Abstract
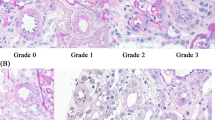
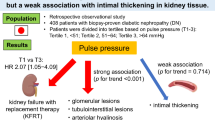
A significant relationship has been established between central hemodynamics and renal microvascular damage. We hypothesized that the increase in the ankle-brachial index (ABI) with age is due to increased arterial stiffness and wave reflection and is thus associated with the pathogenesis of the renal small artery in patients with chronic kidney disease (CKD). We recruited 122 patients with CKD (stages 1–5) who underwent renal biopsy and ABI measurements between 2010 and 2013. Renal small artery intimal thickening (SA-IT) severity was assessed by semiquantitative grading. The median age was 47 years, with a range of 15–86 years (47% women). The median estimated glomerular filtration rate (eGFR) was 62 mL/min/1.73 m2. Compared with patients with the lowest 1–3 SA-IT index quartile, those with the highest quartile of the SA-IT index were older in age had higher mean arterial pressure, ABI, brachial-ankle pulse wave velocity, and lower eGFR. ABI was positively associated with SA-IT severity and inversely associated with eGFR. Multivariate logistic regression analyses showed that ABI was significantly associated with the highest quartile of the SA-IT index (odds ratio per SD increase in ABI, 1.83; 95% confidence interval, 1.08–3.26) and low eGFR (<60 mL/min/1.73 m2) (odds ratio per SD increase in ABI, 1.74; 95% confidence interval, 1.03–3.03). In conclusion, a high normal ABI was associated with severe renal small artery intimal thickening and low eGFR in patients with CKD.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Norgren L, Hiatt WR, Dormandy JA, Nehler MR, Harris KA, Fowkes FGR, et al. Inter-society consensus for the management of peripheral arterial disease (TASC II). J Vasc Surg. 2007;45:S5–S67.
Ishida A, Miyagi M, Kinjo K, Ohya Y. Age- and sex-related effects on ankle-brachial index in a screened cohort of Japanese: the Okinawa Peripheral Arterial Disease Study (OPADS). Eur J Prev Cardiol. 2014;21:712–8.
Wohlfahrt P, Krajčoviechová A, Seidlerová J, Galovcová M, Bruthans J, Filipovský J, et al. Lower-extremity arterial stiffness vs. aortic stiffness in the general population. Hypertens Res. 2013;36:718–24.
Ishida A, Nakachi-Miyagi M, Kinjo K, Iseki K, Ohya Y. A high normal ankle-brachial index is associatedwith proteinuria in a screened cohort of Japanese: the okinawa peripheral arterial disease study. J Hypertens. 2014;32:1435–43.
Khaleghi M, Kullo IJ. Aortic augmentation index is associated with the ankle-brachial index: a community-based study. Atherosclerosis. 2007;195:248–53.
Kinjo Y, Ishida A, Kinjo K, Ohya Y. A high normal ankle-brachial index combined with a high pulse wave velocity is associated with cerebral microbleeds. J Hypertens. 2016;34:1586–93.
O’Rourke MF, Safar ME. Relationship between aortic stiffening and microvascular disease in brain and kidney: cause and logic of therapy. Hypertension. 2005;46:200–4.
Mitchell GF. Effects of central arterial aging on the structure and function of the peripheral vasculature: implications for end-organ damage. J Appl Physiol. 2008;105:1652–60.
Miyagi T, Kohagura K, Ishiki T, Kochi M, Kinjyo T, Kinjyo K, et al. Interrelationship between brachial artery function and renal small artery sclerosis in chronic kidney disease. Hypertens Res. 2014;37:863–9.
Rizzoni D, Porteri E, Guelfi D, Muiesan ML, Valentini U, Cimino A, et al. Structural alterations in subcutaneous small arteries of normotensive and hypertensive patients with non-insulin-dependent diabetes mellitus. Circulation. 2001;103:1238–44.
Rizzoni D, Porteri E, Boari GEM, De Ciuceis C, Sleiman I, Muiesan ML, et al. Prognostic significance of small-artery structure in hypertension. Circulation. 2003;108:2230–5.
Ooi QL, Tow FKN-FH, Deva R, Alias MA, Kawasaki R, Wong TY, et al. The microvasculature in chronic kidney disease. Clin J Am Soc Nephrol. 2011;6:1872–8.
Furuichi K, Shimizu M, Yuzawa Y, Hara A, Toyama T, Kitamura H, et al. Nationwide multicenter kidney biopsy study of Japanese patients with hypertensive nephrosclerosis. Clin Exp Nephrol. 2018;22:629–37.
Matsuo S, Imai E, Horio M, Yasuda Y, Tomita K, Nitta K, et al. Revised equations for estimated GFR from serum creatinine in Japan. Am J Kidney Dis. 2009;53:982–92.
Schwartz GJ, Muñoz A, Schneider MF, Mak RH, Kaskel F, Warady BA, et al. New equations to estimate GFR in children with CKD. J Am Soc Nephrol. 2009;20:629–37.
Jung C-H, Jung S-H, Kim K-J, Kim B-Y, Kim C-H, Kang S-K, et al. Differential associations of central and brachial blood pressure with carotid atherosclerosis and microvascular complications in patients with type 2 diabetes. BMC Cardiovasc Disord. 2014;14:23.
Laurent S, Boutouyrie P. The structural factor of hypertension: large and small artery alterations. Circ Res. 2015;116:1007–21.
Muiesan ML, Salvetti M, Rizzoni D, Paini A, Agabiti-Rosei C, Aggiusti C, et al. Pulsatile hemodynamics and microcirculation: evidence for a close relationship in hypertensive patients. Hypertension. 2013;61:130–6.
Schiffrin EL, Deng LY. Relationship between small-artery structure and systolic, diastolic and pulse pressure in essential hypertension. J Hypertens. 1999;17:381–7.
Safar ME, Rizzoni D, Blacher J, Muiesan ML, Agabiti-Rosei E. Macro and microvasculature in hypertension: therapeutic aspects. J Hum Hypertens. 2008;22:590–5.
Townsend RR, Wilkinson IB, Schiffrin EL, Avolio AP, Chirinos JA, Cockcroft JR, et al. Recommendations for improving and standardizing vascular research on arterial stiffness. Hypertension. 2015;66:698–722.
Hermans MMH, Henry R, Dekker JM, Kooman JP, Kostense PJ, Nijpels G, et al. Estimated glomerular filtration rate and urinary albumin excretion are independently associated with greater arterial stiffness: the Hoorn Study. J Am Soc Nephrol. 2007;18:1942–52.
Upadhyay A, Hwang S-J, Mitchell GF, Vasan RS, Vita JA, Stantchev PI, et al. Arterial stiffness in mild-to-moderate CKD. J Am Soc Nephrol. 2009;20:2044–53.
Rule AD, Amer H, Cornell LD, Taler SJ, Cosio FG, Kremers WK, et al. The association between age and nephrosclerosis on renal biopsy among healthy adults. Ann Intern Med. 2010;152:561–7.
Furuichi K, Shimizu M, Yuzawa Y, Hara A, Toyama T, Kitamura H, et al. Clinicopathological analysis of biopsy-proven diabetic nephropathy based on the Japanese classification of diabetic nephropathy. Clin Exp Nephrol. 2018;22:570–82.
Yamanouchi M, Hoshino J, Ubara Y, Takaichi K, Kinowaki K, Fujii T, et al. Clinicopathological predictors for progression of chronic kidney disease in nephrosclerosis: a biopsy-based cohort study. Nephrol Dial Transplant. 2019;34:1182–8.
Levey AS, Coresh J, Tighiouart H, Greene T, Inker LA. Measured and estimated glomerular filtration rate: current status and future directions. Nat Rev Nephrol. 2020;16:51–64.
Denic A, Mathew J, Lerman LO, Lieske JC, Larson JJ, Alexander MP, et al. Single-nephron glomerular filtration rate in healthy adults. N Engl J Med. 2017;376:2349–57.
Ito S. Cardiorenal syndrome: an evolutionary point of view. Hypertension. 2012;60:589–95.
Aboyans V, Criqui MH, Abraham P, Allison MA, Creager MA, Diehm C, et al. Measurement and interpretation of the ankle-brachial index: a scientific statement from the American Heart Association. Circulation. 2012;126:2890–909.
Acknowledgements
We are grateful to Morihiro Ota, Masato Nohara, and Sakiko Kina-Hatoma for their skillful technical assistance in histological preparation and staining.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
About this article
Cite this article
Zamami, R., Ishida, A., Miyagi, T. et al. A high normal ankle-brachial index is associated with biopsy-proven severe renal small artery intimal thickening and impaired renal function in chronic kidney disease. Hypertens Res 43, 929–937 (2020). https://doi.org/10.1038/s41440-020-0443-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41440-020-0443-z
Keywords
This article is cited by
-
Ankle-brachial index predicts renal outcomes and all-cause mortality in high cardiovascular risk population: a nationwide prospective cohort study in CORE project
International Urology and Nephrology (2022)
-
Combination of low ankle-brachial index and high ankle-brachial index difference for mortality prediction
Hypertension Research (2021)
-
Structural changes in renal arterioles are closely associated with central hemodynamic parameters in patients with renal disease
Hypertension Research (2021)