Abstract
To compare the differences between asleep and awake robot-assisted deep brain stimulation (DBS) surgery for Parkinson’s Disease (PD), we conducted this retrospective cohort study included 153 PD patients undergoing bilateral robot-assisted DBS from June 2017 to August 2019, of which 58 cases were performed under general anesthesia (GA) and 95 cases under local anesthesia (LA). Procedure duration, stimulation parameters, electrode implantation accuracy, intracranial air, intraoperative electrophysiological signal length, complications, and Unified PD Rating Scale (UPDRS) measurements were recorded and compared. The clinical evaluation was conducted by two raters who were blinded to the choice of anesthesia. Procedure duration was significantly shorter in the GA group, while on stimulation off medication motor scores (UPDRS-III) were significantly improved in both the GA and LA group. ANCOVA covariated for the baseline UPDRS-III and levodopa challenge exhibited no significant differences. In terms of amplitude, frequency, and pulse width, the stimulation parameters used for DBS power-on were similar. There were no significant differences in electrode implantation accuracy, intraoperative electrophysiological signal length, or intracerebral hemorrhage (no occurrences in either group). The pneumocephalus volume was significantly smaller in the GA group. Six patients exhibited transient throat discomfort associated with tracheal intubation in the GA group. The occurrence of surgical incision infection was similar in both groups. Compared with the awake group, the asleep group exhibited a shorter procedure duration with a similar electrode implantation accuracy and short-term motor improvement. Robot-assisted asleep DBS surgery is a promising surgical method for PD.
Similar content being viewed by others
Introduction
Deep brain stimulation (DBS) surgery is an effective treatment for Parkinson’s disease (PD)1,2. To date, awake surgery has been typically performed with intraoperative test stimulations3. Recently, asleep surgery has been performed under general anesthesia (GA) with intraoperative magnetic resonance imaging (iMRI)4 or computed tomography (iCT)5 to confirm the position of the electrode tip. However, limited research and clinical experience has been reported regarding robot-assisted asleep surgery. Herein, this study reports robot-assisted DBS surgical procedures and clinical results in detail.
Results
General data
In total, 153 patients were retrospectively included in this study, with 58 cases in the GA group and 95 cases in the LA group. The mean ages were 63.5 ± 6.6 years (GA group) and 63.2 ± 9.7 years (LA group). Males represented 37.9% (22/58) of the GA group and 58.9% (56/95) of the LA group. The disease durations were 10.5 ± 5.0 years (GA group) and 8.7 ± 4.6 years (LA group). The Hoehn & Yahr (H & Y) stages were 3.01 ± 0.27 (GA group) and 2.99 ± 0.23 (LA group). The preoperative UPDRS scores (OFF) were 63.8 ± 13.6 (GA group) and 62.2 ± 16.3 (LA group). The preoperative UPDRS-III scores (OFF) were 28.9 ± 6.9 (GA group) and 28.6 ± 9.0 (LA group), and the preoperative UPDRS-III scores (ON) were 14.3 ± 6.6 (GA group) and 14.8 ± 7.3 (LA group). The preoperative levodopa daily doses were 652.4 ± 400.3 mg/d (GA group) and 577.6 ± 340.8 mg/d (LA group). The preoperative levodopa responses (UPDRS-III improvement) were 50.0 ± 17.8 % (GA group) and 48.8 ± 16.2% (LA group). There were no significant differences in the baseline values between the two groups apart from sex and disease duration (Table 1).
Procedure duration, pneumocephalus volume, levodopa equivalent of daily dose reduction, and Tao’s DBS surgery scale
All 306 electrodes of the 153 cases were implanted successfully without adjustment of the electrode position during the ROSA robot-assisted surgery. The duration of the procedure was significantly shorter in the GA group (1.09 ± 0.46 h) than in the LA group (1.54 ± 0.57 h, p < 0.0001). The total MER time was significantly shorter in the GA group (12.03 ± 1.77 min) than in the LA group (12.89 ± 2.73 min, p = 0.0338). There was no significant difference in the levodopa equivalent of daily dose reduction between the two groups (162.8 ± 431.6 mg (GA) vs. 208.0 ± 403.5 mg (LA), p = 0.5141). The pneumocephalus volume was significantly smaller in the GA group (4.35 ± 5.61 cm3) than in the LA group (12.25 ± 13.76 cm3, p < 0.0001). The Tao’s DBS surgery scale6 was significantly higher in the GA group (85.2 ± 9.3) than in the LA group (76.5 ± 8.0, p < 0.0001, Fig. 1).
Electrode implantation accuracy
The electrode implantation accuracy did not differ between the GA group (0.71 ± 0.25 mm) and LA group (0.76 ± 0.23 mm, p = 0.3031). The intraoperative electrophysiological signal length also did not differ between the groups (for STN, 5.90 ± 1.45 mm (GA) vs. 5.47 ± 0.82 mm (LA), p = 0.0630; for Gpi, 8.68 ± 2.52 mm (GA) vs. 9.48 ± 2.39 mm (LA), p = 0.2525).
UPDRS and UPDRS-III
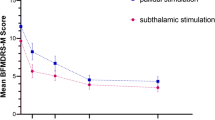
The on stimulation off medication UPDRS scores significantly improved in both groups (GA: 26.84 ± 12.23, p < 0.0001; LA: 25.87 ± 12.59, p < 0.0001). ANCOVA covariated for the baseline UPDRS and levodopa challenge revealed no significant differences (ANCOVA F = 0.009, Bonferroni p = 0.923). The percentage improvement was similar in both groups (42.8 ± 19.0% (GA) and 41.6 ± 15.9% (LA), ANCOVA F = 0.163, Bonferroni p = 0.687). The on stimulation off medication motor scores (UPDRS-III) significantly improved in both group GA (18.31 ± 6.83, p < 0.0001) and LA (16.24 ± 7.14, p < 0.0001). ANCOVA covariated for the baseline UPDRS-III and levodopa challenge revealed no significant differences (ANCOVA F = 1.233, Bonferroni p = 0.269). The percentage improvement was similar in both groups (GA: 63.4 ± 19.2%; LA: 57.0 ± 17.3%; ANCOVA F = 0.718, Bonferroni p = 0.398).
Stimulation parameters
In terms of amplitude, frequency, and pulse width, the stimulation parameters used for DBS power-on were similar (p > 0.05). The amplitude stimulation parameter used for neuromodulation was significantly lower in the GA-STN group (2.33 ± 0.62 V) than in the LA-STN group (2.77 ± 0.70 V, p = 0.0018), yet not significantly different between the GA-GPi (3.94 ± 0.45 V) and LA-GPi groups (3.73 ± 0.70 V, p = 0.2504, Table 2).
Postoperative side effects and complication rates
The postoperative side effects during neuromodulation included muscle contraction (n = 7), dysarthria (n = 1), oculomotor dysfunction (n = 12), dysesthesia (n = 1), dizziness (n = 23), palpitation (n = 10), and dyskinesia (n = 27) in the GA group and muscle contraction (n = 11), dysarthria (n = 2), oculomotor dysfunction (n = 27), dysesthesia (n = 11), dizziness (n = 48), palpitation (n = 3), and dyskinesia (n = 34) in the LA group.
In the LA group, one participant (a 65-year-old male) was allergic to lidocaine. After local administration of lidocaine, the patient immediately exhibited dyspnea, loss of consciousness, and decreased blood pressure. The subsequent rescue was successful. Because of the precise positioning of the robot and fine surgical operations, no intracranial hematomas occurred in any of the cases. Incision infection was similar in both groups (two cases in the GA and two cases in the LA group). In the GA group, one case (a 71-year-old female) exhibited rejection such as swelling and discomfort at the site of skin incision and six patients exhibited transient throat discomfort associated with tracheal intubation.
Discussion
In the last 30 years, awake surgery has been widely performed7, most often under LA with intraoperative test stimulation3. During awake surgery and without intraoperative imaging, neurosurgeons determine electrode placement by relying on electrophysiological typical signals and the patients’ symptom relief without any side effects. In recent years, there have been some reports on the use of intraoperative imaging of DBS under GA. In our current study, we report the application of a robot for DBS of PD patients under GA, with which we achieved good results.
The main difference between asleep and awake DBS surgery is the method of intraoperatively verifying the position of electrode implantation. Some neurosurgical centers have performed asleep DBS surgery under GA with iMRI4,8,9 or iCT merged together with preoperative MRI to verify the accuracy of electrode implantation10,11. It has been demonstrated that the clinical outcomes and complication rates of asleep surgery are comparable to those in historical studies using MER to guide or confirm lead placement under LA12,13,14,15. An advantage of iCT or iMRI guidance is the ability to account for brain shift following dural opening and cerebrospinal fluid loss16. The ideal intraoperative imaging modality for use during asleep DBS surgery remains to be confirmed, and further data are needed to provide accurate comparisons between the outcomes of iCT and iMRI. The disadvantages of iCT and iMRI include longer surgeries and longer times under anesthesia as well as higher risks related to imaging procedures and inevitable errors. The most important disadvantage of iCT and iMRI is that intraoperative images under pneumocephalus or loss of cerebrospinal fluid can merely reflect the real electrode position. The accuracy obtained by merged iMRI or iCT data cannot reflect the real accuracy, which can only be confirmed in postoperative images (after 1–2 weeks) after the brain shift resolves.
Electrode implantation accuracy is a key point related to the prognosis of DBS surgery. The procedures of awake and asleep DBS surgeries rely upon different methods for the verification of the intended target acquisition. There are many factors that affect accuracy, such as head position17, surgical procedures, pneumocephalus, and brain shift. In our study, the electrode implantation accuracy was 0.71 ± 0.25 mm in the GA group and 0.76 ± 0.23 mm in the LA group. Our results indicate that asleep DBS robot-assisted surgery can provide adequate accuracy. In our experience, the specific surgical procedures ensured accuracy by using a modified registration, intraoperative registration, and simulated target verification6,18. Furthermore, intraoperative MER was used to confirm the final position of the electrode.
Despite the support of the above surgical techniques, we used a single-channel microelectrode to record the electrophysiological signals and thus to confirm the target location. Under GA monitored by BIS, many factors can interfere with the MER signal. Sometimes, there was even no MER signal at all. Previously, we reduced the depth of GA. When the BIS value score is >70, BIS monitoring is turned off to avoid interference and to thus obtain a typical MER signal. Once no signal can be recorded, the accuracy of electrode implantation is verified by the merged result of 2 h of postoperative CT and preoperative planning images. Of note, in our 153 cases no adjustment for electrode position was required during surgery.
A meta-analysis found that there were no significant differences between the clinical outcomes of the MER and non-MER groups17. In a retrospective study, the mean (±SD) duration of the procedure for all 323 cases under iCT was 2.51 ± 1.04 h19. Mirzadeh et al.19 found that the MER was an independent contributor to increased procedure durations (+44 min) and that improved accuracy was associated with shorter procedure durations. In our study, the procedure duration was significantly shorter in the GA group. On the basis of the above results, a possible ideal asleep DBS surgery may be performed without MER and intraoperative imaging to reduce excessive surgical procedures. Such an ideal asleep DBS surgery is most likely to be achieved under the following conditions: correction registration, intraoperative registration, and simulated target verification of the robot. Such surgery should be performed by skilled neurosurgeons and their teams. Nevertheless, for this type of surgery to be approved, more prospective clinical studies are necessary.
Most studies that investigated outcomes after DBS asleep surgery and that found comparable results to awake surgery had no control group but rather compared their outcomes with previous studies8,13,20,21. In a retrospective study, Tsai et al.22 found that, in terms of UPDRS score improvement, levodopa equivalent of daily dose reduction and stimulation parameters did not show significant differences between groups after 5 years. Blasberg et al.13 found a significant difference in the percentage reduction of UPDRS-III motor scores due to stimulation after 3 months but not after 1 year. Using the baseline values of UPDRS and levodopa challenge as covariates, we found that the short-term UPDRS-III improvement rate in the GA group was similar to that of the LA group.
The short-term clinical outcomes in this study were consistent with our previous findings regarding the DBS surgical scoring method (Tao’s DBS surgery scale)6. The scale of asleep DBS surgery was higher than that of awake DBS surgery in this study. Although patients undergo extensive pre- and postoperative evaluation, the field lacks a robust scoring system for quantifying DBS surgery. To determine whether a practical scale could assess DBS surgery and predict its clinical significance, we designed the Tao’s DBS surgery scale. The scale draws upon multi-factor statistical analysis of factors that affect the efficacy of DBS surgery in patients with PD and was designed to evaluate the quality of DBS surgery, as well as to help improve its efficacy. It consists of the following parts: electrode implantation duration, postoperative pneumocephalus volume, and electrode fusion error. At present, it is derived from single-center data and thus requires further research and verification.
The current study demonstrated no significant differences in postoperative “freezing” and “speech” between groups. The compared clinical outcome statements still require further observation and long-term follow-up of motor/non-motor and neuropsychological symptoms and side effects (e.g., cognitive, mood, and behavioral effects). Taken together, although most of these studies showed comparable results for both procedures, they were limited because of the absence of control groups or at best unmatched groups with different baseline characteristics, small sample sizes for direct and matched comparisons, or short postoperative observation periods 13.
The literature on the impact of complications associated with the use of GA during asleep DBS surgery is limited. Recent studies15,17,23,24 have demonstrated that the incidence of intracerebral hemorrhages, infections, and epilepsy were similar between asleep and awake DBS surgery, which is generally consistent with our results. In this study, the volume of intracranial air was significantly lower in the GA group than that in the LA group, which is consistent with previous reports17,25. Awake DBS resulted in significantly larger cortical brain shifts25. Additionally, awake DBS surgery has the disadvantage of potential local anesthetic drug allergies26.
A literature review20 published recently revealed that there are no significant differences in cost between awake and asleep DBS surgery. In contrast, in a single academic medical center cost analysis, asleep DBS surgery was associated with lower costs in comparison with the awake procedures27. The cost was influenced by the use of iCT, iMRI, or a robot, as well as anesthesia-related expenses and postoperative incidents or complications.
Certain limitations were present in our study design. First, this study was a retrospective cohort study, which limits its external validity. Nevertheless, the patients in the two groups were consecutively recruited and returned to all postoperative follow-ups. There was no randomization for the group assignment (asleep vs. awake), which implies a selection bias. Second, we investigated robot-assisted asleep DBS surgery for PD in a single center, even though this technique is still not widely used. We insist that this is a DBS surgical method with future prospects. Third, the follow-up time for UPDRS score evaluation (including subscale scores) was 6 months on average. Long-term follow-ups are still needed for both groups. The lack of good-quality randomized clinical trials warrants further research in this field.
Compared with the awake group, the asleep group exhibited a shorter procedure duration and a similar electrode implantation accuracy and short-term motor improvement. In general, DBS surgery should still be performed with the technique that the neurosurgeon and team members are most familiar with, because this provides the patients with the best possible outcome. Robot-assisted asleep DBS surgery is a promising surgical method for PD in the future. However, high-quality epidemiological data are lacking. Thus, a prospective randomized controlled trial with a larger patient population and longer follow-up is needed to confirm the findings and conclusions of this study.
Methods
Patient selection
All PD patients who underwent bilateral subthalamic nucleus (STN) or globus pallidus internus (Gpi) DBS surgery from June 2017 to August 2019 at the General Hospital of Northern Theater Command were included. These patients met the diagnostic criteria of the United Kingdom PD Brain Bank, in which at least two of the cardinal symptoms were present. Before surgery, each patient underwent a levodopa test to ensure a positive levodopa response (Unified PD Rating Scale (UPDRS) part III >30% improvement in scores). The same DBS team, with one senior neurosurgeon (Professor YQ Tao) and one senior anesthesiologist (Doctor DD Song), performed all DBS procedures for these patients at our hospital. The surgical procedures have been described in our previous reports6,18. Because of the lack of DBS surgery guidelines regarding GA or local anesthesia (LA), this choice was made by the patient after we informed the patients and their families about the potential benefits and risks of both GA and LA. The clinical evaluation was conducted by two raters (Doctors Yang Liu and Shimiao Wang) who were blinded to the choice of anesthesia.
Preoperative planning
All patients underwent preoperative MRI (Siemens MAGNETOM Verio 3T Tim) and head contrast-enhanced CT using the parameters7 before DBS surgery. Five metal markers (2023-VG, The ALCIS Company, Besancon, France) were fixed on the patients’ skulls before contrast-enhanced CT scanning. CT data were imported into the ROSA® (Robot of Surgery Assistant, Medtech S.A.S, France) software to create an image fused with the preoperative MRI according to three different blood vessels on the plane of the intended target. MRI and CT images were imported into the ROSA system, and a surgical trajectory was designed according to the location of the nuclei and the optimal cortical puncture point, i.e., where the cortical gyrus was closest to the dura mater, simultaneously avoiding the sulci and blood vessels (Fig. 2). The preoperative planning was led by Professor YQ Tao, and Dr. Hai Jin and Xiao Sun were responsible for inspection and verification.
(There was consent to use the photographs). a Preoperative registration (the first registration) of markers fixed on the skull. b After skin incision and skull hole drilling, intraoperative registration (the second registration) of markers. c, d Intraoperative verification of simulated target before electrode implantation.
The robot-assisted neurosurgical procedure
Preoperative data processing: as metal artifacts of bone markers are present in CT images, we used a modified registration method of robot-assisted DBS surgery, which can reduce the registration error and electrode vector error, as published by our center18. The main surgical procedure: all patients underwent surgery in the supine position with the head elevated at 10–20°. A stereotactic head frame (Leksell Modell G, Elekta Instruments, Inc., GA) was fixed to the patients’ skulls and was mainly used to fix the patients’ heads and then tightly fixed to the connecting rod of the robot. After the laser localization of the robotic manipulator to determine the position of the burr hole, the operator cut the scalp and drilled the burr hole. Preoperative registration (the first registration) of markers was performed (Fig. 3a). Next, intraoperative registration (the second registration) was performed to avoid head shift errors (Fig. 3b); this guaranteed a minimum registration error compared with the preoperative registration. Then the microelectrode on the robotic manipulator verified an implantation error of <0.4 mm (a simulated bone marker notch diameter of 0.8 mm) via simulated target verification (Fig. 3c, d). If the verification error was >0.4 mm (the edge of the bone marker notch), it was slightly corrected by adjusting the screw direction on the microelectrode thruster base to ensure the minimization of any errors before opening the dura mater. The dura mater was opened to a diameter of 2–3 mm so that one parallel steel cannula could be inserted. Then the microelectrode recording (MER) was performed intraoperatively using the alpha-omega microelectrode recording system to confirm that the target was correct. The final placement depth of the electrode was determined according to the intraoperative electrophysiological signal length or the patients’ symptom relief during surgery. When the steel cannula was pushed out after successful implantation of the electrode, the burr hole was immediately closed by bone wax and the lead was fixed. Next, bilateral pulse generators were implanted into subcutaneous pockets of the infraclavicular region. We recorded the duration of surgery beginning with scalp incision and ending with skin suture completion. Electrode implantation duration (~10 min on each side) was recorded beginning with the opening of the dura mater and ending with burr hole closure on each side.
The duration was recorded for the following procedures: (1) skin incision-stage 1 (frontal scalp incisions); (2) dural incision-first side; (3) dural incision-second side; (4) skin closure; (5) skin incision-stage 2 (Impulse Generator, IPG); and (6) skin closure. The duration of the procedure was defined as the interval between skin incision and closure (steps 1–6).
Anesthesia
Awake surgery patients received LA with lidocaine. Asleep surgery patients received GA with endotracheal intubation. Anesthesia was induced by administration of fentanyl (1.5 µg/kg), propofol (1.5 mg/kg), and rocuronium (1 mg/kg). Desflurane inhalation was maintained during surgery and used to keep minimal alveolar concentration during scalp incision and skull hole creation. The depth of GA was adjusted by reducing minimal alveolar concentration of inhalational anesthetics to 0.6 during MER with a bispectral index (BIS) monitor (scores >70). During MER, the BIS monitor was turned off for ~10 min on each side.
Postoperative check
Postoperative CT (Discovery CT750, GE Healthcare), with spiral scanning, 100 KV, 350 mA, and 2.0 mm slice thickness, was performed both 2 h and 1 week after surgery to assess the electrode position, pneumocephalus volume, and complications such as intracranial hemorrhage or electrode offset. The electrode fusion error compared with preoperative planning was defined as the Euclidean difference between the intended and actual trajectories of electrodes on the axial plane of the intended target. The deviation in X- and Y- coordinate vectors of the DBS lead on the intended target Z-plane were measured on each side of the fused images both 2 h and 1 week after surgery, and the total deviation (D) of the electrodes between the intended and actual trajectories was calculated as D = \(\sqrt {{\mathrm{X}}^2 + {\mathrm{Y}}^2}\). The Tao’s DBS surgery scale, including electrode implantation duration, postoperative pneumocephalus volume, and electrode fusion error, was used to assess the DBS surgery.
Clinical evaluation
The UPDRS, the Montreal Cognitive Assessment Scale, and the PD quality of life (PDQL-39) scale were all preoperatively performed on patients. The time of follow-up for postoperative neuromodulation was usually every 3 months. Most patients only required proper adjustment of the original parameters, while some patients required adjustments to the stimulating contacts. For certain patients (with dizziness, blurred vision, or unsatisfactory improvement of gait disturbance), special stimulation modes (such as cross electric pulse, variable frequency stimulation, and low frequency stimulation) were used. Furthermore, different program groups were set up for patients to use in different situations. At the 6-month follow-up, we assessed the short-term clinical efficacy of PD patients with UPDRS and UPDRS-III.
Statistical analysis
Statistical analysis was performed using GraphPad Prism version 8.0 for Mac OS X (GraphPad Software, San Diego, CA, USA). Continuous data were expressed as the mean ± SD, and binary data were provided as percentages. The χ2 test or Fisher’s exact test were used for the binary data, and the paired t-tests or Wilcoxon signed-rank test were used for pre- and postoperative continuous data. The unpaired independent sample t-test or Mann–Whitney U test were used for independent continuous data (depending on whether the variable met the parametric assumptions). Analysis of covariance (ANCOVA) was performed between groups for UPDRS and UPDRS-III improvement comparison, and baseline UPDRS and the levodopa challenge were selected as covariates. The Bonferroni correction was performed for multiple comparisons. P < 0.05 (two-tailed) was considered statistically significant.
Statement regarding ethics committee approval and patient consent
This study was approved by the local ethics committee of the General Hospital of Northern Theater Command and was conducted in accordance with the Declaration of Helsinki. All participants provided written informed consent to take part in this study, and the participants in Fig. 3 provided consent for the photo to be published.
Reporting summary
Further information on research design is available in the Nature Research Reporting Summary linked to this article.
Data availability
The datasets generated and analyzed during the current study are available from the corresponding author upon reasonable request.
References
Michelle, P. et al. Two-year clinical outcomes associated with robotic-assisted subthalamic lead implantation in patients with Parkinson’s disease. J. Robot Surg. 14, 559–565 (2020).
Sato, K. et al. Balance and gait improvements of postoperative rehabilitation in patients with Parkinson’s Disease treated with Subthalamic Nucleus Deep Brain Stimulation (STN-DBS). Parkinsons Dis. 2019, 7104071 (2019).
Sammartino, F., Rege, R. & Krishna, V. Reliability of intraoperative testing during Deep Brain Stimulation surgery. Neuromodulation 23, 525–529 (2020).
Southwell, D. G. et al. Comparison of Deep Brain Stimulation lead targeting accuracy and procedure duration between 1.5-and 3-Tesla interventional magnetic resonance imaging systems: an initial 12-Month experience. Stereotact. Funct. Neurosurg. 94, 102–107 (2016).
Kremer, N. I. et al. Accuracy of intraoperative computed tomography in Deep Brain Stimulation-a prospective non-inferiority study. Neuromodulation 22, 472–477 (2019).
Shun, G. et al. Assessment of Deep Brain Stimulation implantation surgery: a practical scale. World Neurosurg. 134, e1121–e1129 (2020).
Yin, Z. et al. Is awake physiological confirmation necessary for DBS treatment of Parkinson’s disease today? A comparison of intraoperative imaging, physiology, and physiology imaging-guided DBS in the past decade. Brain Stimul. 12, 893–900 (2019).
Mirzadeh, Z. et al. Parkinson’s disease outcomes after intraoperative CT-guided “asleep” deep brain stimulation in the globus pallidus internus. J. Neurosurg. 124, 902–907 (2016).
Chircop, C. et al. MRI-verified “asleep” deep brain stimulation in Malta through cross border collaboration: clinical outcome of the first five years. Br. J. Neurosurg. 32, 365–371 (2018).
Cui, Z. et al. Intraoperative MRI for optimizing electrode placement for deep brain stimulation of the subthalamic nucleus in Parkinson disease. J. Neurosurg. 124, 62–69 (2016).
Bot, M. et al. Accuracy of intraoperative computed tomography during deep brain stimulation procedures: comparison with postoperative magnetic resonance imaging. Stereotact. Funct. Neurosurg. 95, 183–188 (2017).
Chen, T. et al. Clinical outcomes following awake and asleep deep brain stimulation for Parkinson disease. J. Neurosurg. 16, 1–12 (2018).
Blasberg, F. et al. Comparison of awake vs. asleep surgery for subthalamic deep brain stimulation in Parkinson’s disease. Neuromodulation 21, 541–547 (2018).
Brodsky, M. A. et al. Clinical outcomes of asleep vs awake deep brain stimulation for Parkinson disease. Neurology 89, 1944–1950 (2017).
Kochanski, R. B. & Sani, S. Awake versus asleep deep brain stimulation surgery: technical considerations and critical review of the literature. Brain Sci. 8, E17 (2018).
Caio, M. M. et al. Deep brain stimulation outcomes in patients implanted under general anesthesia with frame-based stereotaxy and intraoperative MRI. J. Neurosurg. 129, 1572–1578 (2018).
Liu, Z., He, S. & Li, L. General anesthesia versus local anesthesia for deep brain stimulation in Parkinson’s disease: a meta-analysis. Stereotact. Funct. Neurosurg. 97, 381–390 (2019).
Feng, X. et al. Improved accuracy using a modified registration method of ROSA in deep brain stimulation surgery. Neurosurg. Focus 45, E18 (2018).
Mirzadeh, Z. et al. Procedural variables influencing stereotactic accuracy and efficiency in deep brain stimulation surgery. Oper. Neurosurg. 17, 70–78 (2019).
Jun, W. et al. Comparison of awake and asleep deep brain stimulation for Parkinson’s disease: a detailed analysis through literature review. Neuromodulation 23, 444–450 (2020).
Harries, A. M. et al. Deep brain stimulation of the subthalamic nucleus for advanced Parkinson disease using general anesthesia: long-term results. J. Neurosurg. 116, 107–113 (2012).
Sheng-Tzung, T. et al. Five-year clinical outcomes of local versus general anesthesia deep brain stimulation for Parkinson’s disease. Parkinson’s Dis. 2019, 5676345 (2019).
Ho, A. et al. Awake versus asleep deep brain stimulation for Parkinson’s disease: a critical comparison and meta-analysis. J. Neurol. Neurosurg. Psychiatry 89, 687–691 (2018).
Chen, T. et al. Complication rates, lengths of stay, and readmission rates in “awake” and “asleep” deep brain stimulation. J. Neurosurg. 127, 360–369 (2017).
Ko, A. L. et al. Asleep deep brain stimulation reduces incidence of intracranial air during electrode implantation. Stereotact. Funct. Neurosurg. 96, 83–90 (2018).
Chen, T., Mirzadeh, Z. & Ponce, F. A. “Asleep” Deep Brain Stimulation surgery: acritical review of the literature. World Neurosurg. 105, 191–198 (2017).
Jacob, R. L. et al. Cost analysis of awake versus asleep deep brain stimulation: a single academic health center experience. J. Neurosurg. 124, 1517–1523 (2016).
Acknowledgements
This study was supported by grants from the National Natural Science Foundation of China (81870890 and 81701227), and the Doctoral Research Initiation Foundation of Liaoning province (2020-BS-031).
Author information
Authors and Affiliations
Contributions
H.J., S.G., and X.S. contribute equally to this article. H.J.: research project conception and writing of the first draft of the manuscript. S.G.: design, execution, and review of statistical analysis; and review of the manuscript. X.S.: data collecting and review of the manuscript. Y.T.: research project conception, organisation, and execution; and review of the manuscript. H.H.: design and supervision. D.S., M.X., and Z.X.: anesthesia, review and critique, and final approval. Y.L. and S.W.: patients’ follow-up and clinical evaluation. L.Y. and T.W.: research project-execution and data acquisition. W.S. and H.P.: review and critique, and final approval.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Jin, H., Gong, S., Tao, Y. et al. A comparative study of asleep and awake deep brain stimulation robot-assisted surgery for Parkinson’s disease. npj Parkinsons Dis. 6, 27 (2020). https://doi.org/10.1038/s41531-020-00130-1
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41531-020-00130-1
This article is cited by
-
Effectiveness and reliability of hypnosis in stereotaxy: a randomized study
Acta Neurochirurgica (2024)
-
Frameless Robot-Assisted Asleep Centromedian Thalamic Nucleus Deep Brain Stimulation Surgery in Patients with Drug-Resistant Epilepsy: Technical Description and Short-Term Clinical Results
Neurology and Therapy (2023)
-
Lidocaine
Reactions Weekly (2021)