Abstract
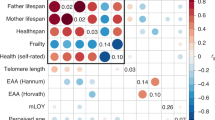
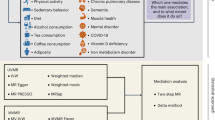
Mental well-being relates to multitudinous lifestyle behaviours and morbidities and underpins healthy aging. Thus far, causal evidence on whether and in what pattern mental well-being impacts healthy aging and the underlying mediating pathways is unknown. Applying genetic instruments of the well-being spectrum and its four dimensions including life satisfaction, positive affect, neuroticism and depressive symptoms (n = 80,852 to 2,370,390), we performed two-sample Mendelian randomization analyses to estimate the causal effect of mental well-being on the genetically independent phenotype of aging (aging-GIP), a robust and representative aging phenotype, and its components including resilience, self-rated health, healthspan, parental lifespan and longevity (n = 36,745 to 1,012,240). Analyses were adjusted for income, education and occupation. All the data were from the largest available genome-wide association studies in populations of European descent. Better mental well-being spectrum (each one Z-score higher) was causally associated with a higher aging-GIP (β [95% confidence interval (CI)] in different models ranging from 1.00 [0.82–1.18] to 1.07 [0.91–1.24] standard deviations (s.d.)) independent of socioeconomic indicators. Similar association patterns were seen for resilience (β [95% CI] ranging from 0.97 [0.82–1.12] to 1.04 [0.91–1.17] s.d.), self-rated health (0.61 [0.43–0.79] to 0.76 [0.59–0.93] points), healthspan (odds ratio [95% CI] ranging from 1.23 [1.02–1.48] to 1.35 [1.11–1.65]) and parental lifespan (1.77 [0.010–3.54] to 2.95 [1.13–4.76] years). Two-step Mendelian randomization mediation analyses identified 33 out of 106 candidates as mediators between the well-being spectrum and the aging-GIP: mainly lifestyles (for example, TV watching and smoking), behaviours (for example, medication use) and diseases (for example, heart failure, attention-deficit hyperactivity disorder, stroke, coronary atherosclerosis and ischaemic heart disease), each exhibiting a mediation proportion of >5%. These findings underscore the importance of mental well-being in promoting healthy aging and inform preventive targets for bridging aging disparities attributable to suboptimal mental health.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Data availability
All GWAS summary statistics analysed in this study are publicly available as shown in Table 1 and Supplementary Table 1 for download by qualified researchers. The GWAS data for mental well-being traits can be obtained from the GWAS catalogue38 (https://www.ebi.ac.uk/gwas/publications/30643256). The GWAS data for aging phenotypes can be retrieved or requested from the study authors at https://doi.org/10.7488/ds/2972 (the aging-GIP14), https://doi.org/10.6084/m9.figshare.9204998.v3 (frailty index42), http://ftp.ebi.ac.uk/pub/databases/gwas/summary_statistics/GCST006001-GCST007000/GCST006620 (self-rated health43), https://doi.org/10.5281/zenodo.1302861 (healthspan44), https://doi.org/10.7488/ds/2463 (parental lifespan45) and https://www.longevitygenomics.org/downloads (longevity46). All data generated in this study are included in the Supplementary Information.
Code availability
All the MR analyses were conducted using R packages TwoSampleMR (version 0.5.7), MVMR (version 0.4), MRPRESSO (version 1.0) and MRlap (version 0.0.3.0) in R software (version 4.3.1). Custom code that supports the findings of this study is available at https://github.com/yechaojie/mental_aging.
References
Oeppen, J. & Vaupel, J. W. Demography. Broken limits to life expectancy. Science 296, 1029–1031 (2002).
Ageing: a 21st century public health challenge? Lancet Public Health 2, e297 (2017).
Garmany, A., Yamada, S. & Terzic, A. Longevity leap: mind the healthspan gap. NPJ Regen. Med. 6, 57 (2021).
Mental health: strengthening our response. World Health Organization https://www.who.int/news-room/fact-sheets/detail/mental-health-strengthening-our-response (17 June 2022).
Prince, M. et al. No health without mental health. Lancet 370, 859–877 (2007).
Mak, H. W. et al. Hobby engagement and mental wellbeing among people aged 65 years and older in 16 countries. Nat. Med. 29, 2233–2240 (2023).
Steptoe, A. et al. Subjective wellbeing, health and ageing. Lancet 385, 640–648 (2015).
Jacobs, J. M. et al. Optimism and longevity beyond age 85. J. Gerontol. A Biol. Sci. Med. Sci. 76, 1806–1813 (2021).
Santamaria-Garcia, H. et al. Factors associated with healthy aging in Latin American populations. Nat. Med. 29, 2248–2258 (2023).
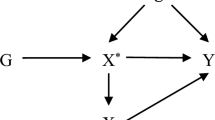
Sekula, P., Del Greco M, F., Pattaro, C. & Köttgen, A. Mendelian randomization as an approach to assess causality using observational data. J. Am. Soc. Nephrol. 27, 3253–3265 (2016).
Killingsworth, M. A., Kahneman, D. & Mellers, B. Income and emotional well-being: a conflict resolved. Proc. Natl Acad. Sci. USA 120, e2208661120 (2023).
Ridley, M., Rao, G., Schilbach, F. & Patel, V. Poverty, depression, and anxiety: causal evidence and mechanisms. Science 370, eaay0214 (2020).
Ye, C. J. et al. Mendelian randomization evidence for the causal effects of socio-economic inequality on human longevity among Europeans. Nat. Hum. Behav. 7, 1357–1370 (2023).
Timmers, P. R. H. J. et al. Mendelian randomization of genetically independent aging phenotypes identifies LPA and VCAM1 as biological targets for human aging. Nat. Aging 2, 19–30 (2022).
Rosoff, D. B. et al. Multivariate genome-wide analysis of aging-related traits identifies novel loci and new drug targets for healthy aging. Nat. Aging 3, 1020–1035 (2023).
Emdin, C. A., Khera, A. V. & Kathiresan, S. Mendelian randomization. JAMA 318, 1925–1926 (2017).
Carter, A. R. et al. Mendelian randomisation for mediation analysis: current methods and challenges for implementation. Eur. J. Epidemiol. 36, 465–478 (2021).
Killingsworth, M. A. Experienced well-being rises with income, even above $75,000 per year. Proc. Natl Acad. Sci. USA 118, e2016976118 (2021).
Krieger, N., Williams, D. R. & Moss, N. E. Measuring social class in US public health research: concepts, methodologies, and guidelines. Annu. Rev. Public Health 18, 341–378 (1997).
Khan, I. et al. Surrogate adiposity markers and mortality. JAMA Netw. Open 6, e2334836 (2023).
Johnson, A. A. & Stolzing, A. The role of lipid metabolism in aging, lifespan regulation, and age-related disease. Aging Cell 18, e13048 (2019).
Ye, C. et al. Causal associations of sarcopenia-related traits with cardiometabolic disease and Alzheimer’s disease and the mediating role of insulin resistance: a Mendelian randomization study. Aging Cell 22, e13923 (2023).
Furman, D. et al. Chronic inflammation in the etiology of disease across the life span. Nat. Med. 25, 1822–1832 (2019).
Yusuf, S. et al. Modifiable risk factors, cardiovascular disease, and mortality in 155 722 individuals from 21 high-income, middle-income, and low-income countries (PURE): a prospective cohort study. Lancet 395, 795–808 (2020).
Levine, G. N. et al. Psychological health, well-being, and the mind-heart-body connection: a scientific statement from the American Heart Association. Circulation 143, e763–e783 (2021).
Dalsgaard, S., Østergaard, S. D., Leckman, J. F., Mortensen, P. B. & Pedersen, M. G. Mortality in children, adolescents, and adults with attention deficit hyperactivity disorder: a nationwide cohort study. Lancet 385, 2190–2196 (2015).
Sun, S. et al. Association of psychiatric comorbidity with the risk of premature death among children and adults with attention-deficit/hyperactivity disorder. JAMA Psychiatry 76, 1141–1149 (2019).
GBD 2017 Causes of Death Collaborators. Global, regional, and national age-sex-specific mortality for 282 causes of death in 195 countries and territories, 1980-2017: a systematic analysis for the Global Burden of Disease Study 2017. Lancet 392, 1736–1788 (2018).
Robson, D. & Gray, R. Serious mental illness and physical health problems: a discussion paper. Int. J. Nurs. Stud. 44, 457–466 (2007).
Wang, Y. et al. Independent associations of education, intelligence, and cognition with hypertension and the mediating effects of cardiometabolic risk factors: a mendelian randomization study. Hypertension 80, 192–203 (2023).
Sturgeon, J. A., Finan, P. H. & Zautra, A. J. Affective disturbance in rheumatoid arthritis: psychological and disease-related pathways. Nat. Rev. Rheumatol. 12, 532–542 (2016).
Wu, X. et al. Investigating the relationship between depression and breast cancer: observational and genetic analyses. BMC Med. 21, 170 (2023).
Dockray, S. & Steptoe, A. Positive affect and psychobiological processes. Neurosci. Biobehav. Rev. 35, 69–75 (2010).
Zhao, W. et al. Identification of new susceptibility loci for type 2 diabetes and shared etiological pathways with coronary heart disease. Nat. Genet. 49, 1450–1457 (2017).
Carreras-Torres, R. et al. Role of obesity in smoking behaviour: Mendelian randomisation study in UK Biobank. Br. Med. J. 361, k1767 (2018).
Klimentidis, Y. C. et al. Genome-wide association study of habitual physical activity in over 377,000 UK Biobank participants identifies multiple variants including CADM2 and APOE. Int. J. Obes. 42, 1161–1176 (2018).
Skrivankova, V. W. et al. Strengthening the reporting of observational studies in epidemiology using Mendelian randomization: the STROBE-MR Statement. JAMA 326, 1614–1621 (2021).
Baselmans, B. M. L. et al. Multivariate genome-wide analyses of the well-being spectrum. Nat. Genet. 51, 445–451 (2019).
Lee, J. J. et al. Gene discovery and polygenic prediction from a genome-wide association study of educational attainment in 1.1 million individuals. Nat. Genet. 50, 1112–1121 (2018).
Mitchell, R. E. et al. MRC IEU UK Biobank GWAS Pipeline Version 2. University of Bristol https://doi.org/10.5523/bris.pnoat8cxo0u52p6ynfaekeigi (2019).
Ko, H. et al. Genome-wide association study of occupational attainment as a proxy for cognitive reserve. Brain 145, 1436–1448 (2022).
Atkins, J. L. et al. A genome-wide association study of the frailty index highlights brain pathways in ageing. Aging Cell 20, e13459 (2021).
Harris, S. E. et al. Molecular genetic contributions to self-rated health. Int. J. Epidemiol. 46, 994–1009 (2017).
Zenin, A. et al. Identification of 12 genetic loci associated with human healthspan. Commun. Biol. 2, 41 (2019).
Timmers, P. R. et al. Genomics of 1 million parent lifespans implicates novel pathways and common diseases and distinguishes survival chances. eLife 8, e39856 (2019).
Deelen, J. et al. A meta-analysis of genome-wide association studies identifies multiple longevity genes. Nat. Commun. 10, 3669 (2019).
1000 Genomes Project Consortium et al. A global reference for human genetic variation. Nature 526, 68–74 (2015).
International HapMap 3 Consortium et al. Integrating common and rare genetic variation in diverse human populations. Nature 467, 52–58 (2010).
Machiela, M. J. & Chanock, S. J. LDlink: a web-based application for exploring population-specific haplotype structure and linking correlated alleles of possible functional variants. Bioinformatics 31, 3555–3557 (2015).
Bowden, J. et al. A framework for the investigation of pleiotropy in two-sample summary data Mendelian randomization. Stat. Med. 36, 1783–1802 (2017).
Grant, A. J. & Burgess, S. Pleiotropy robust methods for multivariable Mendelian randomization. Stat. Med. 40, 5813–5830 (2021).
Burgess, S. et al. Guidelines for performing Mendelian randomization investigations: update for summer 2023. Wellcome Open Res. 4, 186 (2023).
Burgess, S., Davies, N. M. & Thompson, S. G. Bias due to participant overlap in two-sample Mendelian randomization. Genet. Epidemiol. 40, 597–608 (2016).
Mounier, N. & Kutalik, Z. Bias correction for inverse variance weighting Mendelian randomization. Genet. Epidemiol. 47, 314–331 (2023).
MacKinnon, D. P., Lockwood, C. M., Hoffman, J. M., West, S. G. & Sheets, V. A comparison of methods to test mediation and other intervening variable effects. Psychol. Methods 7, 83–104 (2002).
Bowden, J., Davey Smith, G., Haycock, P. C. & Burgess, S. Consistent estimation in Mendelian randomization with some invalid instruments using a weighted median estimator. Genet. Epidemiol. 40, 304–314 (2016).
Hartwig, F. P., Davey Smith, G. & Bowden, J. Robust inference in summary data Mendelian randomization via the zero modal pleiotropy assumption. Int. J. Epidemiol. 46, 1985–1998 (2017).
Bowden, J., Davey Smith, G. & Burgess, S. Mendelian randomization with invalid instruments: effect estimation and bias detection through Egger regression. Int. J. Epidemiol. 44, 512–525 (2015).
Verbanck, M., Chen, C. Y., Neale, B. & Do, R. Detection of widespread horizontal pleiotropy in causal relationships inferred from Mendelian randomization between complex traits and diseases. Nat. Genet. 50, 693–698 (2018).
Burgess, S. & Thompson, S. G. Multivariable Mendelian randomization: the use of pleiotropic genetic variants to estimate causal effects. Am. J. Epidemiol. 181, 251–260 (2015).
Glickman, M. E., Rao, S. R. & Schultz, M. R. False discovery rate control is a recommended alternative to Bonferroni-type adjustments in health studies. J. Clin. Epidemiol. 67, 850–857 (2014).
Acknowledgements
This work was supported by the grants from the National Natural Science Foundation of China (82370820, 82088102, 91857205, 823B2014 and 81930021), the ‘Shanghai Municipal Education Commission–Gaofeng Clinical Medicine Grant Support’ from Shanghai Jiao Tong University School of Medicine (20171901 Round 2), and the Innovative Research Team of High-level Local Universities in Shanghai. The funders had no role in study design, data collection and analysis, decision to publish or preparation of the manuscript. The authors are grateful to the participants of all the GWASs used in this manuscript and the investigators who made these GWAS data publicly available.
Author information
Authors and Affiliations
Contributions
C.-J.Y. and T.-G.W. contributed to the conception and design of the study. C.-J.Y. performed statistical analyses and drafted the manuscript. T.-G.W. critically revised the manuscript. D.L., M.-L.C. and T.-G.W. checked the statistical analysis and proofread the manuscript. T.-G.W., G.N., W.-Q.W. and C.-J.Y. obtained funding. All authors contributed to the acquisition or interpretation of data, proofreading of the manuscript for important intellectual content and the final approval of the version to be published. T.-G.W. is the guarantor of this work and takes responsibility for the integrity of the data and the accuracy of the data analysis.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Human Behaviour thanks Taiji Noguchi, Yongfu Yu and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
Supplementary Methods 1 and 2 and Tables 1–19.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Ye, CJ., Liu, D., Chen, ML. et al. Mendelian randomization evidence for the causal effect of mental well-being on healthy aging. Nat Hum Behav 8, 1798–1809 (2024). https://doi.org/10.1038/s41562-024-01905-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41562-024-01905-9