Abstract
Receiving touch is of critical importance, as many studies have shown that touch promotes mental and physical well-being. We conducted a pre-registered (PROSPERO: CRD42022304281) systematic review and multilevel meta-analysis encompassing 137 studies in the meta-analysis and 75 additional studies in the systematic review (n = 12,966 individuals, search via Google Scholar, PubMed and Web of Science until 1 October 2022) to identify critical factors moderating touch intervention efficacy. Included studies always featured a touch versus no touch control intervention with diverse health outcomes as dependent variables. Risk of bias was assessed via small study, randomization, sequencing, performance and attrition bias. Touch interventions were especially effective in regulating cortisol levels (Hedges’ g = 0.78, 95% confidence interval (CI) 0.24 to 1.31) and increasing weight (0.65, 95% CI 0.37 to 0.94) in newborns as well as in reducing pain (0.69, 95% CI 0.48 to 0.89), feelings of depression (0.59, 95% CI 0.40 to 0.78) and state (0.64, 95% CI 0.44 to 0.84) or trait anxiety (0.59, 95% CI 0.40 to 0.77) for adults. Comparing touch interventions involving objects or robots resulted in similar physical (0.56, 95% CI 0.24 to 0.88 versus 0.51, 95% CI 0.38 to 0.64) but lower mental health benefits (0.34, 95% CI 0.19 to 0.49 versus 0.58, 95% CI 0.43 to 0.73). Adult clinical cohorts profited more strongly in mental health domains compared with healthy individuals (0.63, 95% CI 0.46 to 0.80 versus 0.37, 95% CI 0.20 to 0.55). We found no difference in health benefits in adults when comparing touch applied by a familiar person or a health care professional (0.51, 95% CI 0.29 to 0.73 versus 0.50, 95% CI 0.38 to 0.61), but parental touch was more beneficial in newborns (0.69, 95% CI 0.50 to 0.88 versus 0.39, 95% CI 0.18 to 0.61). Small but significant small study bias and the impossibility to blind experimental conditions need to be considered. Leveraging factors that influence touch intervention efficacy will help maximize the benefits of future interventions and focus research in this field.
Similar content being viewed by others
Main
The sense of touch has immense importance for many aspects of our life. It is the first of all the senses to develop in newborns1 and the most direct experience of contact with our physical and social environment2. Complementing our own touch experience, we also regularly receive touch from others around us, for example, through consensual hugs, kisses or massages3.
The recent coronavirus pandemic has raised awareness regarding the need to better understand the effects that touch—and its reduction during social distancing—can have on our mental and physical well-being. The most common touch interventions, for example, massage for adults or kangaroo care for newborns, have been shown to have a wide range of both mental and physical health benefits, from facilitating growth and development to buffering against anxiety and stress, over the lifespan of humans and animals alike4. Despite the substantial weight this literature gives to support the benefits of touch, it is also characterized by a large variability in, for example, studied cohorts (adults, children, newborns and animals), type and duration of applied touch (for example, one-time hug versus repeated 60-min massages), measured health outcomes (ranging from physical health outcomes such as sleep and blood pressure to mental health outcomes such as depression or mood) and who actually applies the touch (for example, partner versus stranger).
A meaningful tool to make sense of this vast amount of research is through meta-analysis. While previous meta-analyses on this topic exist, they were limited in scope, focusing only on particular types of touch, cohorts or specific health outcomes (for example, refs. 5,6). Furthermore, despite best efforts, meaningful variables that moderate the efficacy of touch interventions could not yet be identified. However, understanding these variables is critical to tailor touch interventions and guide future research to navigate this diverse field with the ultimate aim of promoting well-being in the population.
In this Article, we describe a pre-registered, large-scale systematic review and multilevel, multivariate meta-analysis to address this need with quantitative evidence for (1) the effect of touch interventions on physical and mental health and (2) which moderators influence the efficacy of the intervention. In particular, we ask whether and how strongly health outcomes depend on the dynamics of the touching dyad (for example, humans or robots/objects, familiarity and touch directionality), demographics (for example, clinical status, age or sex), delivery means (for example, type of touch intervention or touched body part) and procedure (for example, duration or number of sessions). We did so separately for newborns and for children and adults, as the health outcomes in newborns differed substantially from those in the other age groups. Despite the focus of the analysis being on humans, it is widely known that many animal species benefit from touch interactions and that engaging in touch promotes their well-being as well7. Since animal models are essential for the investigation of the mechanisms underlying biological processes and for the development of therapeutic approaches, we accordingly included health benefits of touch interventions in non-human animals as part of our systematic review. However, this search yielded only a small number of studies, suggesting a lack of research in this domain, and as such, was insufficient to be included in the meta-analysis. We evaluate the identified animal studies and their findings in the discussion.
Results
Touch interventions have a medium-sized effect
The pre-registration can be found at ref. 8. The flowchart for data collection and extraction is depicted in Fig. 1.
For adults, a total of n = 2,841 and n = 2,556 individuals in the touch and control groups, respectively, across 85 studies and 103 cohorts were included. The effect of touch overall was medium-sized (t(102) = 9.74, P < 0.001, Hedges’ g = 0.52, 95% confidence interval (CI) 0.42 to 0.63; Fig. 2a). For newborns, we could include 63 cohorts across 52 studies comprising a total of n = 2,134 and n = 2,086 newborns in the touch and control groups, respectively, with an overall effect almost identical to the older age group (t(62) = 7.53, P < 0.001, Hedges’ g = 0.56, 95% CI 0.41 to 0.71; Fig. 2b), suggesting that, despite distinct health outcomes, touch interventions show comparable effects across newborns and adults. Using these overall effect estimates, we conducted a power sensitivity analysis of all the included primary studies to investigate whether such effects could be reliably detected9. Sufficient power to detect such effect sizes was rare in individual studies, as investigated by firepower plots10 (Supplementary Figs. 1 and 2). No individual effect size from either meta-analysis was overly influential (Cook’s D < 0.06). The benefits were similar for mental and physical outcomes (mental versus physical; adults: t(101) = 0.79, P = 0.432, Hedges’ g difference of −0.05, 95% CI −0.16 to 0.07, Fig. 2c; newborns: t(61) = 1.08, P = 0.284, Hedges’ g difference of −0.19, 95% CI −0.53 to 0.16, Fig. 2d).
a, Orchard plot illustrating the overall benefits across all health outcomes for adults/children across 469 in part dependent effect sizes from 85 studies and 103 cohorts. b, The same as a but for newborns across 174 in part dependent effect sizes from 52 studies and 63 cohorts. c, The same as a but separating the results for physical versus mental health benefits across 469 in part dependent effect sizes from 85 studies and 103 cohorts. d, The same as b but separating the results for physical versus mental health benefits across 172 in part dependent effect sizes from 52 studies and 63 cohorts. Each dot reflects a measured effect, and the number of effects (k) included in the analysis is depicted in the bottom left. Mean effects and 95% CIs are presented in the bottom right and are indicated by the central black dot (mean effect) and its error bars (95% CI). The heterogeneity Q statistic is presented in the top left. Overall effects of moderator impact were assessed via an F test, and post hoc comparisons were done using t tests (two-sided test). Note that the P values above the mean effects indicate whether an effect differed significantly from a zero effect. P values were not corrected for multiple comparisons. The dot size reflects the precision of each individual effect (larger indicates higher precision). Small-study bias for the overall effect was significant (F test, two-sided test) in the adult meta-analysis (F(1, 101) = 21.24, P < 0.001; Supplementary Fig. 3) as well as in the newborn meta-analysis (F(1, 61) = 5.25, P = 0.025; Supplementary Fig. 4).
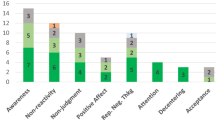
On the basis of the overall effect of both meta-analyses as well as their median sample sizes, the minimum number of studies necessary for subgroup analyses to achieve 80% power was k = 9 effects for adults and k = 8 effects for newborns (Supplementary Figs. 5 and 6). Assessing specific health outcomes with sufficient power in more detail in adults (Fig. 3a) revealed smaller benefits to sleep and heart rate parameters, moderate benefits to positive and negative affect, diastolic blood and systolic blood pressure, mobility and reductions of the stress hormone cortisol and larger benefits to trait and state anxiety, depression, fatigue and pain. Post hoc tests revealed stronger benefits for pain, state anxiety, depression and trait anxiety compared with respiratory, sleep and heart rate parameters (see Fig. 3 for all post hoc comparisons). Reductions in pain and state anxiety were increased compared with reductions in negative affect (t(83) = 2.54, P = 0.013, Hedges’ g difference of 0.31, 95% CI 0.07 to 0.55; t(83) = 2.31, P = 0.024, Hedges’ g difference of 0.27, 95% CI 0.03 to 0.51). Benefits to pain symptoms were higher compared with benefits to positive affect (t(83) = 2.22, P = 0.030, Hedges’ g difference of 0.29, 95% CI 0.04 to 0.54). Finally, touch resulted in larger benefits to cortisol release compared with heart rate parameters (t(83) = 2.30, P = 0.024, Hedges’ g difference of 0.26, 95% CI 0.04–0.48).
a,b, Health outcomes in adults analysed across 405 in part dependent effect sizes from 79 studies and 97 cohorts (a) and in newborns analysed across 105 in part dependent effect sizes from 46 studies and 56 cohorts (b). The type of health outcomes measured differed between adults and newborns and were thus analysed separately. Numbers on the right represent the mean effect with its 95% CI in square brackets and the significance level estimating the likelihood that the effect is equal to zero. Overall effects of moderator impact were assessed via an F test, and post hoc comparisons were done using t tests (two-sided test). The F value in the top right represents a test of the hypothesis that all effects within the subpanel are equal. The Q statistic represents the heterogeneity. P values of post hoc tests are depicted whenever significant. P values above the horizontal whiskers indicate whether an effect differed significantly from a zero effect. Vertical lines indicate significant post hoc tests between moderator levels. P values were not corrected for multiple comparisons. Physical outcomes are marked in red. Mental outcomes are marked in blue.
In newborns, only physical health effects offered sufficient data for further analysis. We found no benefits for digestion and heart rate parameters. All other health outcomes (cortisol, liver enzymes, respiration, temperature regulation and weight gain) showed medium to large effects (Fig. 3b). We found no significant differences among any specific health outcomes.
Non-human touch and skin-to-skin contact
In some situations, a fellow human is not readily available to provide affective touch, raising the question of the efficacy of touch delivered by objects and robots11. Overall, we found humans engaging in touch with other humans or objects to have medium-sized health benefits in adults, without significant differences (t(99) = 1.05, P = 0.295, Hedges’ g difference of 0.12, 95% CI −0.11 to 0.35; Fig. 4a). However, differentiating physical versus mental health benefits revealed similar benefits for human and object touch on physical health outcomes, but larger benefits on mental outcomes when humans were touched by humans (t(97) = 2.32, P = 0.022, Hedges’ g difference of 0.24, 95% CI 0.04 to 0.44; Fig. 4b). It must be noted that touching with an object still showed a significant effect (see Supplementary Fig. 7 for the corresponding orchard plot).
a, Forest plot comparing humans versus objects touching a human on health outcomes overall across 467 in part dependent effect sizes from 85 studies and 101 cohorts. b, The same as a but separately for mental versus physical health outcomes across 467 in part dependent effect sizes from 85 studies and 101 cohorts. c, Results with the removal of all object studies, leaving 406 in part dependent effect sizes from 71 studies and 88 cohorts to identify whether missing skin-to-skin contact is the relevant mediator of higher mental health effects in human–human interactions. Numbers on the right represent the mean effect with its 95% CI in square brackets and the significance level estimating the likelihood that the effect is equal to zero. Overall effects of moderator impact were assessed via an F test, and post hoc comparisons were done using t tests (two-sided test). The F value in the top right represents a test of the hypothesis that all effects within the subpanel are equal. The Q statistic represents the heterogeneity. P values of post hoc tests are depicted whenever significant. P values above the horizontal whiskers indicate whether an effect differed significantly from a zero effect. Vertical lines indicate significant post hoc tests between moderator levels. P values were not corrected for multiple comparisons. Physical outcomes are marked in red. Mental outcomes are marked in blue.
We considered the possibility that this effect was due to missing skin-to-skin contact in human–object interactions. Thus, we investigated human–human interactions with and without skin-to-skin contact (Fig. 4c). In line with the hypothesis that skin-to-skin contact is highly relevant, we again found stronger mental health benefits in the presence of skin-to-skin contact that however did not achieve nominal significance (t(69) = 1.95, P = 0.055, Hedges’ g difference of 0.41, 95% CI −0.00 to 0.82), possibly because skin-to-skin contact was rarely absent in human–human interactions, leading to a decrease in power of this analysis. Results for skin-to-skin contact as an overall moderator can be found in Supplementary Fig. 8.
Influences of type of touch
The large majority of touch interventions comprised massage therapy in adults and kangaroo care in newborns (see Supplementary Table 1 for a complete list of interventions across studies). However, comparing the different types of touch explored across studies did not reveal significant differences in effect sizes based on touch type, be it on overall health benefits (adults: t(101) = 0.11, P = 0.916, Hedges’ g difference of 0.02, 95% CI −0.32 to 0.29; Fig. 5a) or comparing different forms of touch separately for physical (massage therapy versus other forms: t(99) = 0.99, P = 0.325, Hedges’ g difference 0.16, 95% CI −0.15 to 0.47) or for mental health benefits (massage therapy versus other forms: t(99) = 0.75, P = 0.458, Hedges’ g difference of 0.13, 95% CI −0.22 to 0.48) in adults (Fig. 5c; see Supplementary Fig. 9 for the corresponding orchard plot). A similar picture emerged for physical health effects in newborns (massage therapy versus kangaroo care: t(58) = 0.94, P = 0.353, Hedges’ g difference of 0.15, 95% CI −0.17 to 0.47; massage therapy versus other forms: t(58) = 0.56, P = 0.577, Hedges’ g difference of 0.13, 95% CI −0.34 to 0.60; kangaroo care versus other forms: t(58) = 0.07, P = 0.947, Hedges’ g difference of 0.02, 95% CI −0.46 to 0.50; Fig. 5d; see also Supplementary Fig. 10 for the corresponding orchard plot). This suggests that touch types may be flexibly adapted to the setting of every touch intervention.
a, Forest plot of health benefits comparing massage therapy versus other forms of touch in adult cohorts across 469 in part dependent effect sizes from 85 studies and 103 cohorts. b, Forest plot of health benefits comparing massage therapy, kangaroo care and other forms of touch for newborns across 174 in part dependent effect sizes from 52 studies and 63 cohorts. c, The same as a but separating mental and physical health benefits across 469 in part dependent effect sizes from 85 studies and 103 cohorts. d, The same as b but separating mental and physical health outcomes where possible across 164 in part dependent effect sizes from 51 studies and 62 cohorts. Note that an insufficient number of studies assessed mental health benefits of massage therapy or other forms of touch to be included. Numbers on the right represent the mean effect with its 95% CI in square brackets and the significance level estimating the likelihood that the effect is equal to zero. Overall effects of moderator impact were assessed via an F test, and post hoc comparisons were done using t tests (two-sided test). The F value in the top right represents a test of the hypothesis that all effects within the subpanel are equal. The Q statistic represents heterogeneity. P values of post hoc tests are depicted whenever significant. P values above the horizontal whiskers indicate whether an effect differed significantly from a zero effect. Vertical lines indicate significant post hoc tests between moderator levels. P values were not corrected for multiple comparisons. Physical outcomes are marked in red. Mental outcomes are marked in blue.
The role of clinical status
Most research on touch interventions has focused on clinical samples, but are benefits restricted to clinical cohorts? We found health benefits to be significant in clinical and healthy populations (Fig. 6), whether all outcomes are considered (Fig. 6a,b) or physical and mental health outcomes are separated (Fig. 6c,d, see Supplementary Figs. 11 and 12 for the corresponding orchard plots). In adults, however, we found higher mental health benefits for clinical populations compared with healthy ones (Fig. 6c; t(99) = 2.11, P = 0.037, Hedges’ g difference of 0.25, 95% CI 0.01 to 0.49).
a, Health benefits for clinical cohorts of adults versus healthy cohorts of adults across 469 in part dependent effect sizes from 85 studies and 103 cohorts. b, The same as a but for newborn cohorts across 174 in part dependent effect sizes from 52 studies and 63 cohorts. c, The same as a but separating mental versus physical health benefits across 469 in part dependent effect sizes from 85 studies and 103 cohorts. d, The same as b but separating mental versus physical health benefits across 172 in part dependent effect sizes from 52 studies and 63 cohorts. Numbers on the right represent the mean effect with its 95% CI in square brackets and the significance level estimating the likelihood that the effect is equal to zero. Overall effects of moderator impact were assessed via an F test, and post hoc comparisons were done using t tests (two-sided test).The F value in the top right represents a test of the hypothesis that all effects within the subpanel are equal. The Q statistic represents the heterogeneity. P values of post hoc tests are depicted whenever significant. P values above the horizontal whiskers indicate whether an effect differed significantly from a zero effect. Vertical lines indicate significant post hoc tests between moderator levels. P values were not corrected for multiple comparisons. Physical outcomes are marked in red. Mental outcomes are marked in blue.
A more detailed analysis of specific clinical conditions in adults revealed positive mental and physical health benefits for almost all assessed clinical disorders. Differences between disorders were not found, with the exception of increased effectiveness of touch interventions in neurological disorders (Supplementary Fig. 13).
Familiarity in the touching dyad and intervention location
Touch interventions can be performed either by familiar touchers (partners, family members or friends) or by unfamiliar touchers (health care professionals). In adults, we did not find an impact of familiarity of the toucher (t(99) = 0.12, P = 0.905, Hedges’ g difference of 0.02, 95% CI −0.27 to 0.24; Fig. 7a; see Supplementary Fig. 14 for the corresponding orchard plot). Similarly, investigating the impact on mental and physical health benefits specifically, no significant differences could be detected, suggesting that familiarity is irrelevant in adults. In contrast, touch applied to newborns by their parents (almost all studies only included touch by the mother) was significantly more beneficial compared with unfamiliar touch (t(60) = 2.09, P = 0.041, Hedges’ g difference of 0.30, 95% CI 0.01 to 0.59) (Fig. 7b; see Supplementary Fig. 15 for the corresponding orchard plot). Investigating mental and physical health benefits specifically revealed no significant differences. Familiarity with the location in which the touch was applied (familiar being, for example, the participants’ home) did not influence the efficacy of touch interventions (Supplementary Fig. 16).
a, Health benefits for being touched by a familiar (for example, partner, family member or friend) versus unfamiliar toucher (health care professional) across 463 in part dependent effect sizes from 83 studies and 101 cohorts. b, The same as a but for newborn cohorts across 171 in part dependent effect sizes from 51 studies and 62 cohorts. c, The same as a but separating mental versus physical health benefits across 463 in part dependent effect sizes from 83 studies and 101 cohorts. d, The same as b but separating mental versus physical health benefits across 169 in part dependent effect sizes from 51 studies and 62 cohorts. Numbers on the right represent the mean effect with its 95% CI in square brackets and the significance level estimating the likelihood that the effect is equal to zero. Overall effects of moderator impact were assessed via an F test, and post hoc comparisons were done using t tests (two-sided test). The F value in the top right represents a test of the hypothesis that all effects within the subpanel are equal. The Q statistic represents the heterogeneity. P values of post hoc tests are depicted whenever significant. P values above the horizontal whiskers indicate whether an effect differed significantly from a zero effect. Vertical lines indicate significant post hoc tests between moderator levels. P values were not corrected for multiple comparisons. Physical outcomes are marked in red. Mental outcomes are marked in blue.
Frequency and duration of touch interventions
How often and for how long should touch be delivered? For adults, the median touch duration across studies was 20 min and the median number of touch interventions was four sessions with an average time interval of 2.3 days between each session. For newborns, the median touch duration across studies was 17.5 min and the median number of touch interventions was seven sessions with an average time interval of 1.3 days between each session.
Delivering more touch sessions increased benefits in adults, whether overall (t(101) = 4.90, P < 0.001, Hedges’ g = 0.02, 95% CI 0.01 to 0.03), physical (t(81) = 3.07, P = 0.003, Hedges’ g = 0.02, 95% CI 0.01–0.03) or mental benefits (t(72) = 5.43, P < 0.001, Hedges’ g = 0.02, 95% CI 0.01–0.03) were measured (Fig. 8a). A closer look at specific outcomes for which sufficient data were available revealed that positive associations between the number of sessions and outcomes were found for trait anxiety (t(12) = 7.90, P < 0.001, Hedges’ g = 0.03, 95% CI 0.02–0.04), depression (t(20) = 10.69, P < 0.001, Hedges’ g = 0.03, 95% CI 0.03–0.04) and pain (t(37) = 3.65, P < 0.001, Hedges’ g = 0.03, 95% CI 0.02–0.05), indicating a need for repeated sessions to improve these adverse health outcomes. Neither increasing the number of sessions for newborns nor increasing the duration of touch per session in adults or newborns increased health benefits, be they physical or mental (Fig. 8b–d). For continuous moderators in adults, we also looked at specific health outcomes as sufficient data were generally available for further analysis. Surprisingly, we found significant negative associations between touch duration and reductions of cortisol (t(24) = 2.71, P = 0.012, Hedges’ g = −0.01, 95% CI −0.01 to −0.00) and heart rate parameters (t(21) = 2.35, P = 0.029, Hedges’ g = −0.01, 95% CI −0.02 to −0.00).
a, Meta-regression analysis examining the association between the number of sessions applied and the effect size in adults, either on overall health benefits (left, 469 in part dependent effect sizes from 85 studies and 103 cohorts) or for physical (middle, 245 in part dependent effect sizes from 69 studies and 83 cohorts) or mental benefits (right, 224 in part dependent effect sizes from 60 studies and 74 cohorts) separately. b, The same as a for newborns (overall: 150 in part dependent effect sizes from 46 studies and 53 cohorts; physical health: 127 in part dependent effect sizes from 44 studies and 51 cohorts; mental health: 21 in part dependent effect sizes from 11 studies and 12 cohorts). c,d the same as a (c) and b (d) but for the duration of the individual sessions. For adults, 449 in part dependent effect sizes across 80 studies and 96 cohorts were included in the overall analysis. The analysis of physical health benefits included 240 in part dependent effect sizes across 67 studies and 80 cohorts, and the analysis of mental health benefits included 209 in part dependent effect sizes from 56 studies and 69 cohorts. For newborns, 145 in part dependent effect sizes across 45 studies and 52 cohorts were included in the overall analysis. The analysis of physical health benefits included 122 in part dependent effect sizes across 43 studies and 50 cohorts, and the analysis of mental health benefits included 21 in part dependent effect sizes from 11 studies and 12 cohorts. Each dot represents an effect size. Its size indicates the precision of the study (larger indicates better). Overall effects of moderator impact were assessed via an F test (two-sided test). The P values in each panel represent the result of a regression analysis testing the hypothesis that the slope of the relationship is equal to zero. P values are not corrected for multiple testing. The shaded area around the regression line represents the 95% CI.
Demographic influences of sex and age
We used the ratio between women and men in the single-study samples as a proxy for sex-specific effects. Sex ratios were heavily skewed towards larger numbers of women in each cohort (median 83% women), and we could not find significant associations between sex ratio and overall (t(62) = 0.08, P = 0.935, Hedges’ g = 0.00, 95% CI −0.00 to 0.01), mental (t(43) = 0.55, P = 0.588, Hedges’ g = 0.00, 95% CI −0.00 to 0.01) or physical health benefits (t(51) = 0.15, P = 0.882, Hedges’ g = −0.00, 95% CI −0.01 to 0.01). For specific outcomes that could be further analysed, we found a significant positive association of sex ratio with reductions in cortisol secretion (t(18) = 2.31, P = 0.033, Hedges’ g = 0.01, 95% CI 0.00 to 0.01) suggesting stronger benefits in women. In contrast to adults, sex ratios were balanced in samples of newborns (median 53% girls). For newborns, there was no significant association with overall (t(36) = 0.77, P = 0.447, Hedges’ g = −0.01, 95% CI −0.02 to 0.01) and physical health benefits of touch (t(35) = 0.93, P = 0.359, Hedges’ g = −0.01, 95% CI −0.02 to 0.01). Mental health benefits did not provide sufficient data for further analysis.
The median age in the adult meta-analysis was 42.6 years (s.d. 21.16 years, range 4.5–88.4 years). There was no association between age and the overall (t(73) = 0.35, P = 0.727, Hedges’ g = 0.00, 95% CI −0.01 to 0.01), mental (t(53) = 0.94, P = 0.353, Hedges’ g = 0.01, 95% CI −0.01 to 0.02) and physical health benefits of touch (t(60) = 0.16, P = 0.870, Hedges’ g = 0.00, 95% CI −0.01 to 0.01). Looking at specific health outcomes, we found significant positive associations between mean age and improved positive affect (t(10) = 2.54, P = 0.030, Hedges’ g = 0.01, 95% CI 0.00 to 0.02) as well as systolic blood pressure (t(11) = 2.39, P = 0.036, Hedges’ g = 0.02, 95% CI 0.00 to 0.04).
Body part
A list of touched body parts can be found in Supplementary Table 1. For the touched body part, we found significantly higher health benefits for head touch compared with arm touch (t(40) = 2.14, P = 0.039, Hedges’ g difference of 0.78, 95% CI 0.07 to 1.49) and torso touch (t(40) = 2.23, P = 0.031; Hedges’ g difference of 0.84, 95% CI 0.10 to 1.58; Supplementary Fig. 17). Touching the arm resulted in lower mental health compared with physical health benefits (t(37) = 2.29, P = 0.028, Hedges’ g difference of −0.35, 95% CI −0.65 to −0.05). Furthermore, we found a significantly increased physical health benefit when the head was touched as opposed to the torso (t(37) = 2.10, P = 0.043, Hedges’ g difference of 0.96, 95% CI 0.06 to 1.86). Thus, head touch such as a face or scalp massage could be especially beneficial.
Directionality
In adults, we tested whether a uni- or bidirectional application of touch mattered. The large majority of touch was applied unidirectionally (k = 442 of 469 effects). Unidirectional touch had higher health benefits (t(101) = 2.17, P = 0.032, Hedges’ g difference of 0.30, 95% CI 0.03 to 0.58) than bidirectional touch. Specifically, mental health benefits were higher in unidirectional touch (t(99) = 2.33, P = 0.022, Hedges’ g difference of 0.46, 95% CI 0.06 to 0.66).
Study location
For adults, we found significantly stronger health benefits of touch in South American compared with North American cohorts (t(95) = 2.03, P = 0.046, Hedges’ g difference of 0.37, 95% CI 0.01 to 0.73) and European cohorts (t(95) = 2.22, P = 0.029, Hedges’ g difference of 0.36, 95% CI 0.04 to 0.68). For newborns, we found weaker effects in North American cohorts compared to Asian (t(55) = 2.28, P = 0.026, Hedges’ g difference of −0.37, 95% CI −0.69 to −0.05) and European cohorts (t(55) = 2.36, P = 0.022, Hedges’ g difference of −0.40, 95% CI −0.74 to −0.06). Investigating the interaction with mental and physical health benefits did not reveal any effects of study location in both meta-analyses (Supplementary Fig. 18).
Systematic review of studies without effect sizes
All studies where effect size data could not be obtained or that did not meet the meta-analysis inclusion criteria can be found on the OSF project12 in the file ‘Study_lists_final_revised.xlsx’ (sheet ‘Studies_without_effect_sizes’). Specific reasons for exclusion are furthermore documented in Supplementary Table 2. For human health outcomes assessed across 56 studies and n = 2,438 individuals, interventions mostly comprised massage therapy (k = 86 health outcomes) and kangaroo care (k = 33 health outcomes). For datasets where no effect size could be computed, 90.0% of mental health and 84.3% of physical health parameters were positively impacted by touch. Positive impact of touch did not differ between types of touch interventions. These results match well with the observations of the meta-analysis of a highly positive benefit of touch overall, irrespective of whether a massage or any other intervention is applied.
We also assessed health outcomes in animals across 19 studies and n = 911 subjects. Most research was conducted in rodents. Animals that received touch were rats (ten studies, k = 16 health outcomes), mice (four studies, k = 7 health outcomes), macaques (two studies, k = 3 health outcomes), cats (one study, k = 3 health outcomes), lambs (one study, k = 2 health outcomes) and coral reef fish (one study, k = 1 health outcome). Touch interventions mostly comprised stroking (k = 13 health outcomes) and tickling (k = 10 health outcomes). For animal studies, 71.4% of effects showed benefits to mental health-like parameters and 81.8% showed positive physical health effects. We thus found strong evidence that touch interventions, which were mostly conducted by humans (16 studies with human touch versus 3 studies with object touch), had positive health effects in animal species as well.
Discussion
The key aim of the present study was twofold: (1) to provide an estimate of the effect size of touch interventions and (2) to disambiguate moderating factors to potentially tailor future interventions more precisely. Overall, touch interventions were beneficial for both physical and mental health, with a medium effect size. Our work illustrates that touch interventions are best suited for reducing pain, depression and anxiety in adults and children as well as for increasing weight gain in newborns. These findings are in line with previous meta-analyses on this topic, supporting their conclusions and their robustness to the addition of more datasets. One limitation of previous meta-analyses is that they focused on specific health outcomes or populations, despite primary studies often reporting effects on multiple health parameters simultaneously (for example, ref. 13 focusing on neck and shoulder pain and ref. 14 focusing on massage therapy in preterms). To our knowledge, only ref. 5 provides a multivariate picture for a large number of dependent variables. However, this study analysed their data in separate random effects models that did not account for multivariate reporting nor for the multilevel structure of the data, as such approaches have only become available recently. Thus, in addition to adding a substantial amount of new data, our statistical approach provides a more accurate depiction of effect size estimates. Additionally, our study investigated a variety of moderating effects that did not reach significance (for example, sex ratio, mean age or intervention duration) or were not considered (for example, the benefits of robot or object touch) in previous meta-analyses in relation to touch intervention efficacy5, probably because of the small number of studies with information on these moderators in the past. Owing to our large-scale approach, we reached high statistical power for many moderator analyses. Finally, previous meta-analyses on this topic exclusively focused on massage therapy in adults or kangaroo care in newborns15, leaving out a large number of interventions that are being carried out in research as well as in everyday life to improve well-being. Incorporating these studies into our study, we found that, in general, both massages and other types of touch, such as gentle touch, stroking or kangaroo care, showed similar health benefits.
While it seems to be less critical which touch intervention is applied, the frequency of interventions seems to matter. More sessions were positively associated with the improvement of trait outcomes such as depression and anxiety but also pain reductions in adults. In contrast to session number, increasing the duration of individual sessions did not improve health effects. In fact, we found some indications of negative relationships in adults for cortisol and blood pressure. This could be due to habituating effects of touch on the sympathetic nervous system and hypothalamic–pituitary–adrenal axis, ultimately resulting in diminished effects with longer exposure, or decreased pleasantness ratings of affective touch with increasing duration16. For newborns, we could not support previous notions that the duration of the touch intervention is linked to benefits in weight gain17. Thus, an ideal intervention protocol does not seem to have to be excessively long. It should be noted that very few interventions lasted less than 5 min, and it therefore remains unclear whether very short interventions have the same effect.
A critical issue highlighted in the pandemic was the lack of touch due to social restrictions18. To accommodate the need for touch in individuals with small social networks (for example, institutionalized or isolated individuals), touch interventions using objects/robots have been explored in the past (for a review, see ref. 11). We show here that touch interactions outside of the human–human domain are beneficial for mental and physical health outcomes. Importantly, object/robot touch was not as effective in improving mental health as human-applied touch. A sub-analysis of missing skin-to-skin contact among humans indicated that mental health effects of touch might be mediated by the presence of skin-to-skin contact. Thus, it seems profitable to include skin-to-skin contact in future touch interventions, in line with previous findings in newborns19. In robots, recent advancements in synthetic skin20 should be investigated further in this regard. It should be noted that, although we did not observe significant differences in physical health benefits between human–human and human–object touch, the variability of effect sizes was higher in human–object touch. The conditions enabling object or robot interactions to improve well-being should therefore be explored in more detail in the future.
Touch was beneficial for both healthy and clinical cohorts. These data are critical as most previous meta-analytic research has focused on individuals diagnosed with clinical disorders (for example, ref. 6). For mental health outcomes, we found larger effects in clinical cohorts. A possible reason could relate to increased touch wanting21 in patients. For example, loneliness often co-occurs with chronic illnesses22, which are linked to depressed mood and feelings of anxiety23. Touch can be used to counteract this negative development24,25. In adults and children, knowing the toucher did not influence health benefits. In contrast, familiarity affected overall health benefits in newborns, with parental touch being more beneficial than touch applied by medical staff. Previous studies have suggested that early skin-to-skin contact and exposure to maternal odour is critical for a newborn’s ability to adapt to a new environment26, supporting the notion that parental care is difficult to substitute in this time period.
With respect to age-related effects, our data further suggest that increasing age was associated with a higher benefit through touch for systolic blood pressure. These findings could potentially be attributed to higher basal blood pressure27 with increasing age, allowing for a stronger modulation of this parameter. For sex differences, our study provides some evidence that there are differences between women and men with respect to health benefits of touch. Overall, research on sex differences in touch processing is relatively sparse (but see refs. 28,29). Our results suggest that buffering effects against physiological stress are stronger in women. This is in line with increased buffering effects of hugs in women compared with men30. The female-biased primary research in adults, however, begs for more research in men or non-binary individuals. Unfortunately, our study could not dive deeper into this topic as health benefits broken down by sex or gender were almost never provided. Recent research has demonstrated that sensory pleasantness is affected by sex and that this also interacts with the familiarity of the other person in the touching dyad29,31. In general, contextual factors such as sex and gender or the relationship of the touching dyad, differences in cultural background or internal states such as stress have been demonstrated to be highly influential in the perception of affective touch and are thus relevant to maximizing the pleasantness and ultimately the health benefits of touch interactions32,33,34. As a positive personal relationship within the touching dyad is paramount to induce positive health effects, future research applying robot touch to promote well-being should therefore not only explore synthetic skin options but also focus on improving robots as social agents that form a close relationship with the person receiving the touch35.
As part of the systematic review, we also assessed the effects of touch interventions in non-human animals. Mimicking the results of the meta-analysis in humans, beneficial effects of touch in animals were comparably strong for mental health-like and physical health outcomes. This may inform interventions to promote animal welfare in the context of animal experiments36, farming37 and pets38. While most studies investigated effects in rodents, which are mostly used as laboratory animals, these results probably transfer to livestock and common pets as well. Indeed, touch was beneficial in lambs, fish and cats39,40,41. The positive impact of human touch in rodents also allows for future mechanistic studies in animal models to investigate how interventions such as tickling or stroking modulate hormonal and neuronal responses to touch in the brain. Furthermore, the commonly proposed oxytocin hypothesis can be causally investigated in these animal models through, for example, optogenetic or chemogenetic techniques42. We believe that such translational approaches will further help in optimizing future interventions in humans by uncovering the underlying mechanisms and brain circuits involved in touch.
Our results offer many promising avenues to improve future touch interventions, but they also need to be discussed in light of their limitations. While the majority of findings showed robust health benefits of touch interventions across moderators when compared with a null effect, post hoc tests of, for example, familiarity effects in newborns or mental health benefit differences between human and object touch only barely reached significance. Since we computed a large number of statistical tests in the present study, there is a risk that these results are false positives. We hope that researchers in this field are stimulated by these intriguing results and target these questions by primary research through controlled experimental designs within a well-powered study. Furthermore, the presence of small-study bias in both meta-analyses is indicative that the effect size estimates presented here might be overestimated as null results are often unpublished. We want to stress however that this bias is probably reduced by the multivariate reporting of primary studies. Most studies that reported on multiple health outcomes only showed significant findings for one or two among many. Thus, the multivariate nature of primary research in this field allowed us to include many non-significant findings in the present study. Another limitation pertains to the fact that we only included articles in languages mostly spoken in Western countries. As a large body of evidence comes from Asian countries, it could be that primary research was published in languages other than specified in the inclusion criteria. Thus, despite the large and inclusive nature of our study, some studies could have been missed regardless. Another factor that could not be accounted for in our meta-analysis was that an important prerequisite for touch to be beneficial is its perceived pleasantness. The level of pleasantness associated with being touched is modulated by several parameters34 including cultural acceptability43, perceived humanness44 or a need for touch45, which could explain the observed differences for certain moderators, such as human–human versus robot–human interaction. Moreover, the fact that secondary categorical moderators could not be investigated with respect to specific health outcomes, owing to the lack of data points, limits the specificity of our conclusions in this regard. It thus remains unclear whether, for example, a decreased mental health benefit in the absence of skin-to-skin contact is linked mostly to decreased anxiolytic effects, changes in positive/negative affect or something else. Since these health outcomes are however highly correlated46, it is likely that such effects are driven by multiple health outcomes. Similarly, it is important to note that our conclusions mainly refer to outcomes measured close to the touch intervention as we did not include long-term outcomes. Finally, it needs to be noted that blinding towards the experimental condition is essentially impossible in touch interventions. Although we compared the touch intervention with other interventions, such as relaxation therapy, as control whenever possible, contributions of placebo effects cannot be ruled out.
In conclusion, we show clear evidence that touch interventions are beneficial across a large number of both physical and mental health outcomes, for both healthy and clinical cohorts, and for all ages. These benefits, while influenced in their magnitude by study cohorts and intervention characteristics, were robustly present, promoting the conclusion that touch interventions can be systematically employed across the population to preserve and improve our health.
Methods
Open science practices
All data and code are accessible in the corresponding OSF project12. The systematic review was registered on PROSPERO (CRD42022304281) before the start of data collection. We deviated from the pre-registered plan as follows:
Deviation 1: During our initial screening for the systematic review, we were confronted with a large number of potential health outcomes to look at. This observation of multivariate outcomes led us to register an amendment during data collection (but before any effect size or moderator screening). In doing so, we aimed to additionally extract meta-analytic effects for a more quantitative assessment of our review question that can account for multivariate data reporting and dependencies of effects within the same study. Furthermore, as we noted a severe lack of studies with respect to health outcomes for animals during the inclusion assessment for the systematic review, we decided that the meta-analysis would only focus on outcomes that could be meaningfully analysed on the meta-analytic level and therefore only included health outcomes of human participants.
Deviation 2: In the pre-registration, we did not explicitly exclude non-randomized trials. Since an explicit use of non-randomization for group allocation significantly increases the risk of bias, we decided to exclude them a posteriori from data analysis.
Deviation 3: In the pre-registration, we outlined a tertiary moderator level, namely benefits of touch application versus touch reception. This level was ignored since no included study specifically investigated the benefits of touch application by itself.
Deviation 4: In the pre-registration, we suggested using the RoBMA function47 to provide a Bayesian framework that allows for a more accurate assessment of publication bias beyond small-study bias. Unfortunately, neither multilevel nor multivariate data structures are supported by the RoBMA function, to our knowledge. For this reason, we did not further pursue this analysis, as the hierarchical nature of the data would not be accounted for.
Deviation 5: Beyond the pre-registered inclusion and exclusion criteria, we also excluded dissertations owing to their lack of peer review.
Deviation 6: In the pre-registration, we stated to investigate the impact of sex of the person applying the touch. This moderator was not further analysed, as this information was rarely given and the individuals applying the touch were almost exclusively women (7 males, 24 mixed and 85 females in studies on adults/children; 3 males, 17 mixed and 80 females in studied on newborns).
Deviation 7: The time span of the touch intervention as assessed by subtracting the final day of the intervention from the first day was not investigated further owing to its very high correlation with the number of sessions (r(461) = 0.81 in the adult meta-analysis, r(145) = 0.84 in the newborn meta-analysis).
Inclusion and exclusion criteria
To be included in the systematic review, studies had to investigate the relationship between at least one health outcome (physical and/or mental) in humans or animals and a touch intervention, include explicit physical touch by another human, animal or object as part of an intervention and include an experimental and control condition/group that are differentiated by touch alone. Of note, as a result of this selection process, no animal-to-animal touch intervention study was included, as they never featured a proper no-touch control. Human touch was always explicit touch by a human (that is, no brushes or other tools), either with or without skin-to-skin contact. Regarding the included health outcomes, we aimed to be as broad as possible but excluded parameters such as neurophysiological responses or pleasantness ratings after touch application as they do not reflect health outcomes. All included studies in the meta-analysis and systematic review48,49,50,51,52,53,54,55,56,57,58,59,60,61,62,63,64,65,66,67,68,69,70,71,72,73,74,75,76,77,78,79,80,81,82,83,84,85,86,87,88,89,90,91,92,93,94,95,96,97,98,99,100,101,102,103,104,105,106,107,108,109,110,111,112,113,114,115,116,117,118,119,120,121,122,123,124,125,126,127,128,129,130,131,132,133,134,135,136,137,138,139,140,141,142,143,144,145,146,147,148,149,150,151,152,153,154,155,156,157,158,159,160,161,162,163,164,165,166,167,168,169,170,171,172,173,174,175,176,177,178,179,180,181,182,183,184,185,186,187,188,189,190,191,192,193,194,195,196,197,198,199,200,201,202,203,204,205,206,207,208,209,210,211,212,213,214,215,216,217,218,219,220,221,222,223,224,225,226,227,228,229,230,231,232,233,234,235,236,237,238,239,240,241,242,243,244,245,246,247,248,249,250,251,252,253,254,255,256,257,258,259,260,261,262,263 are listed in Supplementary Table 2. All excluded studies are listed in Supplementary Table 3, together with a reason for exclusion. We then applied a two-step process: First, we identified all potential health outcomes and extracted qualitative information on those outcomes (for example, direction of effect). Second, we extracted quantitative information from all possible outcomes (for example, effect sizes). The meta-analysis additionally required a between-subjects design (to clearly distinguish touch from no-touch effects and owing to missing information about the correlation between repeated measurements264). Studies that explicitly did not apply a randomized protocol were excluded before further analysis to reduce risk of bias. The full study lists for excluded and included studies can be found in the OSF project12 in the file ‘Study_lists_final_revised.xlsx’. In terms of the time frame, we conducted an open-start search of studies until 2022 and identified studies conducted between 1965 and 2022.
Data collection
We used Google Scholar, PubMed and Web of Science for our literature search, with no limitations regarding the publication date and using pre-specified search queries (see Supplementary Information for the exact keywords used). All procedures were in accordance with the updated Preferred Reporting Items for Systematic Reviews and Meta-Analyses guidelines265. Articles were assessed in French, Dutch, German or English. The above databases were searched from 2 December 2021 until 1 October 2022. Two independent coders evaluated each paper against the inclusion and exclusion criteria. Inconsistencies between coders were checked and resolved by J.P. and H.H. Studies excluded/included for the review and meta-analysis can be found on the OSF project.
Search queries
We used the following keywords to search the chosen databases. Agents (human versus animal versus object versus robot) and touch outcome (physical versus mental) were searched separately together with keywords searching for touch.
-
1.
TOUCH: Touch OR Social OR Affective OR Contact OR Tactile interaction OR Hug OR Massage OR Embrace OR Kiss OR Cradling OR Stroking OR Haptic interaction OR tickling
-
2.
AGENT: Object OR Robot OR human OR animal OR rodent OR primate
-
3.
MENTAL OUTCOME: Health OR mood OR Depression OR Loneliness OR happiness OR life satisfaction OR Mental Disorder OR well-being OR welfare OR dementia OR psychological OR psychiatric OR anxiety OR Distress
-
4.
PHYSICAL OUTCOME: Health OR Stress OR Pain OR cardiovascular health OR infection risk OR immune response OR blood pressure OR heart rate
Data extraction and preparation
Data extraction began on 10 October 2022 and was concluded on 25 February 2023. J.P. and H.H. oversaw the data collection process, and checked and resolved all inconsistencies between coders.
Health benefits of touch were always coded by positive summary effects, whereas adverse health effects of touch were represented by negative summary effects. If multiple time points were measured for the same outcome on the same day after a single touch intervention, we extracted the peak effect size (in either the positive or negative direction). If the touch intervention occurred multiple times and health outcomes were assessed for each time point, we extracted data points separately. However, we only extracted immediate effects, as long-term effects not controlled through the experimental conditions could be due to influences other than the initial touch intervention. Measurements assessing long-term effects without explicit touch sessions in the breaks were excluded for the same reason. Common control groups for touch interventions comprised active (for example, relaxation therapy) as well as passive control groups (for example, standard medical care). In the case of multiple control groups, we always contrasted the touch group to the group that most closely matched the touch condition (for example, relaxation therapy was preferred over standard medical care). We extracted information from all moderators listed in the pre-registration (Supplementary Table 4). A list of included and excluded health outcomes is presented in Supplementary Table 5. Authors of studies with possible effects but missing information to calculate those effects were contacted via email and asked to provide the missing data (response rate 35.7%).
After finalizing the list of included studies for the systematic review, we added columns for moderators and the coding schema for our meta-analysis per our updated registration. Then, each study was assessed for its eligibility in the meta-analysis by two independent coders (J.P., H.H., K.F. or F.M.). To this end, all coders followed an a priori specified procedure: First, the PDF was skimmed for possible effects to extract, and the study was excluded if no PDF was available or the study was in a language different from the ones specified in ‘Data collection’. Effects from studies that met the inclusion criteria were extracted from all studies listing descriptive values or statistical parameters to calculate effect sizes. A website266 was used to convert descriptive and statistical values available in the included studies (means and standard deviations/standard errors/confidence intervals, sample sizes, F values, t values, t test P values or frequencies) into Cohen’s d, which were then converted in Hedges’ g. If only P value thresholds were reported (for example, P < 0.01), we used this, most conservative, value as the P value to calculate the effect size (for example, P = 0.01). If only the total sample size was given but that number was even and the participants were randomly assigned to each group, we assumed equal sample sizes for each group. If delta change scores (for example, pre- to post-touch intervention) were reported, we used those over post-touch only scores. In case frequencies were 0 when frequency tables were used to determine effect sizes, we used a value of 0.5 as a substitute to calculate the effect (the default setting in the ‘metafor’ function267). From these data, Hedges’ g and its variance could be derived. Effect sizes were always computed between the experimental and the control group.
Statistical analysis and risk of bias assessment
Owing to the lack of identified studies, health benefits to animals were not included as part of the statistical analysis. One meta-analysis was performed for adults, adolescents and children, as outcomes were highly comparable. We refer to this meta-analysis as the adult meta-analysis, as children/adolescent cohorts were only targeted in a minority of studies. A separate meta-analysis was performed for newborns, as their health outcomes differed substantially from any other age group.
Data were analysed using R (version 4.2.2) with the ‘rma.mv’ function from the ‘metafor’ package267 in a multistep, multivariate and multilevel fashion.
We calculated an overall effect of touch interventions across all studies, cohorts and health outcomes. To account for the hierarchical structure of the data, we used a multilevel structure with random effects at the study, cohort and effects level. Furthermore, we calculated the variance–covariance matrix of all data points to account for the dependencies of measured effects within each individual cohort and study. The variance–covariance matrix was calculated by default with an assumed correlation of effect sizes within each cohort of ρ = 0.6. As ρ needed to be assumed, sensitivity analyses for all computed effect estimates were conducted using correlations between effects of 0, 0.2, 0.4 and 0.8. The results of these sensitivity analyses can be found in ref. 12. No conclusion drawn in the present manuscript was altered by changing the level of ρ. The sensitivity analyses, however, showed that higher assumed correlations lead to more conservative effect size estimates (see Supplementary Figs. 19 and 20 for the adult and newborn meta-analyses, respectively), reducing the type I error risk in general268. In addition to these procedures, we used robust variance estimation with cluster-robust inference at the cohort level. This step is recommended to more accurately determine the confidence intervals in complex multivariate models269. The data distribution was assumed to be normal, but this was not formally tested.
To determine whether individual effects had a strong influence on our results, we calculated Cook’s distance D. Here, a threshold of D > 0.5 was used to qualify a study as influential270. Heterogeneity in the present study was assessed using Cochran’s Q, which determines whether the extracted effect sizes estimate a common population effect size. Although the Q statistic in the ‘rma.mv’ function accounts for the hierarchical nature of the data, we also quantified the heterogeneity estimator σ² for each random-effects level to provide a comprehensive overview of heterogeneity indicators. These indicators for all models can be found on the OSF project12 in the Table ‘Model estimates’. To assess small study bias, we visually inspected the funnel plot and used the standard error as a moderator in the overarching meta-analyses.
Before any sub-group analysis, the overall effect size was used as input for power calculations. While such post hoc power calculations might be limited, we believe that a minimum number of effects to be included in subgroup analyses was necessary to allow for meaningful conclusions. Such medium effect sizes would also probably be the minimum effect sizes of interest for researchers as well as clinical practitioners. Power calculation for random-effects models further requires a sample size for each individual effect as well as an approximation of the expected heterogeneity between studies. For the sample size input, we used the median sample size in each of our studies. For heterogeneity, we assumed a value between medium and high levels of heterogeneity (I² = 62.5%271), as moderator analyses typically aim at reducing heterogeneity overall. Subgroups were only further investigated if the number of observed effects achieved ~80% power under these circumstances, to allow for a more robust interpretation of the observed effects (see Supplementary Figs. 5 and 6 for the adult and newborn meta-analysis, respectively). In a next step, we investigated all pre-registered moderators for which sufficient power was detected. We first looked at our primary moderators (mental versus physical health) and how the effect sizes systematically varied as a function of our secondary moderators (for example, human–human or human–object touch, duration, skin-to-skin presence, etc.). We always included random slopes to allow for our moderators to vary with the random effects at our clustering variable, which is recommended in multilevel models to reduce false positives272. All statistical tests were performed two-sided. Significance of moderators was determined using omnibus F tests. Effect size differences between moderator levels and their confidence intervals were assessed via t tests.
Post hoc t tests were performed comparing mental and physical health benefits within each interacting moderator (for example, mental versus physical health benefits in cancer patients) and mental or physical health benefits across levels of the interacting moderator (for example, mental health benefits in cancer versus pain patients). The post hoc tests were not pre-registered. Data were visualized using forest plots and orchard plots273 for categorical moderators and scatter plots for continuous moderators.
For a broad overview of prior work and their biases, risk of bias was assessed for all studies included in both meta-analyses and the systematic review. We assessed the risk of bias for the following parameters:
-
(1)
Bias from randomization, including whether a randomization procedure was performed, whether it was a between- or within-participant design and whether there were any baseline differences for demographic or dependent variables.
-
(2)
Sequence bias resulting from a lack of counterbalancing in within-subject designs.
-
(3)
Performance bias resulting from the participants or experiments not being blinded to the experimental conditions.
-
(4)
Attrition bias resulting from different dropout rates between experimental groups.
Note that four studies in the adult meta-analysis did not explicitly mention randomization as part of their protocol. However, since these studies never showed any baseline differences in all relevant variables (see ‘Risk of Bias’ table on the OSF project), we assumed that randomization was performed but not mentioned. Sequence bias was of no concern for studies for the meta-analysis since cross-over designs were excluded. It was, however, assessed for studies within the scope of the systematic review. Importantly, performance bias was always high in the adult/children meta-analysis, as blinding of the participants and experimenters to the experimental conditions was not possible owing to the nature of the intervention (touch versus no touch). For studies with newborns and animals, we assessed the performance bias as medium since neither newborns or animals are likely to be aware of being part of an experiment or specific group. An overview of the results is presented in Supplementary Fig. 21, and the precise assessment for each study can be found on the OSF project12 in the ‘Risk of Bias’ table.
Reporting summary
Further information on research design is available in the Nature Portfolio Reporting Summary linked to this article.
Data availability
All data are available via Open Science Framework at https://doi.org/10.17605/OSF.IO/C8RVW (ref. 12). Source data are provided with this paper.
Code availability
All code is available via Open Science Framework at https://doi.org/10.17605/OSF.IO/C8RVW (ref. 12).
References
Fulkerson, M. The First Sense: a Philosophical Study of Human Touch (MIT Press, 2013).
Farroni, T., Della Longa, L. & Valori, I. The self-regulatory affective touch: a speculative framework for the development of executive functioning. Curr. Opin. Behav. Sci. 43, 167–173 (2022).
Ocklenburg, S. et al. Hugs and kisses—the role of motor preferences and emotional lateralization for hemispheric asymmetries in human social touch. Neurosci. Biobehav. Rev. 95, 353–360 (2018).
Ardiel, E. L. & Rankin, C. H. The importance of touch in development. Paediatr. Child Health 15, 153–156 (2010).
Moyer, C. A., Rounds, J. & Hannum, J. W. A meta-analysis of massage therapy research. Psychol. Bull. 130, 3–18 (2004).
Lee, S. H., Kim, J. Y., Yeo, S., Kim, S. H. & Lim, S. Meta-analysis of massage therapy on cancer pain. Integr. Cancer Ther. 14, 297–304 (2015).
LaFollette, M. R., O’Haire, M. E., Cloutier, S. & Gaskill, B. N. A happier rat pack: the impacts of tickling pet store rats on human–animal interactions and rat welfare. Appl. Anim. Behav. Sci. 203, 92–102 (2018).
Packheiser, J., Michon, F. Eva, C., Fredriksen, K. & Hartmann H. The physical and mental health benefits of social touch: a comparative systematic review and meta-analysis. PROSPERO https://www.crd.york.ac.uk/prospero/display_record.php?ID=CRD42022304281 (2023).
Lakens, D. Sample size justification. Collabra. Psychol. 8, 33267 (2022).
Quintana, D. S. A guide for calculating study-level statistical power for meta-analyses. Adv. Meth. Pract. Psychol. Sci. https://doi.org/10.1177/25152459221147260 (2023).
Eckstein, M., Mamaev, I., Ditzen, B. & Sailer, U. Calming effects of touch in human, animal, and robotic interaction—scientific state-of-the-art and technical advances. Front. Psychiatry 11, 555058 (2020).
Packheiser, J. et al. The physical and mental health benefits of affective touch: a comparative systematic review and multivariate meta-analysis. Open Science Framework https://doi.org/10.17605/OSF.IO/C8RVW (2023).
Kong, L. J. et al. Massage therapy for neck and shoulder pain: a systematic review and meta-analysis. Evid. Based Complement. Altern. Med. 2013, 613279 (2013).
Wang, L., He, J. L. & Zhang, X. H. The efficacy of massage on preterm infants: a meta-analysis. Am. J. Perinatol. 30, 731–738 (2013).
Field, T. Massage therapy research review. Complement. Ther. Clin. Pract. 24, 19–31 (2016).
Bendas, J., Ree, A., Pabel, L., Sailer, U. & Croy, I. Dynamics of affective habituation to touch differ on the group and individual level. Neuroscience 464, 44–52 (2021).
Charpak, N., Montealegre‐Pomar, A. & Bohorquez, A. Systematic review and meta‐analysis suggest that the duration of Kangaroo mother care has a direct impact on neonatal growth. Acta Paediatr. 110, 45–59 (2021).
Packheiser, J. et al. A comparison of hugging frequency and its association with momentary mood before and during COVID-19 using ecological momentary assessment. Health Commun. https://doi.org/10.1080/10410236.2023.2198058 (2023).
Whitelaw, A., Heisterkamp, G., Sleath, K., Acolet, D. & Richards, M. Skin to skin contact for very low birthweight infants and their mothers. Arch. Dis. Child. 63, 1377–1381 (1988).
Yogeswaran, N. et al. New materials and advances in making electronic skin for interactive robots. Adv. Robot. 29, 1359–1373 (2015).
Durkin, J., Jackson, D. & Usher, K. Touch in times of COVID‐19: touch hunger hurts. J. Clin. Nurs. https://doi.org/10.1111/jocn.15488 (2021).
Rokach, A., Lechcier-Kimel, R. & Safarov, A. Loneliness of people with physical disabilities. Soc. Behav. Personal. Int. J. 34, 681–700 (2006).
Palgi, Y. et al. The loneliness pandemic: loneliness and other concomitants of depression, anxiety and their comorbidity during the COVID-19 outbreak. J. Affect. Disord. 275, 109–111 (2020).
Heatley-Tejada, A., Dunbar, R. I. M. & Montero, M. Physical contact and loneliness: being touched reduces perceptions of loneliness. Adapt. Hum. Behav. Physiol. 6, 292–306 (2020).
Packheiser, J. et al. The association of embracing with daily mood and general life satisfaction: an ecological momentary assessment study. J. Nonverbal Behav. 46, 519–536 (2022).
Porter, R. The biological significance of skin-to-skin contact and maternal odours. Acta Paediatr. 93, 1560–1562 (2007).
Hawkley, L. C., Masi, C. M., Berry, J. D. & Cacioppo, J. T. Loneliness is a unique predictor of age-related differences in systolic blood pressure. Psychol. Aging 21, 152–164 (2006).
Russo, V., Ottaviani, C. & Spitoni, G. F. Affective touch: a meta-analysis on sex differences. Neurosci. Biobehav. Rev. 108, 445–452 (2020).
Schirmer, A. et al. Understanding sex differences in affective touch: sensory pleasantness, social comfort, and precursive experiences. Physiol. Behav. 250, 113797 (2022).
Berretz, G. et al. Romantic partner embraces reduce cortisol release after acute stress induction in women but not in men. PLoS ONE 17, e0266887 (2022).
Gazzola, V. et al. Primary somatosensory cortex discriminates affective significance in social touch. Proc. Natl Acad. Sci. USA 109, E1657–E1666 (2012).
Sorokowska, A. et al. Affective interpersonal touch in close relationships: a cross-cultural perspective. Personal. Soc. Psychol. Bull. 47, 1705–1721 (2021).
Ravaja, N., Harjunen, V., Ahmed, I., Jacucci, G. & Spapé, M. M. Feeling touched: emotional modulation of somatosensory potentials to interpersonal touch. Sci. Rep. 7, 40504 (2017).
Saarinen, A., Harjunen, V., Jasinskaja-Lahti, I., Jääskeläinen, I. P. & Ravaja, N. Social touch experience in different contexts: a review. Neurosci. Biobehav. Rev. 131, 360–372 (2021).
Huisman, G. Social touch technology: a survey of haptic technology for social touch. IEEE Trans. Haptics 10, 391–408 (2017).
Lewejohann, L., Schwabe, K., Häger, C. & Jirkof, P. Impulse for animal welfare outside the experiment. Lab. Anim. https://doi.org/10.17169/REFUBIUM-26765 (2020).
Sørensen, J. T., Sandøe, P. & Halberg, N. Animal welfare as one among several values to be considered at farm level: the idea of an ethical account for livestock farming. Acta Agric. Scand. A 51, 11–16 (2001).
Verga, M. & Michelazzi, M. Companion animal welfare and possible implications on the human–pet relationship. Ital. J. Anim. Sci. 8, 231–240 (2009).
Coulon, M. et al. Do lambs perceive regular human stroking as pleasant? Behavior and heart rate variability analyses. PLoS ONE 10, e0118617 (2015).
Soares, M. C., Oliveira, R. F., Ros, A. F. H., Grutter, A. S. & Bshary, R. Tactile stimulation lowers stress in fish. Nat. Commun. 2, 534 (2011).
Gourkow, N., Hamon, S. C. & Phillips, C. J. C. Effect of gentle stroking and vocalization on behaviour, mucosal immunity and upper respiratory disease in anxious shelter cats. Prev. Vet. Med. 117, 266–275 (2014).
Oliveira, V. E. et al. Oxytocin and vasopressin within the ventral and dorsal lateral septum modulate aggression in female rats. Nat. Commun. 12, 2900 (2021).
Burleson, M. H., Roberts, N. A., Coon, D. W. & Soto, J. A. Perceived cultural acceptability and comfort with affectionate touch: differences between Mexican Americans and European Americans. J. Soc. Personal. Relatsh. 36, 1000–1022 (2019).
Wijaya, M. et al. The human ‘feel’ of touch contributes to its perceived pleasantness. J. Exp. Psychol. Hum. Percept. Perform. 46, 155–171 (2020).
Golaya, S. Touch-hunger: an unexplored consequence of the COVID-19 pandemic. Indian J. Psychol. Med. 43, 362–363 (2021).
Ng, T. W. H., Sorensen, K. L., Zhang, Y. & Yim, F. H. K. Anger, anxiety, depression, and negative affect: convergent or divergent? J. Vocat. Behav. 110, 186–202 (2019).
Maier, M., Bartoš, F. & Wagenmakers, E.-J. Robust Bayesian meta-analysis: addressing publication bias with model-averaging. Psychol. Methods 28, 107–122 (2022).
Ahles, T. A. et al. Massage therapy for patients undergoing autologous bone marrow transplantation. J. Pain. Symptom Manag. 18, 157–163 (1999).
Albert, N. M. et al. A randomized trial of massage therapy after heart surgery. Heart Lung 38, 480–490 (2009).
Ang, J. Y. et al. A randomized placebo-controlled trial of massage therapy on the immune system of preterm infants. Pediatrics 130, e1549–e1558 (2012).
Arditi, H., Feldman, R. & Eidelman, A. I. Effects of human contact and vagal regulation on pain reactivity and visual attention in newborns. Dev. Psychobiol. 48, 561–573 (2006).
Arora, J., Kumar, A. & Ramji, S. Effect of oil massage on growth and neurobehavior in very low birth weight preterm neonates. Indian Pediatr. 42, 1092–1100 (2005).
Asadollahi, M., Jabraeili, M., Mahallei, M., Asgari Jafarabadi, M. & Ebrahimi, S. Effects of gentle human touch and field massage on urine cortisol level in premature infants: a randomized, controlled clinical trial. J. Caring Sci. 5, 187–194 (2016).
Basiri-Moghadam, M., Basiri-Moghadam, K., Kianmehr, M. & Jani, S. The effect of massage on neonatal jaundice in stable preterm newborn infants: a randomized controlled trial. J. Pak. Med. Assoc. 65, 602–606 (2015).
Bauer, B. A. et al. Effect of massage therapy on pain, anxiety, and tension after cardiac surgery: a randomized study. Complement. Ther. Clin. Pract. 16, 70–75 (2010).
Beijers, R., Cillessen, L. & Zijlmans, M. A. C. An experimental study on mother-infant skin-to-skin contact in full-terms. Infant Behav. Dev. 43, 58–65 (2016).
Bennett, S. et al. Acute effects of traditional Thai massage on cortisol levels, arterial blood pressure and stress perception in academic stress condition: a single blind randomised controlled trial. J. Bodyw. Mov. Therapies 20, 286–292 (2016).
Bergman, N., Linley, L. & Fawcus, S. Randomized controlled trial of skin-to-skin contact from birth versus conventional incubator for physiological stabilization in 1200- to 2199-gram newborns. Acta Paediatr. 93, 779–785 (2004).
Bigelow, A., Power, M., MacLellan‐Peters, J., Alex, M. & McDonald, C. Effect of mother/infant skin‐to‐skin contact on postpartum depressive symptoms and maternal physiological stress. J. Obstet. Gynecol. Neonatal Nurs. 41, 369–382 (2012).
Billhult, A., Bergbom, I. & Stener-Victorin, E. Massage relieves nausea in women with breast cancer who are undergoing chemotherapy. J. Altern. Complement. Med. 13, 53–57 (2007).
Billhult, A., Lindholm, C., Gunnarsson, R. & Stener-Victorin, E. The effect of massage on cellular immunity, endocrine and psychological factors in women with breast cancer—a randomized controlled clinical trial. Auton. Neurosci. 140, 88–95 (2008).
Braun, L. A. et al. Massage therapy for cardiac surgery patients—a randomized trial. J. Thorac. Cardiovasc. Surg. 144, 1453–1459 (2012).
Cabibihan, J.-J. & Chauhan, S. S. Physiological responses to affective tele-touch during induced emotional stimuli. IEEE Trans. Affect. Comput. 8, 108–118 (2017).
Campeau, M.-P. et al. Impact of massage therapy on anxiety levels in patients undergoing radiation therapy: randomized controlled trial. J. Soc. Integr. Oncol. 5, 133–138 (2007).
Can, Ş. & Kaya, H. The effects of yakson or gentle human touch training given to mothers with preterm babies on attachment levels and the responses of the baby: a randomized controlled trial. Health Care Women Int. 43, 479–498 (2021).
Carfoot, S., Williamson, P. & Dickson, R. A randomised controlled trial in the north of England examining the effects of skin-to-skin care on breast feeding. Midwifery 21, 71–79 (2005).
Castral, T. C., Warnock, F., Leite, A. M., Haas, V. J. & Scochi, C. G. S. The effects of skin-to-skin contact during acute pain in preterm newborns. Eur. J. Pain. 12, 464–471 (2008).
Cattaneo, A. et al. Kangaroo mother care for low birthweight infants: a randomized controlled trial in different settings. Acta Paediatr. 87, 976–985 (1998).
Charpak, N., Ruiz-Peláez, J. G. & Charpak, Y. Rey-Martinez kangaroo mother program: an alternative way of caring for low birth weight infants? One year mortality in a two cohort study. Pediatrics 94, 804–810 (1994).
Chermont, A. G., Falcão, L. F. M., de Souza Silva, E. H. L., de Cássia Xavier Balda, R. & Guinsburg, R. Skin-to-skin contact and/or oral 25% dextrose for procedural pain relief for term newborn infants. Pediatrics 124, e1101–e1107 (2009).
Chi Luong, K., Long Nguyen, T., Huynh Thi, D. H., Carrara, H. P. O. & Bergman, N. J. Newly born low birthweight infants stabilise better in skin-to-skin contact than when separated from their mothers: a randomised controlled trial. Acta Paediatr. 105, 381–390 (2016).
Cho, E.-S. et al. The effects of kangaroo care in the neonatal intensive care unit on the physiological functions of preterm infants, maternal–infant attachment, and maternal stress. J. Pediatr. Nurs. 31, 430–438 (2016).
Choi, H. et al. The effects of massage therapy on physical growth and gastrointestinal function in premature infants: a pilot study. J. Child Health Care 20, 394–404 (2016).
Choudhary, M. et al. To study the effect of Kangaroo mother care on pain response in preterm neonates and to determine the behavioral and physiological responses to painful stimuli in preterm neonates: a study from western Rajasthan. J. Matern. Fetal Neonatal Med. 29, 826–831 (2016).
Christensson, K. et al. Temperature, metabolic adaptation and crying in healthy full-term newborns cared for skin-to-skin or in a cot. Acta Paediatr. 81, 488–493 (1992).
Cloutier, S. & Newberry, R. C. Use of a conditioning technique to reduce stress associated with repeated intra-peritoneal injections in laboratory rats. Appl. Anim. Behav. Sci. 112, 158–173 (2008).
Cloutier, S., Wahl, K., Baker, C. & Newberry, R. C. The social buffering effect of playful handling on responses to repeated intraperitoneal injections in laboratory rats. J. Am. Assoc. Lab. Anim. Sci. 53, 168–173 (2014).
Cloutier, S., Wahl, K. L., Panksepp, J. & Newberry, R. C. Playful handling of laboratory rats is more beneficial when applied before than after routine injections. Appl. Anim. Behav. Sci. 164, 81–90 (2015).
Cong, X. et al. Effects of skin-to-skin contact on autonomic pain responses in preterm infants. J. Pain. 13, 636–645 (2012).
Cong, X., Ludington-Hoe, S. M., McCain, G. & Fu, P. Kangaroo care modifies preterm infant heart rate variability in response to heel stick pain: pilot study. Early Hum. Dev. 85, 561–567 (2009).
Cong, X., Ludington-Hoe, S. M. & Walsh, S. Randomized crossover trial of kangaroo care to reduce biobehavioral pain responses in preterm infants: a pilot study. Biol. Res. Nurs. 13, 204–216 (2011).
Costa, R. et al. Tactile stimulation of adult rats modulates hormonal responses, depression-like behaviors, and memory impairment induced by chronic mild stress: role of angiotensin II. Behav. Brain Res. 379, 112250 (2020).
Cutshall, S. M. et al. Effect of massage therapy on pain, anxiety, and tension in cardiac surgical patients: a pilot study. Complement. Ther. Clin. Pract. 16, 92–95 (2010).
Dalili, H., Sheikhi, S., Shariat, M. & Haghnazarian, E. Effects of baby massage on neonatal jaundice in healthy Iranian infants: a pilot study. Infant Behav. Dev. 42, 22–26 (2016).
Diego, M. A., Field, T. & Hernandez-Reif, M. Vagal activity, gastric motility, and weight gain in massaged preterm neonates. J. Pediatr. 147, 50–55 (2005).
Diego, M. A., Field, T. & Hernandez-Reif, M. Temperature increases in preterm infants during massage therapy. Infant Behav. Dev. 31, 149–152 (2008).
Diego, M. A. et al. Preterm infant massage elicits consistent increases in vagal activity and gastric motility that are associated with greater weight gain. Acta Paediatr. 96, 1588–1591 (2007).
Diego, M. A. et al. Spinal cord patients benefit from massage therapy. Int. J. Neurosci. 112, 133–142 (2002).
Diego, M. A. et al. Aggressive adolescents benefit from massage therapy. Adolescence 37, 597–607 (2002).
Diego, M. A. et al. HIV adolescents show improved immune function following massage therapy. Int. J. Neurosci. 106, 35–45 (2001).
Dieter, J. N. I., Field, T., Hernandez-Reif, M., Emory, E. K. & Redzepi, M. Stable preterm infants gain more weight and sleep less after five days of massage therapy. J. Pediatr. Psychol. 28, 403–411 (2003).
Ditzen, B. et al. Effects of different kinds of couple interaction on cortisol and heart rate responses to stress in women. Psychoneuroendocrinology 32, 565–574 (2007).
Dreisoerner, A. et al. Self-soothing touch and being hugged reduce cortisol responses to stress: a randomized controlled trial on stress, physical touch, and social identity. Compr. Psychoneuroendocrinol. 8, 100091 (2021).
Eaton, M., Mitchell-Bonair, I. L. & Friedmann, E. The effect of touch on nutritional intake of chronic organic brain syndrome patients. J. Gerontol. 41, 611–616 (1986).
Edens, J. L., Larkin, K. T. & Abel, J. L. The effect of social support and physical touch on cardiovascular reactions to mental stress. J. Psychosom. Res. 36, 371–382 (1992).
El-Farrash, R. A. et al. Longer duration of kangaroo care improves neurobehavioral performance and feeding in preterm infants: a randomized controlled trial. Pediatr. Res. 87, 683–688 (2020).
Erlandsson, K., Dsilna, A., Fagerberg, I. & Christensson, K. Skin-to-skin care with the father after cesarean birth and its effect on newborn crying and prefeeding behavior. Birth 34, 105–114 (2007).
Escalona, A., Field, T., Singer-Strunck, R., Cullen, C. & Hartshorn, K. Brief report: improvements in the behavior of children with autism following massage therapy. J. Autism Dev. Disord. 31, 513–516 (2001).
Fattah, M. A. & Hamdy, B. Pulmonary functions of children with asthma improve following massage therapy. J. Altern. Complement. Med. 17, 1065–1068 (2011).
Feldman, R. & Eidelman, A. I. Skin-to-skin contact (kangaroo care) accelerates autonomic and neurobehavioural maturation in preterm infants. Dev. Med. Child Neurol. 45, 274–281 (2003).
Feldman, R., Eidelman, A. I., Sirota, L. & Weller, A. Comparison of skin-to-skin (kangaroo) and traditional care: parenting outcomes and preterm infant development. Pediatrics 110, 16–26 (2002).
Feldman, R., Singer, M. & Zagoory, O. Touch attenuates infants’ physiological reactivity to stress. Dev. Sci. 13, 271–278 (2010).
Feldman, R., Weller, A., Sirota, L. & Eidelman, A. I. Testing a family intervention hypothesis: the contribution of mother–infant skin-to-skin contact (kangaroo care) to family interaction, proximity, and touch. J. Fam. Psychol. 17, 94–107 (2003).
Ferber, S. G. et al. Massage therapy by mothers and trained professionals enhances weight gain in preterm infants. Early Hum. Dev. 67, 37–45 (2002).
Ferber, S. G. & Makhoul, I. R. The effect of skin-to-skin contact (kangaroo care) shortly after birth on the neurobehavioral responses of the term newborn: a randomized, controlled trial. Pediatrics 113, 858–865 (2004).
Ferreira, A. M. & Bergamasco, N. H. P. Behavioral analysis of preterm neonates included in a tactile and kinesthetic stimulation program during hospitalization. Rev. Bras. Fisioter. 14, 141–148 (2010).
Fidanza, F., Polimeni, E., Pierangeli, V. & Martini, M. A better touch: C-tactile fibers related activity is associated to pain reduction during temporal summation of second pain. J. Pain. 22, 567–576 (2021).
Field, T. et al. Leukemia immune changes following massage therapy. J. Bodyw. Mov. Ther. 5, 271–274 (2001).
Field, T. et al. Benefits of combining massage therapy with group interpersonal psychotherapy in prenatally depressed women. J. Bodyw. Mov. Ther. 13, 297–303 (2009).
Field, T., Delage, J. & Hernandez-Reif, M. Movement and massage therapy reduce fibromyalgia pain. J. Bodyw. Mov. Ther. 7, 49–52 (2003).
Field, T. et al. Fibromyalgia pain and substance P decrease and sleep improves after massage therapy. J. Clin. Rheumatol. 8, 72–76 (2002).
Field, T., Diego, M., Gonzalez, G. & Funk, C. G. Neck arthritis pain is reduced and range of motion is increased by massage therapy. Complement. Ther. Clin. Pract. 20, 219–223 (2014).
Field, T., Diego, M., Hernandez-Reif, M., Deeds, O. & Figueiredo, B. Pregnancy massage reduces prematurity, low birthweight and postpartum depression. Infant Behav. Dev. 32, 454–460 (2009).
Field, T. et al. Insulin and insulin-like growth factor-1 increased in preterm neonates following massage therapy. J. Dev. Behav. Pediatr. 29, 463–466 (2008).
Field, T. et al. Yoga and massage therapy reduce prenatal depression and prematurity. J. Bodyw. Mov. Ther. 16, 204–209 (2012).
Field, T., Diego, M., Hernandez-Reif, M., Schanberg, S. & Kuhn, C. Massage therapy effects on depressed pregnant women. J. Psychosom. Obstet. Gynecol. 25, 115–122 (2004).
Field, T., Diego, M., Hernandez-Reif, M. & Shea, J. Hand arthritis pain is reduced by massage therapy. J. Bodyw. Mov. Ther. 11, 21–24 (2007).
Field, T., Gonzalez, G., Diego, M. & Mindell, J. Mothers massaging their newborns with lotion versus no lotion enhances mothers’ and newborns’ sleep. Infant Behav. Dev. 45, 31–37 (2016).
Field, T. et al. Children with asthma have improved pulmonary functions after massage therapy. J. Pediatr. 132, 854–858 (1998).
Field, T., Hernandez-Reif, M., Diego, M. & Fraser, M. Lower back pain and sleep disturbance are reduced following massage therapy. J. Bodyw. Mov. Ther. 11, 141–145 (2007).
Field, T. et al. Effects of sexual abuse are lessened by massage therapy. J. Bodyw. Mov. Ther. 1, 65–69 (1997).
Field, T. et al. Pregnant women benefit from massage therapy. J. Psychosom. Obstet. Gynecol. 20, 31–38 (1999).
Field, T. et al. Juvenilerheumatoid arthritis: benefits from massage therapy. J. Pediatr. Psychol. 22, 607–617 (1997).
Field, T., Hernandez-Reif, M., Taylor, S., Quintino, O. & Burman, I. Labor pain is reduced by massage therapy. J. Psychosom. Obstet. Gynecol. 18, 286–291 (1997).
Field, T. et al. Massage therapy reduces anxiety and enhances EEG pattern of alertness and math computations. Int. J. Neurosci. 86, 197–205 (1996).
Field, T. et al. Brief report: autistic children’s attentiveness and responsivity improve after touch therapy. J. Autism Dev. Disord. 27, 333–338 (1997).
Field, T. M. et al. Tactile/kinesthetic stimulation effects on preterm neonates. Pediatrics 77, 654–658 (1986).
Field, T. et al. Massage reduces anxiety in child and adolescent psychiatric patients. J. Am. Acad. Child Adolesc. Psychiatry 31, 125–131 (1992).
Field, T. et al. Burn injuries benefit from massage therapy. J. Burn Care Res. 19, 241–244 (1998).
Filho, F. L. et al. Effect of maternal skin-to-skin contact on decolonization of methicillin-oxacillin-resistant Staphylococcus in neonatal intensive care units: a randomized controlled trial. BMC Pregnancy Childbirth https://doi.org/10.1186/s12884-015-0496-1 (2015).
Forward, J. B., Greuter, N. E., Crisall, S. J. & Lester, H. F. Effect of structured touch and guided imagery for pain and anxiety in elective joint replacement patients—a randomized controlled trial: M-TIJRP. Perm. J. 19, 18–28 (2015).
Fraser, J. & Ross Kerr, J. Psychophysiological effects of back massage on elderly institutionalized patients. J. Adv. Nurs. 18, 238–245 (1993).
Frey Law, L. A. et al. Massage reduces pain perception and hyperalgesia in experimental muscle pain: a randomized, controlled trial. J. Pain. 9, 714–721 (2008).
Gao, H. et al. Effect of repeated kangaroo mother care on repeated procedural pain in preterm infants: a randomized controlled trial. Int. J. Nurs. Stud. 52, 1157–1165 (2015).
Garner, B. et al. Pilot study evaluating the effect of massage therapy on stress, anxiety and aggression in a young adult psychiatric inpatient unit. Aust. N. Z. J. Psychiatry 42, 414–422 (2008).
Gathwala, G., Singh, B. & Singh, J. Effect of kangaroo mother care on physical growth, breastfeeding and its acceptability. Trop. Dr. 40, 199–202 (2010).
Geva, N., Uzefovsky, F. & Levy-Tzedek, S. Touching the social robot PARO reduces pain perception and salivary oxytocin levels. Sci. Rep. 10, 9814 (2020).
Gitau, R. et al. Acute effects of maternal skin-to-skin contact and massage on saliva cortisol in preterm babies. J. Reprod. Infant Psychol. 20, 83–88 (2002).
Givi, M. Durability of effect of massage therapy on blood pressure. Int. J. Prev. Med. 4, 511–516 (2013).
Glover, V., Onozawa, K. & Hodgkinson, A. Benefits of infant massage for mothers with postnatal depression. Semin. Neonatol. 7, 495–500 (2002).
Gonzalez, A. et al. Weight gain in preterm infants following parent-administered vimala massage: a randomized controlled trial. Am. J. Perinatol. 26, 247–252 (2009).
Gray, L., Watt, L. & Blass, E. M. Skin-to-skin contact is analgesic in healthy newborns. Pediatrics 105, e14 (2000).
Grewen, K. M., Anderson, B. J., Girdler, S. S. & Light, K. C. Warm partner contact is related to lower cardiovascular reactivity. Behav. Med. 29, 123–130 (2003).
Groër, M. W., Hill, J., Wilkinson, J. E. & Stuart, A. Effects of separation and separation with supplemental stroking in BALB/c infant mice. Biol. Res. Nurs. 3, 119–131 (2002).
Gürol, A. P., Polat, S. & Nuran Akçay, M. Itching, pain, and anxiety levels are reduced with massage therapy in burned adolescents. J. Burn Care Res. 31, 429–432 (2010).
Haley, S. et al. Tactile/kinesthetic stimulation (TKS) increases tibial speed of sound and urinary osteocalcin (U-MidOC and unOC) in premature infants (29–32 weeks PMA). Bone 51, 661–666 (2012).
Harris, M., Richards, K. C. & Grando, V. T. The effects of slow-stroke back massage on minutes of nighttime sleep in persons with dementia and sleep disturbances in the nursing home: a pilot study. J. Holist. Nurs. 30, 255–263 (2012).
Hart, S. et al. Anorexia nervosa symptoms are reduced by massage therapy. Eat. Disord. 9, 289–299 (2001).
Hattan, J., King, L. & Griffiths, P. The impact of foot massage and guided relaxation following cardiac surgery: a randomized controlled trial. Issues Innov. Nurs. Pract. 37, 199–207 (2002).
Haynes, A. C. et al. A calming hug: design and validation of a tactile aid to ease anxiety. PLoS ONE 17, e0259838 (2022).
Henricson, M., Ersson, A., Määttä, S., Segesten, K. & Berglund, A.-L. The outcome of tactile touch on stress parameters in intensive care: a randomized controlled trial. Complement. Ther. Clin. Pract. 14, 244–254 (2008).
Hernandez-Reif, M., Diego, M. & Field, T. Preterm infants show reduced stress behaviors and activity after 5 days of massage therapy. Infant Behav. Dev. 30, 557–561 (2007).
Hernandez-Reif, M., Dieter, J. N. I., Field, T., Swerdlow, B. & Diego, M. Migraine headaches are reduced by massage therapy. Int. J. Neurosci. 96, 1–11 (1998).
Hernandez-Reif, M. et al. Natural killer cells and lymphocytes increase in women with breast cancer following massage therapy. Int. J. Neurosci. 115, 495–510 (2005).
Hernandez-Reif, M. et al. Children with cystic fibrosis benefit from massage therapy. J. Pediatr. Psychol. 24, 175–181 (1999).
Hernandez-Reif, M., Field, T., Krasnegor, J. & Theakston, H. Lower back pain is reduced and range of motion increased after massage therapy. Int. J. Neurosci. 106, 131–145 (2001).
Hernandez-Reif, M. et al. High blood pressure and associated symptoms were reduced by massage therapy. J. Bodyw. Mov. Ther. 4, 31–38 (2000).
Hernandez-Reif, M. et al. Parkinson’s disease symptoms are differentially affected by massage therapy vs. progressive muscle relaxation: a pilot study. J. Bodyw. Mov. Ther. 6, 177–182 (2002).
Hernandez-Reif, M., Field, T. & Theakston, H. Multiple sclerosis patients benefit from massage therapy. J. Bodyw. Mov. Ther. 2, 168–174 (1998).
Hernandez-Reif, M. et al. Breast cancer patients have improved immune and neuroendocrine functions following massage therapy. J. Psychosom. Res. 57, 45–52 (2004).
Hertenstein, M. J. & Campos, J. J. Emotion regulation via maternal touch. Infancy 2, 549–566 (2001).
Hinchcliffe, J. K., Mendl, M. & Robinson, E. S. J. Rat 50 kHz calls reflect graded tickling-induced positive emotion. Curr. Biol. 30, R1034–R1035 (2020).
Hodgson, N. A. & Andersen, S. The clinical efficacy of reflexology in nursing home residents with dementia. J. Altern. Complement. Med. 14, 269–275 (2008).
Hoffmann, L. & Krämer, N. C. The persuasive power of robot touch. Behavioral and evaluative consequences of non-functional touch from a robot. PLoS ONE 16, e0249554 (2021).
Holst, S., Lund, I., Petersson, M. & Uvnäs-Moberg, K. Massage-like stroking influences plasma levels of gastrointestinal hormones, including insulin, and increases weight gain in male rats. Auton. Neurosci. 120, 73–79 (2005).
Hori, M. et al. Tickling during adolescence alters fear-related and cognitive behaviors in rats after prolonged isolation. Physiol. Behav. 131, 62–67 (2014).
Hori, M. et al. Effects of repeated tickling on conditioned fear and hormonal responses in socially isolated rats. Neurosci. Lett. 536, 85–89 (2013).
Hucklenbruch-Rother, E. et al. Delivery room skin-to-skin contact in preterm infants affects long-term expression of stress response genes. Psychoneuroendocrinology 122, 104883 (2020).
Im, H. & Kim, E. Effect of yakson and gentle human touch versus usual care on urine stress hormones and behaviors in preterm infants: a quasi-experimental study. Int. J. Nurs. Stud. 46, 450–458 (2009).
Jain, S., Kumar, P. & McMillan, D. D. Prior leg massage decreases pain responses to heel stick in preterm babies. J. Paediatr. Child Health 42, 505–508 (2006).
Jane, S.-W. et al. Effects of massage on pain, mood status, relaxation, and sleep in Taiwanese patients with metastatic bone pain: a randomized clinical trial. Pain 152, 2432–2442 (2011).
Johnston, C. C. et al. Kangaroo mother care diminishes pain from heel lance in very preterm neonates: a crossover trial. BMC Pediatr. 8, 13 (2008).
Johnston, C. C. et al. Kangaroo care is effective in diminishing pain response in preterm neonates. Arch. Pediatr. Adolesc. Med. 157, 1084–1088 (2003).
Jung, M. J., Shin, B.-C., Kim, Y.-S., Shin, Y.-I. & Lee, M. S. Is there any difference in the effects of QI therapy (external QIGONG) with and without touching? a pilot study. Int. J. Neurosci. 116, 1055–1064 (2006).
Kapoor, Y. & Orr, R. Effect of therapeutic massage on pain in patients with dementia. Dementia 16, 119–125 (2017).
Karagozoglu, S. & Kahve, E. Effects of back massage on chemotherapy-related fatigue and anxiety: supportive care and therapeutic touch in cancer nursing. Appl. Nurs. Res. 26, 210–217 (2013).
Karbasi, S. A., Golestan, M., Fallah, R., Golshan, M. & Dehghan, Z. Effect of body massage on increase of low birth weight neonates growth parameters: a randomized clinical trial. Iran. J. Reprod. Med. 11, 583–588 (2013).
Kashaninia, Z., Sajedi, F., Rahgozar, M. & Noghabi, F. A. The effect of kangaroo care on behavioral responses to pain of an intramuscular injection in neonates. J. Pediatr. Nurs. 3, 275–280 (2008).
Kelling, C., Pitaro, D. & Rantala, J. Good vibes: The impact of haptic patterns on stress levels. In Proc. 20th International Academic Mindtrek Conference 130–136 (Association for Computing Machinery, 2016).
Khilnani, S., Field, T., Hernandez-Reif, M. & Schanberg, S. Massage therapy improves mood and behavior of students with attention-deficit/hyperactivity disorder. Adolescence 38, 623–638 (2003).
Kianmehr, M. et al. The effect of massage on serum bilirubin levels in term neonates with hyperbilirubinemia undergoing phototherapy. Nautilus 128, 36–41 (2014).
Kim, I.-H., Kim, T.-Y. & Ko, Y.-W. The effect of a scalp massage on stress hormone, blood pressure, and heart rate of healthy female. J. Phys. Ther. Sci. 28, 2703–2707 (2016).
Kim, M. A., Kim, S.-J. & Cho, H. Effects of tactile stimulation by fathers on physiological responses and paternal attachment in infants in the NICU: a pilot study. J. Child Health Care 21, 36–45 (2017).
Kim, M. S., Sook Cho, K., Woo, H.-M. & Kim, J. H. Effects of hand massage on anxiety in cataract surgery using local anesthesia. J. Cataract Refr. Surg. 27, 884–890 (2001).
Koole, S. L., Tjew A Sin, M. & Schneider, I. K. Embodied terror management: interpersonal touch alleviates existential concerns among individuals with low self-esteem. Psychol. Sci. 25, 30–37 (2014).
Krohn, M. et al. Depression, mood, stress, and Th1/Th2 immune balance in primary breast cancer patients undergoing classical massage therapy. Support. Care Cancer 19, 1303–1311 (2011).
Kuhn, C. et al. Tactile-kinesthetic stimulation effects sympathetic and adrenocortical function in preterm infants. J. Pediatr. 119, 434–440 (1991).
Kumar, J. et al. Effect of oil massage on growth in preterm neonates less than 1800 g: a randomized control trial. Indian J. Pediatr. 80, 465–469 (2013).
Lee, H.-K. The effects of infant massage on weight, height, and mother–infant interaction. J. Korean Acad. Nurs. 36, 1331–1339 (2006).
Leivadi, S. et al. Massage therapy and relaxation effects on university dance students. J. Dance Med. Sci. 3, 108–112 (1999).
Lindgren, L. et al. Touch massage: a pilot study of a complex intervention. Nurs. Crit. Care 18, 269–277 (2013).
Lindgren, L. et al. Physiological responses to touch massage in healthy volunteers. Auton. Neurosci. Basic Clin. 158, 105–110 (2010).
Listing, M. et al. Massage therapy reduces physical discomfort and improves mood disturbances in women with breast cancer. Psycho-Oncol. 18, 1290–1299 (2009).
Ludington-Hoe, S. M., Cranston Anderson, G., Swinth, J. Y., Thompson, C. & Hadeed, A. J. Randomized controlled trial of kangaroo care: cardiorespiratory and thermal effects on healthy preterm infants. Neonatal Netw. 23, 39–48 (2004).
Lund, I. et al. Corticotropin releasing factor in urine—a possible biochemical marker of fibromyalgia. Neurosci. Lett. 403, 166–171 (2006).
Ma, Y.-K. et al. Lack of social touch alters anxiety-like and social behaviors in male mice. Stress 25, 134–144 (2022).
Massaro, A. N., Hammad, T. A., Jazzo, B. & Aly, H. Massage with kinesthetic stimulation improves weight gain in preterm infants. J. Perinatol. 29, 352–357 (2009).
Mathai, S., Fernandez, A., Mondkar, J. & Kanbur, W. Effects of tactile-kinesthetic stimulation in preterms–a controlled trial. Indian Pediatr. 38, 1091–1098 (2001).
Matsunaga, M. et al. Profiling of serum proteins influenced by warm partner contact in healthy couples. Neuroenocrinol. Lett. 30, 227–236 (2009).
Mendes, E. W. & Procianoy, R. S. Massage therapy reduces hospital stay and occurrence of late-onset sepsis in very preterm neonates. J. Perinatol. 28, 815–820 (2008).
Mirnia, K., Arshadi Bostanabad, M., Asadollahi, M. & Hamid Razzaghi, M. Paternal skin-to-skin care and its effect on cortisol levels of the infants. Iran. J. Pediatrics 27, e8151 (2017).
Mitchell, A. J., Yates, C., Williams, K. & Hall, R. W. Effects of daily kangaroo care on cardiorespiratory parameters in preterm infants. J. Neonatal-Perinat. Med. 6, 243–249 (2013).
Mitchinson, A. R. et al. Acute postoperative pain management using massage as an adjuvant therapy: a randomized trial. Arch. Surg. 142, 1158–1167 (2007).
Modrcin-Talbott, M. A., Harrison, L. L., Groer, M. W. & Younger, M. S. The biobehavioral effects of gentle human touch on preterm infants. Nurs. Sci. Q. 16, 60–67 (2003).
Mok, E. & Pang Woo, C. The effects of slow-stroke back massage on anxiety and shoulder pain in elderly stroke patients. Complement. Ther. Nurs. Midwifery 10, 209–216 (2004).
Mokaberian, M., Noripour, S., Sheikh, M. & Mills, P. J. Examining the effectiveness of body massage on physical status of premature neonates and their mothers’ psychological status. Early Child Dev. Care 192, 2311–2325 (2021).
Mori, H. et al. Effect of massage on blood flow and muscle fatigue following isometric lumbar exercise. Med. Sci. Monit. Int. Med. J. Exp. Clin. Res. 10, CR173–CR178 (2004).
Moyer-Mileur, L. J., Haley, S., Slater, H., Beachy, J. & Smith, S. L. Massage improves growth quality by decreasing body fat deposition in male preterm infants. J. Pediatr. 162, 490–495 (2013).
Moyle, W. et al. Foot massage and physiological stress in people with dementia: a randomized controlled trial. J. Altern. Complement. Med. 20, 305–311 (2014).
Muntsant, A., Shrivastava, K., Recasens, M. & Giménez-Llort, L. Severe perinatal hypoxic-ischemic brain injury induces long-term sensorimotor deficits, anxiety-like behaviors and cognitive impairment in a sex-, age- and task-selective manner in C57BL/6 mice but can be modulated by neonatal handling. Front. Behav. Neurosci. 13, 7 (2019).
Negahban, H., Rezaie, S. & Goharpey, S. Massage therapy and exercise therapy in patients with multiple sclerosis: a randomized controlled pilot study. Clin. Rehabil. 27, 1126–1136 (2013).
Nelson, D., Heitman, R. & Jennings, C. Effects of tactile stimulation on premature infant weight gain. J. Obstet. Gynecol. Neonatal Nurs. 15, 262–267 (1986).
Griffin, J. W. Calculating statistical power for meta-analysis using metapower. Quant. Meth. Psychol. 17, 24–39 (2021).
Nunes, G. S. et al. Massage therapy decreases pain and perceived fatigue after long-distance Ironman triathlon: a randomised trial. J. Physiother. 62, 83–87 (2016).
Ohgi, S. et al. Comparison of kangaroo care and standard care: behavioral organization, development, and temperament in healthy, low-birth-weight infants through 1 year. J. Perinatol. 22, 374–379 (2002).
O′Higgins, M., St. James Roberts, I. & Glover, V. Postnatal depression and mother and infant outcomes after infant massage. J. Affect. Disord. 109, 189–192 (2008).
Okan, F., Ozdil, A., Bulbul, A., Yapici, Z. & Nuhoglu, A. Analgesic effects of skin-to-skin contact and breastfeeding in procedural pain in healthy term neonates. Ann. Trop. Paediatr. 30, 119–128 (2010).
Oliveira, D. S., Hachul, H., Goto, V., Tufik, S. & Bittencourt, L. R. A. Effect of therapeutic massage on insomnia and climacteric symptoms in postmenopausal women. Climacteric 15, 21–29 (2012).
Olsson, E., Ahlsén, G. & Eriksson, M. Skin-to-skin contact reduces near-infrared spectroscopy pain responses in premature infants during blood sampling. Acta Paediatr. 105, 376–380 (2016).
Pauk, J., Kuhn, C. M., Field, T. M. & Schanberg, S. M. Positive effects of tactile versus kinesthetic or vestibular stimulation on neuroendocrine and ODC activity in maternally-deprived rat pups. Life Sci. 39, 2081–2087 (1986).
Pinazo, D., Arahuete, L. & Correas, N. Hugging as a buffer against distal fear of death. Calid. Vida Salud 13, 11–20 (2020).
Pope, M. H. et al. A prospective randomized three-week trial of spinal manipulation, transcutaneous muscle stimulation, massage and corset in the treatment of subacute low back pain. Spine 19, 2571–2577 (1994).
Preyde, M. Effectiveness of massage therapy for subacute low-back pain: a randomized controlled trial. Can. Med. Assoc. J. 162, 1815–1820 (2000).
Ramanathan, K., Paul, V. K., Deorari, A. K., Taneja, U. & George, G. Kangaroo mother care in very low birth weight infants. Indian J. Pediatr. 68, 1019–1023 (2001).
Reddan, M. C., Young, H., Falkner, J., López-Solà, M. & Wager, T. D. Touch and social support influence interpersonal synchrony and pain. Soc. Cogn. Affect. Neurosci. 15, 1064–1075 (2020).
Rodríguez-Mansilla, J. et al. The effects of ear acupressure, massage therapy and no therapy on symptoms of dementia: a randomized controlled trial. Clin. Rehabil. 29, 683–693 (2015).
Rose, S. A., Schmidt, K., Riese, M. L. & Bridger, W. H. Effects of prematurity and early intervention on responsivity to tactual stimuli: a comparison of preterm and full-term infants. Child Dev. 51, 416–425 (1980).
Scafidi, F. A. et al. Massage stimulates growth in preterm infants: a replication. Infant Behav. Dev. 13, 167–188 (1990).
Scafidi, F. A. et al. Effects of tactile/kinesthetic stimulation on the clinical course and sleep/wake behavior of preterm neonates. Infant Behav. Dev. 9, 91–105 (1986).
Scafidi, F. & Field, T. Massage therapy improves behavior in neonates born to HIV-positive mothers. J. Pediatr. Psychol. 21, 889–897 (1996).
Scarr-Salapatek, S. & Williams, M. L. A stimulation program for low birth weight infants. Am. J. Public Health 62, 662–667 (1972).
Serrano, B., Baños, R. M. & Botella, C. Virtual reality and stimulation of touch and smell for inducing relaxation: a randomized controlled trial. Comput. Hum. Behav. 55, 1–8 (2016).
Seyyedrasooli, A., Valizadeh, L., Hosseini, M. B., Asgari Jafarabadi, M. & Mohammadzad, M. Effect of vimala massage on physiological jaundice in infants: a randomized controlled trial. J. Caring Sci. 3, 165–173 (2014).
Sharpe, P. A., Williams, H. G., Granner, M. L. & Hussey, J. R. A randomised study of the effects of massage therapy compared to guided relaxation on well-being and stress perception among older adults. Complement. Therap. Med. 15, 157–163 (2007).
Sherman, K. J., Cherkin, D. C., Hawkes, R. J., Miglioretti, D. L. & Deyo, R. A. Randomized trial of therapeutic massage for chronic neck pain. Clin. J. Pain. 25, 233–238 (2009).
Shiloh, S., Sorek, G. & Terkel, J. Reduction of state-anxiety by petting animals in a controlled laboratory experiment. Anxiety, Stress Coping 16, 387–395 (2003).
Shor-Posner, G. et al. Impact of a massage therapy clinical trial on immune status in young Dominican children infected with HIV-1. J. Altern. Complement. Med. 12, 511–516 (2006).
Simpson, E. A. et al. Social touch alters newborn monkey behavior. Infant Behav. Dev. 57, 101368 (2019).
Smith, S. L., Haley, S., Slater, H. & Moyer-Mileur, L. J. Heart rate variability during caregiving and sleep after massage therapy in preterm infants. Early Hum. Dev. 89, 525–529 (2013).
Smith, S. L. et al. The effect of massage on heart rate variability in preterm infants. J. Perinatol. 33, 59–64 (2013).
Solkoff, N. & Matuszak, D. Tactile stimulation and behavioral development among low-birthweight infants. Child Psychiatry Hum. Dev. 6, 3337 (1975).
Srivastava, S., Gupta, A., Bhatnagar, A. & Dutta, S. Effect of very early skin to skin contact on success at breastfeeding and preventing early hypothermia in neonates. Indian J. Public Health 58, 22–26 (2014).
Stringer, J., Swindell, R. & Dennis, M. Massage in patients undergoing intensive chemotherapy reduces serum cortisol and prolactin: massage in oncology patients reduces serum cortisol. Psycho-Oncol. 17, 1024–1031 (2008).
Suman Rao, P. N., Udani, R. & Nanavati, R. Kangaroo mother care for low birth weight infants: a randomized controlled trial. Indian Pediatr. 45, 17–23 (2008).
Sumioka, H. et al. A huggable device can reduce the stress of calling an unfamiliar person on the phone for individuals with ASD. PLoS ONE 16, e0254675 (2021).
Sumioka, H., Nakae, A., Kanai, R. & Ishiguro, H. Huggable communication medium decreases cortisol levels. Sci. Rep. 3, 3034 (2013).
Suzuki, M. et al. Physical and psychological effects of 6-week tactile massage on elderly patients with severe dementia. Am. J. Alzheimer’s Dis. Other Dement. 25, 680–686 (2010).
Thomson, L. J. M., Ander, E. E., Menon, U., Lanceley, A. & Chatterjee, H. J. Quantitative evidence for wellbeing benefits from a heritage-in-health intervention with hospital patients. Int. J. Art. Ther. 17, 63–79 (2012).
Triplett, J. L. & Arneson, S. W. The use of verbal and tactile comfort to alleviate distress in young hospitalized children. Res. Nurs. Health 2, 17–23 (1979).
Walach, H., Güthlin, C. & König, M. Efficacy of massage therapy in chronic pain: a pragmatic randomized trial. J. Altern. Complement. Med. 9, 837–846 (2003).
Walker, S. C. et al. C‐low threshold mechanoafferent targeted dynamic touch modulates stress resilience in rats exposed to chronic mild stress. Eur. J. Neurosci. 55, 2925–2938 (2022).
Weinrich, S. P. & Weinrich, M. C. The effect of massage on pain in cancer patients. Appl. Nurs. Res. 3, 140–145 (1990).
Wheeden, A. et al. Massage effects on cocaine-exposed preterm neonates. Dev. Behav. Pediatr. 14, 318–322 (1993).
White, J. L. & Labarba, R. C. The effects of tactile and kinesthetic stimulation on neonatal development in the premature infant. Dev. Psychobiol. 9, 569–577 (1976).
Wilkie, D. J. et al. Effects of massage on pain intensity, analgesics and quality of life in patients with cancer pain: a pilot study of a randomized clinical trial conducted within hospice care delivery. Hosp. J. 15, 31–53 (2000).
Willemse, C. J. A. M., Toet, A. & van Erp, J. B. F. Affective and behavioral responses to robot-initiated social touch: toward understanding the opportunities and limitations of physical contact in human–robot interaction. Front. ICT 4, 12 (2017).
Willemse, C. J. A. M. & van Erp, J. B. F. Social touch in human–robot interaction: robot-initiated touches can induce positive responses without extensive prior bonding. Int. J. Soc. Robot. 11, 285–304 (2019).
Woods, D. L., Beck, C. & Sinha, K. The effect of therapeutic touch on behavioral symptoms and cortisol in persons with dementia. Res. Complement. Med. 16, 181–189 (2009).
Yamaguchi, M., Sekine, T. & Shetty, V. A salivary cytokine panel discriminates moods states following a touch massage intervention. Int. J. Affect. Eng. 19, 189–198 (2020).
Yamazaki, R. et al. Intimacy in phone conversations: anxiety reduction for Danish seniors with hugvie. Front. Psychol. 7, 537 (2016).
Yang, M.-H. et al. Comparison of the efficacy of aroma-acupressure and aromatherapy for the treatment of dementia-associated agitation. BMC Complement. Altern. Med. 15, 93 (2015).
Yates, C. C. et al. The effects of massage therapy to induce sleep in infants born preterm. Pediatr. Phys. Ther. 26, 405–410 (2014).
Yu, H. et al. Social touch-like tactile stimulation activates a tachykinin 1-oxytocin pathway to promote social interactions. Neuron 110, 1051–1067 (2022).
Lakens, D. Calculating and reporting effect sizes to facilitate cumulative science: a practical primer for t-tests and ANOVAs. Front. Psychol. 4, 863 (2013).
Page, M. J., et al. The PRISMA 2020 statement: an updated guideline for reporting systematic reviews. Syst. Rev. https://doi.org/10.1186/s13643-021-01626-4 (2021).
Wilson, D. B. Practical meta-analysis effect size calculator (Version 2023.11.27). https://campbellcollaboration.org/research-resources/effect-size-calculator.html (2023).
Viechtbauer, W. Conducting meta-analyses in R with the metafor package. J. Stat. Softw https://doi.org/10.18637/jss.v036.i03 (2010).
Scammacca, N., Roberts, G. & Stuebing, K. K. Meta-analysis with complex research designs: dealing with dependence from multiple measures and multiple group comparisons. Rev. Educ. Res. 84, 328–364 (2014).
Pustejovsky, J. E. & Tipton, E. Meta-analysis with robust variance estimation: expanding the range of working models. Prev. Sci. Off. J. Soc. Prev. Res. 23, 425–438 (2022).
Cook, R. D. in International Encyclopedia of Statistical Science (ed. M. Lovric) S. 301–302 (Springer, 2011).
Higgins, J. P. T., Thompson, S. & Deeks, J. Measuring inconsistency in meta-analyses. BMJ https://doi.org/10.1136/bmj.327.7414.557 (2003).
Oberauer, K. The importance of random slopes in mixed models for Bayesian hypothesis testing. Psychol. Sci. 33, 648–665 (2022).
Nakagawa, S. et al. The orchard plot: cultivating a forest plot for use in ecology, evolution, and beyond. Res. Synth. Methods 12, 4–12 (2021).
Acknowledgements
We thank A. Frick and E. Chris for supporting the initial literature search and coding. We also thank A. Dreisoerner, T. Field, S. Koole, C. Kuhn, M. Henricson, L. Frey Law, J. Fraser, M. Cumella Reddan, and J. Stringer, who kindly responded to our data requests and provided additional information or data with respect to single studies. J.P. was supported by the German National Academy of Sciences Leopoldina (LPDS 2021-05). H.H. was supported by the Marietta-Blau scholarship of the Austrian Agency for Education and Internationalisation (OeAD) and the Deutsche Forschungsgemeinschaft (DFG, German Research Foundation, project ID 422744262 – TRR 289). C.K. received funding from OCENW.XL21.XL21.069 and V.G. from the European Research Council (ERC) under European Union’s Horizon 2020 research and innovation programme, grant ‘HelpUS’ (758703) and from the Dutch Research Council (NWO) grant OCENW.XL21.XL21.069. The funders had no role in study design, data collection and analysis, decision to publish or preparation of the manuscript.
Funding
Open access funding provided by Ruhr-Universität Bochum.
Author information
Authors and Affiliations
Contributions
J.P. contributed to conceptualization, methodology, formal analysis, investigation, data curation, writing the original draft, review and editing, visualization, supervision and project administration. HH contributed to conceptualization, methodology, formal analysis, investigation, data curation, writing the original draft, review and editing, visualization, supervision and project administration. K.F. contributed to investigation, data curation, and review and editing. C.K. and V.G. contributed to conceptualization, and review and editing. F.M. contributed to conceptualization, methodology, formal analysis, investigation, writing the original draft, and review and editing.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Human Behaviour thanks Ville Harjunen, Rebecca Boehme and the other, anonymous, reviewer(s) for their contribution to the peer review of this work. Peer reviewer reports are available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
Supplementary Figs. 1–21 and Tables 1–4.
Supplementary Table 1
List of studies included in and excluded from the meta-analyses/review.
Supplementary Table 2
PRISMA checklist, manuscript.
Supplementary Table 3
PRISMA checklist, abstract.
Source data
Source Data Fig. 2
Effect size/error (columns ‘Hedges_g’ and ‘variance’) information for each study/cohort/effect included in the analysis. Source Data Fig. 3 Effect size/error (columns ‘Hedges_g’ and ‘variance’) together with moderator data (column ‘Outcome’) for each study/cohort/effect included in the analysis. Source Data Fig. 4 Effect size/error (columns ‘Hedges_g’ and ‘variance’) together with moderator data (columns ‘dyad_type’ and ‘skin_to_skin’) for each study/cohort/effect included in the analysis. Source Data Fig. 5 Effect size/error (columns ‘Hedges_g’ and ‘variance’) together with moderator data (column ‘touch_type’) for each study/cohort/effect included in the analysis. Source Data Fig. 6 Effect size/error (columns ‘Hedges_g’ and ‘variance’) together with moderator data (column ‘clin_sample’) for each study/cohort/effect included in the analysis. Source Data Fig. 7 Effect size/error (columns ‘Hedges_g’ and ‘variance’) together with moderator data (column ‘familiarity’) for each study/cohort/effect included in the analysis. Source Data Fig. 7 Effect size/error (columns ‘Hedges_g’ and ‘variance’) together with moderator data (columns ‘touch_duration’ and ‘sessions’) for each study/cohort/effect included in the analysis.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Packheiser, J., Hartmann, H., Fredriksen, K. et al. A systematic review and multivariate meta-analysis of the physical and mental health benefits of touch interventions. Nat Hum Behav 8, 1088–1107 (2024). https://doi.org/10.1038/s41562-024-01841-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41562-024-01841-8