2018).
Acknowledgements
T.M. is the principal investigator of the German Center for Cardiovascular Research (DZHK), Partner Site Rhine-Main, Mainz, Germany. O.H. and A.D. received funding from the DZHK. O.H. and A.D. have received vascular biology research grants from the Foundation Heart of Mainz. M.A. was supported by grants from the National Institute of Environmental Health Sciences (R01ES07331 and R01ES10563). All the authors were supported by the environmental network EXPOHEALTH, funded by the state of Rhineland-Palatinate, Germany.
Author information
Authors and Affiliations
Contributions
The authors contributed substantially to all aspects of the article.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Reviews Cardiology thanks Mark Miller, Annette Peters and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Münzel, T., Hahad, O., Lelieveld, J. et al. Soil and water pollution and cardiovascular disease. Nat Rev Cardiol (2024). https://doi.org/10.1038/s41569-024-01068-0
Accepted:
Published:
DOI: https://doi.org/10.1038/s41569-024-01068-0