Abstract
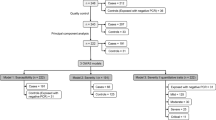
The COVID-19 pandemic remains a significant public health concern despite the new vaccines and therapeutics. The clinical course of acute SARS-CoV-2 infection is highly variable and influenced by several factors related to the virus and the host. Numerous genetic studies, including candidate gene, exome, and genome sequencing studies, genome-wide association studies, and other omics efforts, have proposed various Mendelian and non-Mendelian associations with COVID-19 course. In this study, we conducted whole-exome sequencing on 90 unvaccinated patients from Turkey with no known comorbidities associated with severe COVID-19. Of these patients, 30 had severe, 30 had moderate, and 30 had mild/asymptomatic disease. We identified rare variants in genes associated with SARS-CoV-2 susceptibility and pathogenesis, with an emphasis on genes related to the regulation of inflammation, and discussed these in the context of the clinical course of the patients. In addition, we compared the frequencies of common variants between each group. Even though no variant remained statistically significant after correction for multiple testing, we observed that certain previously associated genes and variants showed significant associations before correction. Our study contributes to the existing literature regarding the genetic susceptibility to SARS-CoV-2. Future studies would be beneficial characterizing the host genetic properties in different populations.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 6 digital issues and online access to articles
$119.00 per year
only $19.83 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout

Similar content being viewed by others
References
Chatterjee S, Bhattacharya M, Nag S, Dhama K, Chakraborty C. A detailed overview of SARS-CoV-2 Omicron: its sub-variants, mutations and pathophysiology, clinical characteristics, immunological landscape, immune escape, and therapies. Viruses. 2023;15:167.
Zhou F, Yu T, Du R, Fan G, Liu Y, Liu Z, et al. Clinical course and risk factors for mortality of adult inpatients with COVID-19 in Wuhan, China: a retrospective cohort study. Lancet. 2020;395:1054–1062.
Zhang JJ, Dong X, Liu GH, Gao YD. Risk and protective factors for COVID-19 morbidity, severity, and mortality. Clin Rev Allergy Immunol. 2023;64:90.
Feng Y, Ling Y, Bai T, Xie Y, Huang J, Li J, et al. COVID-19 with different severities: a multicenter study of clinical features. Am J Respir Crit Care Med. 2020;201:1380–8.
Niederman MS, Richeldi L, Chotirmall SH, Bai C. Rising to the challenge of CovID-19: advice for pulmonary and critical care and an agenda for research. Am J Respir Crit Care Med 2020;201:1019–22.
Wu C, Chen X, Cai Y, Xia J, Zhou X, Xu S, et al. Risk factors associated with acute respiratory distress syndrome and death in patients with coronavirus disease 2019 pneumonia in Wuhan, China. JAMA Intern Med. 2020;180:934–43.
Scully EP, Haverfield J, Ursin RL, Tannenbaum C, Klein SL. Considering how biological sex impacts immune responses and COVID-19 outcomes. Nat Rev Immunol. 2020;20:442–7.
Quillent C, Oberlin E, Braun J, Rousset D, Gonzalez-Canali G, Métais P, et al. HIV-1-resistance phenotype conferred by combination of two separate inherited mutations of CCR5 gene. Lancet. 1998;351:14–18.
Julg B, Moodley ES, Qi Y, Ramduth D, Reddy S, Mncube Z, et al. Possession of HLA class II DRB1*1303 associates with reduced viral loads in chronic HIV-1 clade C and B infection. J Infect Dis. 2011;203:803–9.
Agostini S, Mancuso R, Guerini FR, D’Alfonso S, Agliardi C, Hernis A, et al. HLA alleles modulate EBV viral load in multiple sclerosis. J Transl Med. 2018;16:80 https://doi.org/10.1186/s12967-018-1450-6
Lim HK, Huang SXL, Chen J, Kerner G, Gilliaux O, Bastard P, et al. Severe influenza pneumonitis in children with inherited TLR3 deficiency. J Exp Med. 2019;216:2038–56. https://doi.org/10.1084/jem.20181621
Darbeheshti F, Rezaei N. Genetic predisposition models to COVID-19 infection. Med Hypotheses 2020;142:109818 https://doi.org/10.1016/j.mehy.2020.109818
Ferreira LC, Gomes CEM, Rodrigues-Neto JF, Jeronimo SMB. Genome-wide association studies of COVID-19: connecting the dots. Infect Genet Evol. 2022;106:105379.
Pairo-Castineira E, Clohisey S, Klaric L, Bretherick AD, Rawlik K, Pasko D, et al. Genetic mechanisms of critical illness in COVID-19. Nature. 2021;591:92–98.
Augusto DG, Murdolo LD, Chatzileontiadou DSM, Sabatino JJ, Yusufali T, Peyser ND, et al. A common allele of HLA is associated with asymptomatic SARS-CoV-2 infection. Nature. 2023;620:128–36.
Niemi MEK, Karjalainen J, Liao RG, Neale BM, Daly M, Ganna A, et al. Mapping the human genetic architecture of COVID-19. Nature. 2021;600:472–7.
Casanova JL, Su HC, Abel L, Aiuti A, Almuhsen S, Arias AA, et al. A global effort to define the human genetics of protective immunity to SARS-CoV-2 infection. Cell. 2020;181:1194–9. https://doi.org/10.1016/j.cell.2020.05.016
Zhang Q, Matuozzo D, Le Pen J, Lee D, Moens L, Asano T, et al. Recessive inborn errors of type I IFN immunity in children with COVID-19 pneumonia. J Exp Med. 2022;219:e20220131.
van de Veerdonk FL, Giamarellos-Bourboulis E, Pickkers P, Derde L, Leavis H, van Crevel R, et al. A guide to immunotherapy for COVID-19. Nat Med. 2022;28:39–50.
Carter-Timofte ME, Jørgensen SE, Freytag MR, Thomsen MM, Brinck Andersen NS, Al-Mousawi A, et al. Deciphering the role of host genetics in susceptibility to severe COVID-19. Front Immunol. 2020;11:1606.
Duman N, ALzaidi Z, Aynekin B, Taskin D, Demirors B, Yildirim A, et al. COVID-19 vaccine candidates and vaccine development platforms available worldwide. J Pharm Anal. 2021;11:675–82. https://doi.org/10.1016/j.jpha.2021.09.004
Altmann DM, Boyton RJ. COVID-19 vaccination: the road ahead. Science. 2022;375:1127–32. https://doi.org/10.1126/science.abn1755
Carabelli AM, Peacock TP, Thorne LG, Harvey WT, Hughes J, de Silva TI, et al. SARS-CoV-2 variant biology: immune escape, transmission and fitness. Nat Rev Microbiol. 2023;21:162–77. https://doi.org/10.1038/s41579-022-00841-7
Bowe B, Xie Y, Al-Aly Z. Acute and postacute sequelae associated with SARS-CoV-2 reinfection. Nat Med. 2022;28:2398–405. https://doi.org/10.1038/s41591-022-02051-3
Zeng N, Zhao YM, Yan W, Li C, Lu QD, Liu L, et al. A systematic review and meta-analysis of long term physical and mental sequelae of COVID-19 pandemic: call for research priority and action. Mol Psychiatry. 2023;28:423–33. https://doi.org/10.1038/s41380-022-01614-7
Genetic susceptibility to severe COVID-19. Infect Genet Evol. 2023;110:105426. https://doi.org/10.1016/j.meegid.2023.105426
Kiraz A, Sezer O, Alemdar A, Canbek S, Duman N, Bisgin A, et al. Contribution of genotypes in Prothrombin and Factor V Leiden to COVID‐19 and disease severity in patients at high risk for hereditary thrombophilia. Abst J Med Virol. 2023;95. https://doi.org/10.1002/jmv.28457
Karczewski KJ, Francioli LC, Tiao G, Cummings BB, Alföldi J, Wang Q, et al. The mutational constraint spectrum quantified from variation in 141,456 humans. Nature. 2020;581:434–43.
Wang K, Li M, Hakonarson H. ANNOVAR: functional annotation of genetic variants from high-throughput sequencing data. Nucleic Acids Res. 2010;38:e164 https://doi.org/10.1093/NAR/GKQ603
Bardou P, Mariette J, Escudié F, Djemiel C, Klopp C. Jvenn: an interactive Venn diagram viewer. BMC Bioinforma. 2014;15:1–7.
Köhler S, Gargano M, Matentzoglu N, Carmody LC, Lewis-Smith D, Vasilevsky NA, et al. The human phenotype ontology in 2021. Nucleic Acids Res. 2021;49:D1207–D1217. https://doi.org/10.1093/nar/gkaa1043
Gordon DE, Jang GM, Bouhaddou M, Xu J, Obernier K, White KM, et al. A SARS-CoV-2 protein interaction map reveals targets for drug repurposing. Nature. 2020;583:459–68. https://doi.org/10.1038/s41586-020-2286-9
Martin AR, Williams E, Foulger RE, Leigh S, Daugherty LC, Niblock O, et al. PanelApp crowdsources expert knowledge to establish consensus diagnostic gene panels. Nat Genet. 2019;51:1560–5. https://doi.org/10.1038/s41588-019-0528-2
McLaren W, Gil L, Hunt SE, Riat HS, Ritchie GRS, Thormann A, et al. The Ensembl variant effect predictor. Genome Biol. 2016;17. https://doi.org/10.1186/s13059-016-0974-4.
Rentzsch P, Witten D, Cooper GM, Shendure J, Kircher M. CADD: predicting the deleteriousness of variants throughout the human genome. Nucleic Acids Res. 2019;47:D886–D894. https://doi.org/10.1093/nar/gky1016
Frazer J, Notin P, Dias M, Gomez A, Min JK, Brock K, et al. Disease variant prediction with deep generative models of evolutionary data. Nature. 2021;599:91–95. https://doi.org/10.1038/s41586-021-04043-8
Henikoff S, Henikoff JG. Amino acid substitution matrices from protein blocks. Proc Natl Acad Sci USA. 1992;89:10915–9. https://doi.org/10.1073/pnas.89.22.10915
Ge SX, Jung D, Jung D, Yao R. ShinyGO: a graphical gene-set enrichment tool for animals and plants. Bioinformatics. 2020;36:2628–9. https://doi.org/10.1093/bioinformatics/btz931
Benjamini Y, Hochberg Y. Controlling the false discovery rate: a practical and powerful approach to multiple testing. J R Stat Soc Ser B. 1995;57. https://doi.org/10.1111/j.2517-6161.1995.tb02031.x.
R Core Team. R Core Team 2021 R: a language and environment for statistical computing. R foundation for statistical computing. https://www.R-project.org/. R Found Stat Comput. 2022;2.
Liu Y, Liu FL, Bai ZJ, Zhao N, Zhang LY, Lu X, et al. Defective activities, but not secretions, resulting from gene point mutations of human mannan-binding lectin. Mol Med Rep. 2012;5:1121–7.
Gao, dong Y, Ding M, Dong X, Zhang Jjin, Kursat Azkur A, et al. Risk factors for severe and critically ill COVID-19 patients: a review. Allergy. 2021;76:428–55.
Zhang SY, Zhang Q, Casanova JL, Su HC, Abel L, Bastard P, et al. Severe COVID-19 in the young and healthy: monogenic inborn errors of immunity? Nat Rev Immunol. 2020;20:455–6.
Ishak A, Mehendale M, AlRawashdeh MM, Sestacovschi C, Sharath M, Pandav K, et al. The association of COVID-19 severity and susceptibility and genetic risk factors: a systematic review of the literature. Gene. 2022;836:146674 https://doi.org/10.1016/J.GENE.2022.146674
Van Der Made CI, Simons A, Schuurs-Hoeijmakers J, Van Den Heuvel G, Mantere T, Kersten S, et al. Presence of genetic variants among young men with severe COVID-19. JAMA J Am Med Assoc. 2020;324:663–73. https://doi.org/10.1001/jama.2020.13719
Asano T, Boisson B, Onodi F, Matuozzo D, Moncada-Velez M, Renkilaraj MRLM, et al. X-linked recessive TLR7 deficiency in ~1% of men under 60 years old with life-threatening COVID-19. Sci Immunol. 2021;6:eabl4348 https://doi.org/10.1126/SCIIMMUNOL.ABL4348
Edahiro R, Shirai Y, Takeshima Y, Sakakibara S, Yamaguchi Y, Murakami T, et al. Single-cell analyses and host genetics highlight the role of innate immune cells in COVID-19 severity. Nat Genet. 2023;55:753–67. https://doi.org/10.1038/s41588-023-01375-1
Cen X, Wang F, Huang X, Jovic D, Dubee F, Yang H, et al. Towards precision medicine: Omics approach for COVID-19. Biosaf Heal. 2023;5:78–88. https://doi.org/10.1016/j.bsheal.2023.01.002
Kousathanas A, Pairo-Castineira E, Rawlik K, Stuckey A, Odhams CA, Walker S, et al. Whole-genome sequencing reveals host factors underlying critical COVID-19. Nature. 2022;607:97.
Degenhardt F, Ellinghaus D, Juzenas S, Lerga-Jaso J, Wendorff M, Maya-Miles D, et al. Detailed stratified GWAS analysis for severe COVID-19 in four European populations. Hum Mol Genet. 2022;31:3945.
Rusiñol L, Puig L. Tyk2 targeting in immune-mediated inflammatory diseases. Int J Mol Sci. 2023;24:3391.
Pairo-Castineira E, Rawlik K, Bretherick AD, Qi T, Wu Y, Nassiri I, et al. GWAS and meta-analysis identifies 49 genetic variants underlying critical COVID-19. Nature. 2023;617:764–8. https://doi.org/10.1038/s41586-023-06034-3
Ng ASL, Kramer J, Centurion A, Dalmau J, Huang E, Cotter JA, et al. Clinico-pathological correlation in adenylate kinase 5 autoimmune limbic encephalitis. J Neuroimmunol. 2015;287:31–5. https://doi.org/10.1016/j.jneuroim.2015.08.009
Veleri S. Neurotropism of SARS-CoV-2 and neurological diseases of the central nervous system in COVID-19 patients. Exp Brain Res. 2022;240:9–25. https://doi.org/10.1007/s00221-021-06244-z
Sadeghi MB, Nakhaee A, Saravani R, Sargazi S. Significant association of LXRβ (NR1H2) polymorphisms (rs28514894, rs2303044) with type 2 diabetes mellitus and laboratory characteristics. J Diabetes Metab Disord. 2021;20:261–70. https://doi.org/10.1007/s40200-021-00740-3
Korf H, Vander Beken S, Romano M, Steffensen KR, Stijlemans B, Gustafsson JÅ, et al. Liver X receptors contribute to the protective immune response against Mycobacterium tuberculosis in mice. J Clin Investig. 2009;119:1626–37.
Tangye SG, Al-Herz W, Bousfiha A, Cunningham-Rundles C, Franco JL, Holland SM, et al. Human inborn errors of immunity: 2022 update on the classification from the International Union of Immunological Societies Expert Committee. J Clin Immunol. 2022;42:1473.
Ramasamy S, Subbian S. Critical determinants of cytokine storm and type I interferon response in COVID-19 pathogenesis. Clin Microbiol Rev. 2021;34:e00299–20. https://doi.org/10.1128/CMR.00299-20
Touitou I, Koné-Paut I. Autoinflammatory diseases. Best Pr Res Clin Rheumatol. 2008;22:811–29.
Miano M, Cappelli E, Pezzulla A, Venè R, Grossi A, Terranova P, et al. FAS-mediated apoptosis impairment in patients with ALPS/ALPS-like phenotype carrying variants on CASP10 gene. Br J Haematol. 2019;187:502–8.
Zhang K, Astigarraga I, Bryceson Y, Lehmberg K, Machowicz R, Marsh R, et al. Familial hemophagocytic lymphohistiocytosis. GeneReviews® 2021. https://www.ncbi.nlm.nih.gov/books/NBK1444/ (accessed 6 April 2023).
Cunningham L, Kimber I, Basketter D, Simmonds P, McSweeney S, Tziotzios C, et al. Perforin, COVID-19 and a possible pathogenic auto-inflammatory feedback loop. Scand J Immunol. 2021;94:e13102 https://doi.org/10.1111/SJI.13102
Abidi E, El Nekidy WS, Alefishat E, Rahman N, Petroianu GA, El-Lababidi R, et al. Tocilizumab and COVID-19: timing of administration and efficacy. Front Pharm. 2022;13:825749 https://doi.org/10.3389/FPHAR.2022.825749
Cabrera-Marante O, de Frías ER, Pleguezuelo DE, Allende LM, Serrano A, Laguna-Goya R, et al. Perforin gene variant A91V in young patients with severe COVID-19. Haematologica. 2020;105:2844–6.
Wu J, Fernandes-Alnemri T, Alnemri ES. Involvement of the AIM2, NLRC4, and NLRP3 inflammasomes in caspase-1 activation by Listeria monocytogenes. J Clin Immunol. 2010;30:693–702. https://doi.org/10.1007/s10875-010-9425-2
Strober W, Watanabe T. NOD2, an intracellular innate immune sensor involved in host defense and Crohn’s disease. Mucosal Immunol. 2011;4:484–95. https://doi.org/10.1038/mi.2011.29
Jackson CB, Farzan M, Chen B, Choe H. Mechanisms of SARS-CoV-2 entry into cells. Nat Rev Mol Cell Biol. 2022;23:3–20. https://doi.org/10.1038/s41580-021-00418-x
Duman N, Tuncel G, Bisgin A, Bozdogan ST, Sag SO, Gul S, et al. Analysis of ACE2 and TMPRSS2 coding variants as a risk factor for SARS‐CoV‐2 from 946 whole‐exome sequencing data in the Turkish population. Abst J Med Virol. 2022;94:5225–43. https://doi.org/10.1002/jmv.27976.
Baldassarri M, Fava F, Fallerini C, Daga S, Benetti E, Zguro K, et al. Severe COVID-19 in hospitalized carriers of single CFTR pathogenic variants. J Pers Med. 2021;11:558 https://doi.org/10.3390/jpm11060558
Nunnari G, Sanfilippo C, Castrogiovanni P, Imbesi R, Li Volti G, Barbagallo I, et al. Network perturbation analysis in human bronchial epithelial cells following SARS-CoV2 infection. Exp Cell Res. 2020;395:112204 https://doi.org/10.1016/j.yexcr.2020.112204
Zhang Q, Liu Z, Moncada-Velez M, Chen J, Ogishi M, Bigio B, et al. Inborn errors of type I IFN immunity in patients with life-threatening COVID-19. Science. 2020;370:eabd4570.
Khalil BA, Elemam NM, Maghazachi AA. Chemokines and chemokine receptors during COVID-19 infection. Comput Struct Biotechnol J. 2021;19:976–88. https://doi.org/10.1016/j.csbj.2021.01.034
Kuijpers Y, Chu X, Jaeger M, Moorlag SJCFM, Koeken VACM, Zhang B, et al. The genetic risk for COVID-19 severity is associated with defective immune responses. Front Immunol. 2022;13:859387.
Funding
The study was funded by Erciyes University BAP TCD-2021-10936 and received by Munis Dundar.
Author information
Authors and Affiliations
Contributions
MD, KU, and FO conceived the study. MD was in charge of overall direction and planning. MD, OY, GZ, NT, KG, KU, and FO designed the study protocol. KU, FO, RCY, HS, ZOS, OB, YS, KG, and AKA provided and gathered patient data. KU and HA contributed to the molecular studies. GZ, VE, AC, and IOS performed the statistical analyses. KU and FO performed the molecular analyses. FO, KU, GZ, and VE wrote the manuscript, MD and YO provided supervision and critical review of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Uslu, K., Ozcelik, F., Zararsiz, G. et al. Deciphering the host genetic factors conferring susceptibility to severe COVID-19 using exome sequencing. Genes Immun 25, 14–42 (2024). https://doi.org/10.1038/s41435-023-00232-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41435-023-00232-9