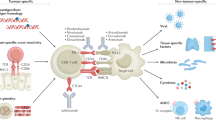
Abstract
Immune checkpoint molecules are physiological regulators of the adaptive immune response. Immune checkpoint inhibitors (ICIs), such as monoclonal antibodies targeting programmed cell death protein 1 or cytotoxic T lymphocyte-associated protein 4, have revolutionized cancer treatment and their clinical use is increasing. However, ICIs can cause various immune-related adverse events, including acute and chronic cardiotoxicity. Of these cardiovascular complications, ICI-induced acute fulminant myocarditis is the most studied, although emerging clinical and preclinical data are uncovering the importance of other ICI-related chronic cardiovascular complications, such as accelerated atherosclerosis and non-myocarditis-related heart failure. These complications could be more difficult to diagnose, given that they might only be present alongside other comorbidities. The occurrence of these complications suggests a potential role of immune checkpoint molecules in maintaining cardiovascular homeostasis, and disruption of physiological immune checkpoint signalling might thus lead to cardiac pathologies, including heart failure. Although inflammation is a long-known contributor to the development of heart failure, the therapeutic targeting of pro-inflammatory pathways has not been successful thus far. The increasingly recognized role of immune checkpoint molecules in the failing heart highlights their potential use as immunotherapeutic targets for heart failure. In this Review, we summarize the available data on ICI-induced cardiac dysfunction and heart failure, and discuss how immune checkpoint signalling is altered in the failing heart. Furthermore, we describe how pharmacological targeting of immune checkpoints could be used to treat heart failure.
Key points
-
Immune checkpoint inhibitors have revolutionized cancer treatment, but their use is associated with immune-related adverse events, including cardiovascular adverse effects.
-
Among the cardiovascular adverse events associated with immune checkpoint inhibitors, myocarditis is the most studied, but heart failure is increasingly being recognized.
-
Immune checkpoint signalling is involved in several physiological and pathological processes in the heart.
-
Modulation of immune checkpoint signalling can result in cardiac dysfunction or provide a target for therapeutic interventions in the failing heart, depending on the targeted checkpoint.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Johnson, D. B. et al. Fulminant myocarditis with combination immune checkpoint blockade. N. Engl. J. Med. 375, 1749–1755 (2016).
Yousif, L. I., Tanja, A. A., de Boer, R. A., Teske, A. J. & Meijers, W. C. The role of immune checkpoints in cardiovascular disease. Front. Pharmacol. 13, 989431 (2022).
Heinzerling, L. et al. Cardiotoxicity associated with CTLA4 and PD1 blocking immunotherapy. J. Immunother. Cancer 4, 50 (2016).
Drobni, Z. D. et al. Association between immune checkpoint inhibitors with cardiovascular events and atherosclerotic plaque. Circulation 142, 2299–2311 (2020).
Suero-Abreu, G. A., Zanni, M. V. & Neilan, T. G. Atherosclerosis with immune checkpoint inhibitor therapy: evidence, diagnosis, and management: JACC: cardiooncology state-of-the-art review. JACC Cardiooncol. 4, 598–615 (2022).
Vuong, J. T. et al. Immune checkpoint therapies and atherosclerosis: mechanisms and clinical implications: JACC state-of-the-art review. J. Am. Coll. Cardiol. 79, 577–593 (2022).
Gong, J. et al. Pericardial disease in patients treated with immune checkpoint inhibitors. J. Immunother. Cancer 9, e002771 (2021).
Daxini, A., Cronin, K. & Sreih, A. G. Vasculitis associated with immune checkpoint inhibitors — a systematic review. Clin. Rheumatol. 37, 2579–2584 (2018).
Power, J. R. et al. Electrocardiographic manifestations of immune checkpoint inhibitor myocarditis. Circulation 144, 1521–1523 (2021).
Zaha, V. G., Meijers, W. C. & Moslehi, J. Cardio-immuno-oncology. Circulation 141, 87–89 (2020).
Laenens, D. et al. Incidence of cardiovascular events in patients treated with immune checkpoint inhibitors. J. Clin. Oncol. 40, 3430–3438 (2022).
Dolladille, C. et al. Cardiovascular immunotoxicities associated with immune checkpoint inhibitors: a safety meta-analysis. Eur. Heart J. 42, 4964–4977 (2021).
Lutgens, E. et al. CHECKPOINT ATHERO: developing immune checkpoint-based therapeutics for atherosclerosis. Eur. Heart J. 44, 1010–1012 (2023).
Levine, B., Kalman, J., Mayer, L., Fillit, H. M. & Packer, M. Elevated circulating levels of tumor necrosis factor in severe chronic heart failure. N. Engl. J. Med. 323, 236–241 (1990).
Chung, E. S., Packer, M., Lo, K. H., Fasanmade, A. A. & Willerson, J. T. Randomized, double-blind, placebo-controlled, pilot trial of infliximab, a chimeric monoclonal antibody to tumor necrosis factor-α, in patients with moderate-to-severe heart failure: results of the anti-TNF therapy against congestive heart failure (ATTACH). Circulation 107, 3133–3140 (2003).
Everett, B. M. et al. Anti-inflammatory therapy with canakinumab for the prevention of hospitalization for heart failure. Circulation 139, 1289–1299 (2019).
Schiattarella, G. G. et al. Immunometabolic mechanisms of heart failure with preserved ejection fraction. Nat. Cardiovasc. Res. 1, 211–222 (2022).
Cohen, C. D. et al. Myocardial immune cells: the basis of cardiac immunology. J. Immunol. 210, 1198–1207 (2023).
Bajpai, G. et al. The human heart contains distinct macrophage subsets with divergent origins and functions. Nat. Med. 24, 1234–1245 (2018).
Liao, X. et al. Distinct roles of resident and nonresident macrophages in nonischemic cardiomyopathy. Proc. Natl Acad. Sci. USA 115, E4661–E4669 (2018).
Patel, B. et al. CCR2+ monocyte-derived infiltrating macrophages are required for adverse cardiac remodeling during pressure overload. JACC Basic Transl. Sci. 3, 230–244 (2018).
Onódi, Z. et al. AIM2-driven inflammasome activation in heart failure. Cardiovasc. Res. 117, 2639–2651 (2021).
Adamo, L., Rocha-Resende, C., Prabhu, S. D. & Mann, D. L. Reappraising the role of inflammation in heart failure. Nat. Rev. Cardiol. 17, 269–285 (2020).
Martini, E. et al. Single-cell sequencing of mouse heart immune infiltrate in pressure overload-driven heart failure reveals extent of immune activation. Circulation 140, 2089–2107 (2019).
Nevers, T. et al. Left ventricular T-cell recruitment contributes to the pathogenesis of heart failure. Circ. Heart Fail. 8, 776–787 (2015).
Bansal, S. S. et al. Activated T lymphocytes are essential drivers of pathological remodeling in ischemic heart failure. Circ. Heart Fail. 10, e003688 (2017).
Laroumanie, F. et al. CD4+ T cells promote the transition from hypertrophy to heart failure during chronic pressure overload. Circulation 129, 2111–2124 (2014).
Ngwenyama, N. et al. CXCR3 regulates CD4+ T cell cardiotropism in pressure overload-induced cardiac dysfunction. JCI Insight 4, e125527 (2019).
Bansal, S. S. et al. Dysfunctional and proinflammatory regulatory T-lymphocytes are essential for adverse cardiac remodeling in ischemic cardiomyopathy. Circulation 139, 206–221 (2019).
Oyewole-Said, D. et al. Beyond T-cells: functional characterization of CTLA-4 expression in immune and non-immune cell types. Front. Immunol. 11, 608024 (2020).
Bracamonte-Baran, W. et al. Endothelial stromal PD-L1 (programmed death ligand 1) modulates CD8+ T-cell infiltration after heart transplantation. Circ. Heart Fail. 14, e007982 (2021).
Cha, J. H., Chan, L. C., Li, C. W., Hsu, J. L. & Hung, M. C. Mechanisms controlling PD-L1 expression in cancer. Mol. Cell 76, 359–370 (2019).
Jhajj, H. S., Lwo, T. S., Yao, E. L. & Tessier, P. M. Unlocking the potential of agonist antibodies for treating cancer using antibody engineering. Trends Mol. Med. 29, 48–60 (2023).
Hodi, F. S. et al. Improved survival with ipilimumab in patients with metastatic melanoma. N. Engl. J. Med. 363, 711–723 (2010).
Robert, C. et al. Ipilimumab plus dacarbazine for previously untreated metastatic melanoma. N. Engl. J. Med. 364, 2517–2526 (2011).
Rizvi, N. A. et al. Activity and safety of nivolumab, an anti-PD-1 immune checkpoint inhibitor, for patients with advanced, refractory squamous non-small-cell lung cancer (CheckMate 063): a phase 2, single-arm trial. Lancet Oncol. 16, 257–265 (2015).
Balar, A. V. et al. First-line pembrolizumab in cisplatin-ineligible patients with locally advanced and unresectable or metastatic urothelial cancer (KEYNOTE-052): a multicentre, single-arm, phase 2 study. Lancet Oncol. 18, 1483–1492 (2017).
Larkin, J. et al. Combined nivolumab and ipilimumab or monotherapy in untreated melanoma. N. Engl. J. Med. 373, 23–34 (2015).
Wolchok, J. D. et al. Overall survival with combined nivolumab and ipilimumab in advanced melanoma. N. Engl. J. Med. 377, 1345–1356 (2017).
Upadhaya, S., Neftelinov, S. T., Hodge, J. & Campbell, J. Challenges and opportunities in the PD1/PDL1 inhibitor clinical trial landscape. Nat. Rev. Drug Discov. 21, 482–483 (2022).
Haslam, A., Gill, J. & Prasad, V. Estimation of the percentage of US patients with cancer who are eligible for immune checkpoint inhibitor drugs. JAMA Netw. Open 3, e200423 (2020).
Tawbi, H. A. et al. Relatlimab and nivolumab versus nivolumab in untreated advanced melanoma. N. Engl. J. Med. 386, 24–34 (2022).
Postow, M. A., Sidlow, R. & Hellmann, M. D. Immune-related adverse events associated with immune checkpoint blockade. N. Engl. J. Med. 378, 158–168 (2018).
Salem, J. E. et al. Cardiovascular toxicities associated with immune checkpoint inhibitors: an observational, retrospective, pharmacovigilance study. Lancet Oncol. 19, 1579–1589 (2018).
Mahmood, S. S. et al. Myocarditis in patients treated with immune checkpoint inhibitors. J. Am. Coll. Cardiol. 71, 1755–1764 (2018).
Moslehi, J., Lichtman, A. H., Sharpe, A. H., Galluzzi, L. & Kitsis, R. N. Immune checkpoint inhibitor-associated myocarditis: manifestations and mechanisms. J. Clin. Invest. 131, e145186 (2021).
Waliany, S. et al. Immune checkpoint inhibitor cardiotoxicity: understanding basic mechanisms and clinical characteristics and finding a cure. Annu. Rev. Pharmacol. Toxicol. 61, 113–134 (2021).
Palaskas, N., Lopez‐Mattei, J., Durand, J. B., Iliescu, C. & Deswal, A. Immune checkpoint inhibitor myocarditis: pathophysiological characteristics, diagnosis, and treatment. J. Am. Heart Assoc. 9, e013757 (2020).
Lyon, A. R. et al. 2022 ESC guidelines on cardio-oncology developed in collaboration with the European Hematology Association (EHA), the European Society for Therapeutic Radiology and Oncology (ESTRO) and the International Cardio-Oncology Society (IC-OS). Eur. Heart J. 43, 4229–4361 (2022).
Thavendiranathan, P. et al. Myocardial T1 and T2 mapping by magnetic resonance in patients with immune checkpoint inhibitor-associated myocarditis. J. Am. Coll. Cardiol. 77, 1503–1516 (2021).
Cadour, F. et al. Cardiac MRI features and prognostic value in immune checkpoint inhibitor-induced myocarditis. Radiology 303, 512–521 (2022).
Roth, M. E., Muluneh, B., Jensen, B. C., Madamanchi, C. & Lee, C. B. Left ventricular dysfunction after treatment with ipilimumab for metastatic melanoma. Am. J. Ther. 23, e1925–e1928 (2016).
Serzan, M., Rapisuwon, S., Krishnan, J., Chang, I. C. & Barac, A. Takotsubo cardiomyopathy associated with checkpoint inhibitor therapy: endomyocardial biopsy provides pathological insights to dual diseases. JACC Cardiooncol. 3, 330–334 (2021).
Waliany, S. et al. Myocarditis surveillance with high-sensitivity troponin I during cancer treatment with immune checkpoint inhibitors. JACC Cardiooncol. 3, 137–139 (2021).
Sakai, T. et al. Nivolumab-induced myocardial necrosis in a patient with lung cancer: a case report. Respir. Med. Case Rep. 27, 100839 (2019).
Samejima, Y. et al. Development of severe heart failure in a patient with squamous non-small-cell lung cancer during nivolumab treatment. Intern. Med. 59, 2003–2008 (2020).
Escudier, M. et al. Clinical features, management, and outcomes of immune checkpoint inhibitor-related cardiotoxicity. Circulation 136, 2085–2087 (2017).
Michel, L. et al. Targeting early stages of cardiotoxicity from anti-PD1 immune checkpoint inhibitor therapy. Eur. Heart J. 43, 316–329 (2022).
Waheed, N. et al. Newly diagnosed cardiovascular disease in patients treated with immune checkpoint inhibitors: a retrospective analysis of patients at an academic tertiary care center. Cardiooncology 7, 10 (2021).
D’Souza, M. et al. The risk of cardiac events in patients receiving immune checkpoint inhibitors: a nationwide Danish study. Eur. Heart J. 42, 1621–1631 (2020).
Dolladille, C. et al. Late cardiac adverse events in patients with cancer treated with immune checkpoint inhibitors. J. Immunother. Cancer 8, e000261 (2020).
Chitturi, K. R. et al. Immune checkpoint inhibitor-related adverse cardiovascular events in patients with lung cancer. JACC Cardiooncol. 1, 182–192 (2019).
Lal, J. C., Brown, S. A., Collier, P. & Cheng, F. A retrospective analysis of cardiovascular adverse events associated with immune checkpoint inhibitors. Cardiooncology 7, 19 (2021).
Naqash, A. R. et al. Major adverse cardiac events with immune checkpoint inhibitors: a pooled analysis of trials sponsored by the National Cancer Institute — cancer therapy evaluation program. J. Clin. Oncol. 40, 3439–3452 (2022).
Korste, S. et al. Anthracycline therapy modifies immune checkpoint signaling in the heart. Int. J. Mol. Sci. 24, 6052 (2023).
Sharma, M., Suero-Abreu, G. A. & Kim, B. A case of acute heart failure due to immune checkpoint blocker nivolumab. Cardiol. Res. 10, 120–123 (2019).
Chahine, J., Collier, P., Maroo, A., Tang, W. H. W. & Klein, A. L. Myocardial and pericardial toxicity associated with immune checkpoint inhibitors in cancer patients. JACC Case Rep. 2, 191–199 (2020).
Okauchi, S., Sasatani, Y., Yamada, H. & Satoh, H. Late-onset pulmonary and cardiac toxicities in a patient treated with immune checkpoint inhibitor monotherapy. Klinicka Onkol. 35, 150–154 (2022).
Rini, B. I. et al. Prospective cardiovascular surveillance of immune checkpoint inhibitor-based combination therapy in patients with advanced renal cell cancer: data from the phase III JAVELIN Renal 101 trial. J. Clin. Oncol. 40, 1929–1938 (2022).
Andres, M. S. et al. The spectrum of cardiovascular complications related to immune-checkpoint inhibitor treatment: including myocarditis and the new entity of non inflammatory left ventricular dysfunction. Cardiooncology 8, 21 (2022).
Geisler, B. P., Raad, R. A., Esaian, D., Sharon, E. & Schwartz, D. R. Apical ballooning and cardiomyopathy in a melanoma patient treated with ipilimumab: a case of takotsubo-like syndrome. J. Immunother. Cancer 3, 4 (2015).
Anderson, R. D. & Brooks, M. Apical takotsubo syndrome in a patient with metastatic breast carcinoma on novel immunotherapy. Int. J. Cardiol. 222, 760–761 (2016).
Ederhy, S. et al. Takotsubo-like syndrome in cancer patients treated with immune checkpoint inhibitors. JACC Cardiovasc. Imaging 11, 1187–1190 (2018).
Schwab, K. S. et al. Long term remission and cardiac toxicity of a combination of ipilimumab and nivolumab in a patient with metastatic head and neck carcinoma after progression following nivolumab monotherapy. Front. Oncol. 9, 403 (2019).
Okamatsu, Y. et al. Rapid onset of takotsubo cardiomyopathy induced by an infusion reaction to pembrolizumab in a patient with NSCLC. JTO Clin. Res. Rep. 1, 100055 (2020).
Khan, N. A. J., Pacioles, T. & Alsharedi, M. Atypical takotsubo cardiomyopathy secondary to combination of chemo-immunotherapy in a patient with non-small cell lung cancer. Cureus 12, 10–13 (2020).
Oldfield, K., Jayasinghe, R., Niranjan, S. & Chadha, S. Immune checkpoint inhibitor-induced takotsubo syndrome and diabetic ketoacidosis: rare reactions. BMJ Case Rep. 14, e237217 (2021).
Ederhy, S., Dolladille, C., Thuny, F., Alexandre, J. & Cohen, A. Takotsubo syndrome in patients with cancer treated with immune checkpoint inhibitors: a new adverse cardiac complication. Eur. J. Heart Fail. 21, 945–947 (2019).
Norikane, T. et al. Immune checkpoint inhibitor myocarditis mimicking takotsubo cardiomyopathy on MPI. J. Nucl. Cardiol. 29, 2694–2698 (2022).
Tan, N. Y. L., Anavekar, N. S. & Wiley, B. M. Concomitant myopericarditis and takotsubo syndrome following immune checkpoint inhibitor therapy. BMJ Case Rep. 13, e235265 (2020).
Norwood, T. G. et al. Smoldering myocarditis following immune checkpoint blockade. J. Immunother. Cancer 5, 91 (2017).
Johnson, D. B., Nebhan, C. A., Moslehi, J. J. & Balko, J. M. Immune-checkpoint inhibitors: long-term implications of toxicity. Nat. Rev. Clin. Oncol. 19, 254–267 (2022).
Drobni, Z. D. et al. Impact of immune checkpoint inhibitors on atherosclerosis progression in patients with lung cancer. J. Immunother. Cancer 11, e007307 (2023).
Poels, K. et al. Immune checkpoint inhibitor therapy aggravates T cell-driven plaque inflammation in atherosclerosis. JACC Cardiooncol. 2, 599–610 (2020).
Ferdinandy, P. et al. Definition of hidden drug cardiotoxicity: paradigm change in cardiac safety testing and its clinical implications. Eur. Heart J. 40, 1771–1777 (2018).
Lau, E. S. et al. Cardiovascular risk factors are associated with future cancer. JACC Cardiooncol. 3, 48–58 (2021).
Torrente, M. et al. Assessing the risk of cardiovascular events in patients receiving immune checkpoint inhibitors. Front. Cardiovasc. Med. 9, 1062858 (2022).
Lehmann, L. H. et al. Cardiomuscular biomarkers in the diagnosis and prognostication of immune checkpoint inhibitor myocarditis. Circulation 148, 473–486 (2023).
Nishimura, H. et al. Autoimmune dilated cardiomyopathy in PD-1 receptor-deficient mice. Science 291, 319–322 (2001).
Okazaki, T. et al. Autoantibodies against cardiac troponin I are responsible for dilated cardiomyopathy in PD-1-deficient mice. Nat. Med. 9, 1477–1483 (2003).
Forte, E. et al. Type 2 MI induced by a single high dose of isoproterenol in C57BL/6J mice triggers a persistent adaptive immune response against the heart J. Cell. Mol. Med. 25, 229–243 (2020).
Furusawa, S. et al. Cardiac autoantibodies against cardiac troponin I in post-myocardial infarction heart failure: evaluation in a novel murine model and applications in therapeutics. Circ. Heart Fail. 16, e010347 (2023).
Nishimura, H., Nose, M., Hiai, H., Minato, N. & Honjo, T. Development of lupus-like autoimmune diseases by disruption of the PD-1 gene encoding an ITIM motif-carrying immunoreceptor. Immunity 11, 141–151 (1999).
Gergely, T. G. et al. Characterization of immune checkpoint inhibitor‐induced cardiotoxicity reveals interleukin‐17A as a driver of cardiac dysfunction after anti‐PD‐1 treatment. Br. J. Pharmacol. 180, 740–761 (2022).
Baldeviano, G. C. et al. Interleukin-17A is dispensable for myocarditis but essential for the progression to dilated cardiomyopathy. Circ. Res. 106, 1646–1655 (2010).
Quagliariello, V. et al. Evidences of CTLA-4 and PD-1 blocking agents-induced cardiotoxicity in cellular and preclinical models. J. Pers. Med. 10, 179 (2020).
Tay, W. T. et al. Programmed cell death-1: programmed cell death-ligand 1 interaction protects human cardiomyocytes against T-cell mediated inflammation and apoptosis response in vitro. Int. J. Mol. Sci. 21, 2399 (2020).
Xia, W., Zou, C., Chen, H., Xie, C. & Hou, M. Immune checkpoint inhibitor induces cardiac injury through polarizing macrophages via modulating microRNA-34a/Kruppel-like factor 4 signaling. Cell Death Dis. 11, 575 (2020).
Xia, W. et al. PD-1 inhibitor inducing exosomal miR-34a-5p expression mediates the cross talk between cardiomyocyte and macrophage in immune checkpoint inhibitor-related cardiac dysfunction. J. Immunother. Cancer 8, e001293 (2020).
Zhu, H. et al. Identification of pathogenic immune cell subsets associated with checkpoint inhibitor-induced myocarditis. Circulation 146, 316–335 (2022).
Wei, S. C. et al. A genetic mouse model recapitulates immune checkpoint inhibitor-associated myocarditis and supports a mechanism-based therapeutic intervention. Cancer Discov. 11, 614–625 (2021).
Axelrod, M. L. et al. T cells specific for α-myosin drive immunotherapy-related myocarditis. Nature 611, 818–826 (2022).
Won, T. et al. Cardiac myosin-specific autoimmune T cells contribute to immune-checkpoint-inhibitor-associated myocarditis. Cell Rep. 41, 111611 (2022).
Ma, P. et al. Expansion of pathogenic cardiac macrophages in immune checkpoint inhibitor myocarditis. Circulation 149, 48–66 (2023).
Lee, C. et al. Pre-existing autoimmune disease increases the risk of cardiovascular and noncardiovascular events after immunotherapy. JACC Cardiooncol. 4, 660–669 (2022).
Tsuruoka, K. et al. Exacerbation of autoimmune myocarditis by an immune checkpoint inhibitor is dependent on its time of administration in mice. Int. J. Cardiol. 313, 67–75 (2020).
Zhang, Y. et al. Hormonal therapies up-regulate MANF and overcome female susceptibility to immune checkpoint inhibitor myocarditis. Sci. Transl. Med. 14, eabo1981 (2022).
Nemeth, B. T., Varga, Z. V., Wu, W. J. & Pacher, P. Trastuzumab cardiotoxicity: from clinical trials to experimental studies. Br. J. Pharmacol. 174, 3727–3748 (2017).
Vermeulen, Z., Segers, V. F. M. & De Keulenaer, G. W. ErbB2 signaling at the crossing between heart failure and cancer. Basic Res. Cardiol. 111, 60 (2016).
Dokmanovic, M., King, K. E., Mohan, N., Endo, Y. & Wu, W. J. Cardiotoxicity of ErbB2-targeted therapies and its impact on drug development, a spotlight on trastuzumab. Expert Opin. Drug Metab. Toxicol. 13, 755–766 (2017).
Xiao, L. et al. Ibrutinib-mediated atrial fibrillation attributable to inhibition of C-terminal Src kinase. Circulation 142, 2443–2455 (2020).
Simons, K. H. et al. T cell co-stimulation and co-inhibition in cardiovascular disease: a double-edged sword. Nat. Rev. Cardiol. 16, 325–343 (2019).
Shami, A. et al. Glucocorticoid-induced tumour necrosis factor receptor family-related protein (GITR) drives atherosclerosis in mice and is associated with an unstable plaque phenotype and cerebrovascular events in humans. Eur. Heart J. 41, 2938–2948 (2020).
Kassiteridi, C. et al. CD200 limits monopoiesis and monocyte recruitment in atherosclerosis. Circ. Res. 129, 280–295 (2021).
Lacy, M. et al. Cell-specific and divergent roles of the CD40L–CD40 axis in atherosclerotic vascular disease. Nat. Commun. 12, 3754 (2021).
Lutgens, E. et al. Immunotherapy for cardiovascular disease. Eur. Heart J. 40, 3937–3946 (2019).
Delgobo, M. et al. Myocardial milieu favors local differentiation of regulatory T cells. Circ. Res. 132, 565–582 (2023).
Kushnareva, E., Kushnarev, V., Artemyeva, A., Mitrofanova, L. & Moiseeva, O. Myocardial PD-L1 expression in patients with ischemic and non-ischemic heart failure. Front. Cardiovasc. Med. 8, 759972 (2022).
Screever, E. M. et al. Circulating immune checkpoints predict heart failure outcomes. ESC Heart Fail. 10, 2330–2337 (2023).
Hayashi, T. et al. The programmed death-1 signaling axis modulates inflammation and LV structure/function in a stress-induced cardiomyopathy model. JACC Basic Transl. Sci. 7, 1120–1139 (2022).
Choudhary, A. et al. PD-L1 (programmed death ligand 1) as a marker of acute cellular rejection after heart transplantation. Circ. Heart Fail. 14, e008563 (2021).
Horckmans, M., Diaz Villamil, E., Horckmans, M., De Roeck, L. & Communi, D. Central role of PD-L1 in cardioprotection resulting from P2Y4 nucleotide receptor loss. Front. Immunol. 13, 1006934 (2022).
Ruppert, V. et al. Evidence for CTLA4 as a susceptibility gene for dilated cardiomyopathy. Eur. J. Hum. Genet. 18, 694–699 (2010).
Wang, H. et al. CD28/B7 deficiency attenuates systolic overload-induced congestive heart failure, myocardial and pulmonary inflammation, and activated T cell accumulation in the heart and lungs. Hypertension 68, 688–696 (2016).
Kallikourdis, M. et al. T cell costimulation blockade blunts pressure overload-induced heart failure. Nat. Commun. 8, 14680 (2017).
Martini, E. et al. T cell costimulation blockade blunts age-related heart failure. Circ. Res. 127, 1115–1117 (2020).
Reynolds, K. et al. 696 Abatacept for immune checkpoint inhibitor associated myocarditis (ATRIUM): a phase 3, investigator-initiated, randomized, double blind, placebo-controlled trial. J. Immunother. Cancer https://doi.org/10.1136/jitc-2022-SITC2022.0696 (2022).
Wolf, Y., Anderson, A. C. & Kuchroo, V. K. TIM3 comes of age as an inhibitory receptor. Nat. Rev. Immunol. 20, 173–185 (2020).
Acharya, N., Acharya, N., Sabatos-Peyton, C., Anderson, A. C. & Anderson, A. C. Tim-3 finds its place in the cancer immunotherapy landscape. J. Immunother. Cancer 8, e000911 (2020).
Meng, X., Xia, G., Zhang, L., Xu, C. & Chen, Z. T cell immunoglobulin and mucin domain-containing protein 3 is highly expressed in patients with acute decompensated heart failure and predicts mid-term prognosis. Front. Cardiovascular Med. 9, 933532 (2022).
Garetto, S. et al. Immunobiology peak inflammation in atherosclerosis, primary biliary cirrhosis and autoimmune arthritis is counter-intuitively associated with regulatory T cell enrichment. Immunobiology 220, 1025–1029 (2015).
Girerd, N. et al. Protein biomarkers of new-onset heart failure: insights from the heart omics and ageing cohort, the atherosclerosis risk in communities study, and the Framingham Heart Study. Circ. Heart Fail. 16, e009694 (2023).
Cauwenberghs, N., Sabovčik, F., Magnus, A., Haddad, F. & Kuznetsova, T. Proteomic profiling for detection of early-stage heart failure in the community. ESC Heart Fail. 8, 2928–2939 (2021).
Kresoja, K.-P. et al. Proteomics to improve phenotyping in obese patients with heart failure with preserved ejection fraction. Eur. J. Heart Fail. 23, 1633–1644 (2021).
Wang, Y. et al. Loss of CEACAM1, a tumor-associated factor, attenuates post-infarction cardiac remodeling by inhibiting apoptosis. Sci. Rep. 6, 1972 (2016).
Han, W. K., Bailly, V., Abichandani, R., Thadhani, R. & Bonventre, J. V. Kidney injury molecule-1 (KIM-1): a novel biomarker for human renal proximal tubule injury. Kidney Int. 62, 237–244 (2002).
Driver, T. H. et al. Urinary kidney injury molecule 1 (KIM-1) and interleukin 18 (IL-18) as risk markers for heart failure in older adults: the health, aging, and body composition (Health ABC) study. Am. J. Kidney Dis. 64, 49–56 (2014).
Jungbauer, C. G. et al. Kidney injury molecule-1 and N-acetyl-β-d-glucosaminidase in chronic heart failure: possible biomarkers of cardiorenal syndrome. Eur. J. Heart Fail. 13, 1104–1110 (2011).
Stenemo, M. et al. Circulating proteins as predictors of incident heart failure in the elderly. Eur. J. Heart Fail. 20, 55–62 (2018).
Emmens, J. E. et al. Plasma kidney injury molecule-1 in heart failure: renal mechanisms and clinical outcome. Eur. J. Heart Fail. 18, 641–649 (2016).
Henry, A. et al. Therapeutic targets for heart failure identified using proteomics and Mendelian randomization. Circulation 145, 1205–1217 (2022).
Xie, X., Li, C., Zhou, B., Dai, X. & Rao, L. Associations between TIM1 polymorphisms and dilated cardiomyopathy in a Han Chinese population. Int. Heart J. 57, 742–746 (2016).
Dong, C. et al. ICOS co-stimulatory receptor is essential for T-cell activation and function. Nature 409, 97–101 (2001).
Yoshinaga, S. K. et al. T-cell co-stimulation through B7RP-1 and ICOS. Nature 402, 827–832 (1999).
Khayyamian, S. et al. ICOS-ligand, expressed on human endothelial cells, costimulates Th1 and Th2 cytokine secretion by memory CD4+ T cells. Proc. Natl Acad. Sci. USA 99, 6198–6203 (2002).
Solinas, C., Gu-Trantien, C. & Willard-Gallo, K. The rationale behind targeting the ICOS–ICOS ligand costimulatory pathway in cancer immunotherapy. ESMO Open 5, e000544 (2020).
Spicer, P. & Runkel, L. Costimulatory pathway targets for autoimmune and inflammatory conditions: clinical successes, failures, and hope for the future. Expert Opin. Investig. Drugs 28, 99–106 (2019).
Voors, A. A. et al. A systems biology study to tailored treatment in chronic heart failure: rationale, design, and baseline characteristics of BIOSTAT-CHF. Eur. J. Heart Fail. 18, 716–726 (2016).
Markousis-Mavrogenis, G. et al. Multimarker profiling identifies protective and harmful immune processes in heart failure: findings from BIOSTAT-CHF. Cardiovasc. Res. 118, 1964–1977 (2022).
Futamatsu, H. et al. Attenuation of experimental autoimmune myocarditis by blocking activated T cells through inducible costimulatory molecule pathway. Cardiovasc. Res. 59, 95–104 (2003).
Liu, W. et al. Adenovirus-mediated ICOSIg gene transfer alleviates cardiac remodeling in experimental autoimmune myocarditis. Immunol. Cell Biol. 86, 659–665 (2008).
Li, D. K. & Wang, W. Characteristics and clinical trial results of agonistic anti-CD40 antibodies in the treatment of malignancies (review). Oncol. Lett. 20, 176 (2020).
Yndestad, A. et al. Increased gene expression of tumor necrosis factor superfamily ligands in peripheral blood mononuclear cells during chronic heart failure. Cardiovasc. Res. 54, 175–182 (2002).
Stumpf, C. et al. Enhanced levels of CD154 (CD40 ligand) on platelets in patients with chronic heart failure. Eur. J. Heart Fail. 5, 629–637 (2003).
Ueland, T. et al. Soluble CD40 ligand in acute and chronic heart failure. Eur. Heart J. 26, 1101–1107 (2005).
Seijkens, T. T. P. et al. Targeting CD40-induced TRAF6 signaling in macrophages reduces atherosclerosis. J. Am. Coll. Cardiol. 71, 527–542 (2018).
Lameijer, M. et al. Efficacy and safety assessment of a TRAF6-targeted nanoimmunotherapy in atherosclerotic mice and non-human primates. Nat. Biomed. Eng. 2, 279–292 (2018).
Bosch, L. et al. Small molecule-mediated inhibition of CD40–TRAF6 reduces adverse cardiac remodelling in pressure overload induced heart failure. Int. J. Cardiol. 279, 141–144 (2019).
Duhen, R. et al. Neoadjuvant anti-OX40 (MEDI6469) therapy in patients with head and neck squamous cell carcinoma activates and expands antigen-specific tumor-infiltrating T cells. Nat. Commun. 12, 1047 (2021).
Wu, Q. Q. et al. OX40 regulates pressure overload-induced cardiac hypertrophy and remodelling via CD4+ T-cells. Clin. Sci. 130, 2061–2071 (2016).
Seko, Y. et al. Expression of tumor necrosis factor ligand superfamily costimulatory molecules CD27L, CD30L, OX40L and 4-1BBL in the heart of patients with acute myocarditis and dilated cardiomyopathy. Cardiovasc. Pathol. 11, 166–170 (2002).
Min, A., Kim, J., Nemeth, M. R. & Lim, S. 4-1BB: a promising target for cancer immunotherapy. Front. Oncol. 12, 968360 (2022).
Cheung, C. T. Y. et al. Neutralizing anti-4-1BBL treatment improves cardiac function in viral myocarditis. Lab. Invest. 87, 651–661 (2007).
Mouton, A. J. et al. Fibroblast polarization over the myocardial infarction time continuum shifts roles from inflammation to angiogenesis. Basic Res. Cardiol. 114, 6 (2019).
Xu, Y. et al. CD137 signal mediates cardiac ischemia — reperfusion injury by regulating the necrosis of cardiomyocytes. J. Cardiovasc. Transl. Res. 15, 1163–1175 (2022).
Tang, H. et al. Facilitating T cell infiltration in tumor microenvironment overcomes resistance to PD-L1 blockade. Cancer Cell 29, 285–296 (2016).
Dahl, C. P. et al. Increased expression of LIGHT/TNFSF14 and its receptors in experimental and clinical heart failure. Eur. J. Heart Fail. 10, 352–359 (2008).
Wu, Y. et al. TNFSF14/LIGHT promotes cardiac fibrosis and atrial fibrillation vulnerability via PI3Kγ/SGK1 pathway-dependent M2 macrophage polarisation. J. Transl. Med. 21, 544 (2023).
Rumpret, M. et al. Functional categories of immune inhibitory receptors. Nat. Rev. Immunol. 20, 771–780 (2020).
Ashour, D., Delgobo, M., Frantz, S. & Ramos, G. C. Coping with sterile inflammation: between risk and necessity. Cardiovasc. Res. 117, e84–e87 (2021).
Vinh, A. et al. Inhibition and genetic ablation of the B7/CD28 T-cell costimulation axis prevents experimental hypertension. Circulation 122, 2529–2537 (2010).
Salem, J.-E. et al. Abatacept for severe immune checkpoint inhibitor-associated myocarditis. N. Engl. J. Med. 380, 2377–2379 (2019).
Nguyen, L. S. et al. Reversal of immune-checkpoint inhibitor fulminant myocarditis using personalized-dose-adjusted abatacept and ruxolitinib: proof of concept. J. Immunother. Cancer 10, e004699 (2022).
Salem, J.-E. et al. Abatacept/ruxolitinib and screening for concomitant respiratory muscle failure to mitigate fatality of immune-checkpoint inhibitor myocarditis. Cancer Discov. 13, 1100–1115 (2023).
Liu, M. et al. Soluble CTLA-4 mutants ameliorate immune-related adverse events but preserve efficacy of CTLA-4- and PD-1-targeted immunotherapy. Sci. Transl. Med. 15, eabm5663 (2023).
Leedy, D. J. et al. The association between heart failure and incident cancer in women: an analysis of the Women’s Health Initiative. Eur. J. Heart Fail. 23, 1712–1721 (2021).
Peleg Hasson, S. et al. Cancer therapeutics-related cardiac dysfunction in patients treated with immune checkpoint inhibitors: an understudied manifestation. J. Immunother. 44, 179–184 (2021).
Isawa, T., Toi, Y., Sugawara, S., Taguri, M. & Toyoda, S. Incidence, clinical characteristics, and predictors of cardiovascular immune-related adverse events associated with immune checkpoint inhibitors. Oncologist 27, e410–e419 (2022).
Zhou, J. et al. Adverse cardiovascular complications following prescription of programmed cell death 1 (PD-1) and programmed cell death ligand 1 (PD-L1) inhibitors: a propensity-score matched cohort study with competing risk analysis. Cardiooncology 8, 5 (2022).
Xu, A. et al. Early detection of immune checkpoint inhibitor-related subclinical cardiotoxicity: a pilot study by using speckle tracking imaging and three-dimensional echocardiography. Front. Cardiovasc. Med. 9, 1087287 (2022).
Brumberger, Z. L. et al. Cardiotoxicity risk factors with immune checkpoint inhibitors. Cardiooncology 8, 3 (2022).
Li, C., Bhatti, S. A. & Ying, J. Immune checkpoint inhibitors-associated cardiotoxicity. Cancers 14, 1145 (2022).
Chan, J. S. K. et al. Cardiovascular outcomes and hospitalizations in Asian patients receiving immune checkpoint inhibitors: a population-based study. Curr. Probl. Cardiol. 48, 101380 (2023).
Zhang, C. et al. Incidence of adverse cardiovascular events associated with immune checkpoint inhibitors and risk factors for left ventricular dysfunction: a single-center prospective clinical study. Front. Cardiovasc. Med. 10, 1052699 (2023).
Cutroneo, P. M. et al. Safety profile of immune checkpoint inhibitors: an analysis of the Italian spontaneous reporting system database. Br. J. Clin. Pharmacol. 87, 527–541 (2021).
Mascolo, A. et al. Immune checkpoint inhibitors and cardiotoxicity: an analysis of spontaneous reports in eudravigilance. Drug Saf. 44, 957–971 (2021).
Jain, P. et al. Cardiovascular adverse events are associated with usage of immune checkpoint inhibitors in real-world clinical data across the United States. ESMO Open 6, 100252 (2021).
Rubio-Infante, N. et al. Cardiotoxicity associated with immune checkpoint inhibitor therapy: a meta-analysis. Eur. J. Heart Fail. 23, 1739–1747 (2021).
Heilbroner, S. P. et al. Predicting cardiac adverse events in patients receiving immune checkpoint inhibitors: a machine learning approach. J. Immunother. Cancer 9, e002545 (2021).
Wu, N. C. et al. Clinical features and outcomes of immune checkpoint inhibitor-associated cardiovascular toxicities. Acta Cardiol. Sin. 38, 39–46 (2022).
Malaty, M. M., Amarasekera, A. T., Li, C., Scherrer-Crosbie, M. & Tan, T. C. Incidence of immune checkpoint inhibitor mediated cardiovascular toxicity: a systematic review and meta-analysis. Eur. J. Clin. Invest. 52, e13831 (2022).
Acknowledgements
Z.V.V. was supported by the EU Horizon 2020 Research and Innovation Programme (grant number 739593), by a Momentum Research Grant from the Hungarian Academy of Sciences, by a National Research, Development and Innovation Office under grant 2022-1.1.1-KK-2022-00005 and an EU grant on project RRF-2.3.1-21-2022-00003. T.G.G. was supported by the Semmelweis 250 + Excellence PhD Scholarship (EFOP-3.6.3-VEKOP-16-2017-00009) and by the Gedeon Richter Talentum Foundation scholarship. Z.D.D. and T.G.G. was supported by the ÚNKP-23-4-II-SE and ÚNKP-23-II-SE-43 New National Excellence Program of the Ministry for Innovation and Technology from the National Research, Development and Innovation fund, respectively. H.Z. is supported by grant funding from the National Institutes of Health and National Heart, Lung, and Blood Institute (K08HL161405). M.K. receives support from AIRC IG grant 24988. W.C.M. is supported by the Mandema–Stipendium of the Junior Scientific Masterclass 2020-10 of the University Medical Center Groningen and by the Dutch Heart Foundation (Dekker grant 03-005-2021-T005). T.G.N. is supported by a gift from A. Curt Greer and P. Kohlberg and from C. and P. Kazilionis, the Michael and Kathryn Park Endowed Chair in Cardiology, by a Hassenfeld Scholar Award, and has additional grant funding from the National Institutes of Health and National Heart, Lung, and Blood Institute (R01HL137562 and K24HL150238).
Author information
Authors and Affiliations
Contributions
T.G.G. researched data for the article. T.G.G. and Z.V.V. wrote the article. All authors contributed to the discussion of content and to reviewing and editing the manuscript before submission.
Corresponding author
Ethics declarations
Competing interests
M.K. is listed as an inventor in a patent on the use of co-stimulation blockade in heart failure. W.C.M. received speaker fees from Daiichi Sankyo and Novartis. T.G.N. has been a consultant for and received fees from Bristol Myers Squibb, CRC Oncology, Genentech, Roche, Roivant, Sanofi and Race Oncology, and has received grant funding from AstraZeneca and Bristol Myers Squibb for work related to immune checkpoint inhibitors. T.R. is a co-founder of Bimyo, a company focusing on the development of cardioprotective peptides; is listed on patents for the use of BNIP3 peptides for the treatment of myocardial infarction; is listed on a patent on the transport and delivery of nitric oxide; and is also listed on a patent of an antihypertensive composition of nitrate or nitrate derivates. P.F. is the founder and CEO of the Pharmahungary Group, a group of research and development companies. All other authors declare no competing interests.
Peer review
Peer review information
Nature Reviews Cardiology thanks Joachim Alexandre, Franck Thuny and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Gergely, T.G., Drobni, Z.D., Kallikourdis, M. et al. Immune checkpoints in cardiac physiology and pathology: therapeutic targets for heart failure. Nat Rev Cardiol (2024). https://doi.org/10.1038/s41569-023-00986-9
Accepted:
Published:
DOI: https://doi.org/10.1038/s41569-023-00986-9