Abstract
Background/Objective
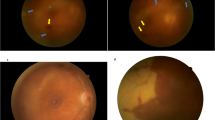
Acute retinal necrosis (ARN) is a vision-threatening disease caused by herpesvirus infection. This study aimed to investigate the visual prognostic factors that could be determined at the initial visit.
Subjects and Methods
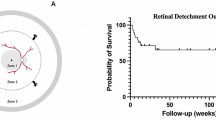
This retrospective study included 34 patients with ARN. Logistic regression analysis was employed to evaluate the associations between poor final visual outcomes and various factors, including poor initial visual acuity, presence of retinal detachment at the initial visit, posterior extension of necrotizing retinitis, and circumferential extension of necrotizing retinitis. Posterior extension was evaluated with three zonings, from the periphery (zone 3), mid-periphery (zone 2), and macula (zone 1). Circumferential extension was evaluated according to the degree of necrotizing retinitis lesions using ultra-wide fundus imaging.
Results
The mean logarithm of the minimum angle of resolution was 0.63 ± 0.68 at the initial visit and 0.83 ± 0.65 at 12 months after the initial visit. Seven patients had a retinal detachment. The distribution of posterior extension at the initial visit was 5 in zone 1, 20 in zone 2, and 9 in zone 3. The average of necrotizing retinitis lesion angle was 249 ± 115°. The logistic regression analysis revealed that participants with wide angles of necrotizing retinitis were associated with final poor vision, with an odds ratio of 1.28 per 30° increase (95%CI: 1.00–1.65, p = 0.03).
Conclusions
Assessment of the widespread circumferential extension of white necrotizing retinal lesions at the initial visit is a crucial risk factor for the visual prognosis in ARN.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 18 print issues and online access
$259.00 per year
only $14.39 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
Data availability
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
References
Fisher JP, Lewis ML, Blumenkranz M, Culbertson WW, Flynn HW Jr, Clarkson JG, et al. The acute retinal necrosis syndrome. Part 1: Clinical manifestations. Ophthalmology 1982;89:1309–16.
Urayama A, Yamada N, Sasaki T. Unilateral acute uveitis with retinal periarteritis and detachment. Rinsho Ganka. 1971;25:607–19.
Cochrane TF, Silvestri G, McDowell C, Foot B, McAvoy CE. Acute retinal necrosis in the United Kingdom: results of a prospective surveillance study. Eye. 2012;26:370–7.
Okazawa R, Iwai S, Nagura K, Sora D, Sato T, Takayama K, et al. Epidemiology and characteristics of common forms of anterior uveitis at initial presentation in a tertiary facility in Japan. Jpn J Ophthalmol. 2023;67:22–31.
Powell B, Wang D, Llop S, Rosen RB. Management strategies of acute retinal necrosis: current perspectives. Clin Ophthalmol. 2020;14:1931–43.
Takase H, Okada AA, Goto H, Mizuki N, Namba K, Ohguro N, et al. Development and validation of new diagnostic criteria for acute retinal necrosis. Jpn J Ophthalmol. 2015;59:14–20.
Kim DY, Jo J, Joe SG, Lee JY, Yoon YH, Kim JG. Clinical feature and visual prognosis of acute retinal necrosis according to the initially involved zone and extent: 10-year experience. Eur J Ophthalmol. 2019;29:244–50.
Paolo M, Marta Z, Stefania F, Viola T, Giacomo C, Arturo C, et al. Visual outcome and poor prognostic factors in acute retinal necrosis syndrome. Graefes Arch Clin Exp Ophthalmol. 2020;258:1851–6.
Lei B, Zhou M, Wang Z, Chang Q, Xu G, Jiang R. Ultra-wide-field fundus imaging of acute retinal necrosis: clinical characteristics and visual significance. Eye. 2020;34:864–72.
Li S, Sun L, Liu C, Wang W, Huang S, Zhang T, et al. Clinical features of ocular toxocariasis: a comparison between ultra-wide-field and conventional camera imaging. Eye. 2021;35:2855–63.
Patel CK, Fung TH, Muqit MM, Mordant DJ, Brett J, Smith L, et al. Non-contact ultra-widefield imaging of retinopathy of prematurity using the Optos dual wavelength scanning laser ophthalmoscope. Eye. 2013;27:589–96.
Fukuda Y, Nakao S, Kohno RI, Ishikawa K, Shimokawa S, Shiose S, et al. Postoperative follow-up of submacular hemorrhage displacement treated with vitrectomy and subretinal injection of tissue plasminogen activator: ultrawide-field fundus autofluorescence imaging in gas-filled eyes. Jpn J Ophthalmol. 2022;66:264–70.
Lei B, Wang Z, Shu Q, Gu R, Zhang Y, Jiang R, et al. Observation of varicella zoster virus-induced acute retinal necrosis: viral load detection and visual outcome. Eye. 2022;36:1209–16.
Bavinger JC, Anthony CL, Lindeke-Myers AT, Lynch S, Xu LT, Barnett J, et al. Risk factors for retinal detachment in acute retinal necrosis. Ophthalmol Retin. 2022;6:478–83.
Butler NJ, Moradi A, Salek SS, Burkholder BM, Leung TG, Dunn JP, et al. Acute retinal necrosis: presenting characteristics and clinical outcomes in a cohort of polymerase chain reaction-positive patients. Am J Ophthalmol. 2017;179:179–89.
Fan S, Lin D, Wang Y. Role of Prophylactic vitrectomy in acute retinal necrosis in preventing rhegmatogenous retinal detachment: systematic review and meta-analysis. Ocul Immunol Inflamm. 2022;30:515–9.
Ishida T, Sugamoto Y, Sugita S, Mochizuki M. Prophylactic vitrectomy for acute retinal necrosis. Jpn J Ophthalmol. 2009;53:486–9.
Iwahashi-Shima C, Azumi A, Ohguro N, Okada AA, Kaburaki T, Goto H, et al. Acute retinal necrosis: factors associated with anatomic and visual outcomes. Jpn J Ophthalmol. 2013;57:98–103.
Luo YH, Duan XC, Chen BH, Tang LS, Guo XJ. Efficacy and necessity of prophylactic vitrectomy for acute retinal necrosis syndrome. Int J Ophthalmol. 2012;5:482–7.
Schulze-Bonsel K, Feltgen N, Burau H, Hansen L, Bach M. Visual acuities “hand motion” and “counting fingers” can be quantified with the freiburg visual acuity test. Invest Ophthalmol Vis Sci. 2006;47:1236–40.
Holland GN, Buhles WC Jr, Mastre B, Kaplan HJ. A controlled retrospective study of ganciclovir treatment for cytomegalovirus retinopathy. Use of a standardized system for the assessment of disease outcome. UCLA CMV Retinopathy. Study Group Arch Ophthalmol. 1989;107:1759–66.
Witmer MT, Pavan PR, Fouraker BD, Levy-Clarke GA. Acute retinal necrosis associated optic neuropathy. Acta Ophthalmol. 2011;89:599–607.
Takase H, Goto H, Namba K, Mizuki N, Okada AA, Ohguro N, et al. Clinical characteristics, management, and factors associated with poor visual prognosis of acute retinal necrosis. Ocul Immunol Inflamm. 2022;30:48–53.
Hillenkamp J, Nolle B, Bruns C, Rautenberg P, Fickenscher H, Roider J. Acute retinal necrosis: clinical features, early vitrectomy, and outcomes. Ophthalmology. 2009;116:1971–5.e2.
Zhao XY, Meng LH, Zhang WF, Wang DY, Chen YX. Retinal detachment after acute retinal necrosis and the efficacies of different interventions: A Systematic Review and Metaanalysis. Retina. 2021;41:965–78.
Clarkson JG, Blumenkranz MS, Culbertson WW, Flynn HW Jr, Lewis ML. Retinal detachment following the acute retinal necrosis syndrome. Ophthalmology 1984;91:1665–8.
Wu CY, Fan J, Davis JL, Berrocal AM, Haddock LJ, Yannuzzi NA, et al. Surgical 0utcomes of acute retinal necrosis-related retinal detachment in polymerase chain reaction-positive patients: a single-center experience. Ophthalmol Retin. 2022;6:992–1000.
Meghpara B, Sulkowski G, Kesen MR, Tessler HH, Goldstein DA. Long-term follow-up of acute retinal necrosis. Retina. 2010;30:795–800.
Acknowledgements
We would like to thank Editage for English language editing.
Funding
This study was supported by grants from the Japan Society for the Promotion of Science (JSPS) through JP21K09745 and JP24K12810 (Grant C to AT), the Ministry of Education, Science, Sports, and Culture of Japan (Tokyo, Japan), the Japan Agency for Medical Research and Development (AMED) through grand number 23jk0210042h0001 (NY: Tokyo, Japan), Alcon Pharma Research Grants (AT; Tokyo, Japan) and AMO Japan Research Grants (AT; Tokyo, Japan). The funding sources had no role in the design and conduct of the study.
Author information
Authors and Affiliations
Contributions
Concept and design of the work: CF, AT, NY; Acquisition or interpretation of data: CF, EH, KA, MS, HT, KY, TI, KI, YM, TH; Analysis of data: CF, AT, NY; Drafting of the manuscript: CF, AT; Statistical analysis: CF, MA; Critical revision of the article: EH, NY, KHS.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Fukui, C., Takeda, A., Hasegawa, E. et al. Factors at the initial visit associated with poor visual outcomes in patients with acute retinal necrosis. Eye (2024). https://doi.org/10.1038/s41433-024-03207-w
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41433-024-03207-w