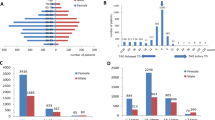
Abstract
Background/objectives
We aimed to investigate the prevalence, risk factors, and prognosis of Graves’ orbitopathy (GO) in patients with thyroid cancer without a history of hyperthyroidism.
Subjects/methods
This retrospective cohort study analysed a sample from the Korean National Health Insurance Service database, which included 1,137,861 subjects from 2002 through 2019. Patients diagnosed with thyroid cancer, without a history of hyperthyroidism, were identified according to the Korean Standard Classification of Disease codes. The study compared the type of surgery, dose of radioactive iodine (RAI), and daily average thyroid hormone dose between patients who developed GO after being diagnosed with thyroid cancer and those who did not develop GO. We analysed the course of GO and the type of treatment.
Results
A total of 8499 cancer patients without a history of hyperthyroidism were identified, among whom 7836 underwent thyroidectomy. Of those who underwent thyroidectomy, 12 developed GO postoperatively. Among the 663 patients who did not undergo thyroidectomy, none developed GO. The prevalence of GO among thyroid cancer patients was 0.14%. The GO group received a significantly higher total RAI dose than the non-GO group (p = 0.036). There were no significant differences in sex, age, type of surgery, rate of RAI treatment, or average thyroid hormone dose between the two groups. One of the 12 patients who developed GO required intravenous steroids.
Conclusions
Although GO rarely develops in thyroid cancer patients without coexisting hyperthyroidism, the total RAI dose may increase its risk. Further research would help clarify GO’s association with thyroid cancer.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 18 print issues and online access
$259.00 per year
only $14.39 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout

Similar content being viewed by others
Data availability
Datasets used and analysed during the current study are available from the corresponding author upon reasonable request.
References
Bartalena L, Kahaly GJ, Baldeschi L, Dayan CM, Eckstein A, Marcocci C, et al. The 2021 European Group on Graves’ orbitopathy (EUGOGO) clinical practice guidelines for the medical management of Graves’ orbitopathy. Eur J Endocrinol. 2021;185:G43–g67.
Bartalena L, Tanda ML. Current concepts regarding Graves’ orbitopathy. J Intern Med. 2022;292:692–716.
Keskin C, Sahin M, Hasanov R, Aydogan B, Demir O, Emral R, et al. Frequency of thyroid nodules and thyroid cancer in thyroidectomized patients with Graves’ disease. Arch Med Sci. 2020;16:302–7.
Ren M, Wu MC, Shang CZ, Wang XY, Zhang JL, Cheng H, et al. Predictive factors of thyroid cancer in patients with Graves’ disease. World J Surg. 2014;38:80–7.
Pazaitou-Panayiotou K, Michalakis K, Paschke R. Thyroid cancer in patients with hyperthyroidism. Horm Metab Res. 2012;44:255–62.
Louvet C, De Bellis A, Pereira B, Bournaud C, Kelly A, Maqdasy S, et al. Time course of Graves’ orbitopathy after total thyroidectomy and radioiodine therapy for thyroid cancer. Medicines. 2016;95:e5474.
Yu P, Liu S, Zhou X, Huang T, Li Y, Wang H, et al. Thyroid-associated orbitopathy in patients with thyroid carcinoma: a case report of 5 cases. Medicines. 2017;96:e8768.
Lee SH, Lee JK. Clinical manifestations of thyroid-associated ophthalmopathy accompanied by thyroid cancer. J Korean Ophthalmol Soc. 2022;63:807–13.
Murayama D, Toda S, Okubo Y, Hayashi H, Matsui A, Yasukawa M, et al. Thyroid-associated ophthalmopathy after radioactive iodine therapy for metastatic follicular thyroid carcinoma. Case Rep. Endocrinol. 2021;2021:3024639.
Jang SY, Lee KH, Oh JR, Kim BY, Yoon JS. Development of thyroid-associated ophthalmopathy in patients who underwent total thyroidectomy. Yonsei Med J. 2015;56:1389–94.
Perros P, Hegedüs L, Bartalena L, Marcocci C, Kahaly G, Baldeschi L, et al. Graves’ orbitopathy as a rare disease in Europe: a European Group on Graves’ Orbitopathy (EUGOGO) position statement. Orphanet J Rare Dis. 2017;12:72.
Lazarus JH. Epidemiology of Graves’ orbitopathy (GO) and relationship with thyroid disease. Best Pract Res Clin Endocrinol Metab. 2012;26:273–9.
Hiromatsu Y, Eguchi H, Tani J, Kasaoka M, Teshima Y. Graves’ ophthalmopathy: epidemiology and natural history. Intern Med. 2014;53:353–60.
Muñoz-Ortiz J, Sierra-Cote MC, Zapata-Bravo E, Valenzuela-Vallejo L, Marin-Noriega M, Uribe-Reina P, et al. Prevalence of hyperthyroidism, hypothyroidism, and euthyroidism in thyroid eye disease: a systematic review of the literature. Syst Rev. 2020;9:201.
Bartalena L, Piantanida E, Gallo D, Lai A, Tanda M. Epidemiology, natural history, risk factors, and prevention of Graves’ orbitopathy. Front Endocrinol (Lausanne). 2020;11:615993.
Lahooti H, Shanmuganathan T, Champion BL, Wall JR. Serum thyroglobulin levels in patients with thyroid autoimmunity with and without ophthalmopathy or isolated upper eyelid retraction. Int Trends Immun. 2015;3:22–7.
Shanmuganathan T, Girgis C, Lahooti H, Champion B, Wall J. Does autoimmunity against thyroglobulin play a role in the pathogenesis of Graves’ ophthalmopathy: a review. Clin Ophthalmol. 2015;9:2271–6.
Adhami M, Michail P, Rao A, Bhatt C, Grodski S, Serpell J, et al. Anti-thyroid antibodies and TSH as potential markers of thyroid carcinoma and aggressive behavior in patients with indeterminate fine-needle aspiration cytology. World J Surg. 2020;44:363–70.
Jo K, Kim MH, Ha J, Lim Y, Lee S, Bae JS, et al. Prognostic value of preoperative anti-thyroglobulin antibody in differentiated thyroid cancer. Clin Endocrinol (Oxf). 2017;87:292–9.
Patell R, Mikhael A, Tabet M, Bena J, Berber E, Nasr C. Assessing the utility of preoperative serum thyroglobulin in differentiated thyroid cancer: a retrospective cohort study. Endocrine. 2018;61:506–10.
Kim H, Kim YN, Kim HI, Park SY, Cheo J, Kim J, et al. Preoperative serum thyroglobulin predicts initial distant metastasis in patients with differentiated thyroid cancer. Sci Rep. 2017;7:16955.
Kim H, Park SY, Choe JH, Kim JS, Hahn SY, Kim SW, et al. Preoperative serum thyroglobulin and its correlation with the burden and extent of differentiated thyroid cancer. Cancers (Basel). 2020;12:625.
Sanyal P, Bing-You RG, Braverman LE. Use of methotrexate to treat isolated Graves ophthalmopathy developing years after thyroidectomy and iodine 131 treatment of papillary thyroid cancer. Endocr Pract. 2008;14:422–5.
Giovansili L, Cayrolle G, Belange G, Clavel G, Herdan M. Graves’ ophthalmopathy after total thyroidectomy for papillary carcinoma. Ann Endocrinol (Paris). 2011;72:42–4.
Kim YI, Im HJ, Paeng JC, Cheon GJ, Kang KW, Lee DS, et al. Serum thyroglobulin level after radioiodine therapy (Day 3) to predict successful ablation of thyroid remnant in postoperative thyroid cancer. Ann Nucl Med. 2015;29:184–9.
Marinò M, Lisi S, Pinchera A, Mazzi B, Latrofa F, Sellari-Franceschini S, et al. Identification of thyroglobulin in orbital tissues of patients with thyroid-associated ophthalmopathy. Thyroid. 2001;11:177–85.
Marinò M, Chiovato L, Lisi S, Altea M, Marcocci C, Pinchera A. Role of thyroglobulin in the pathogenesis of Graves’ ophthalmopathy: the hypothesis of Kriss revisited. J Endocrinol Invest. 2004;27:230–6.
Lisi S, Marinò M, Pinchera A, Mazzi B, Cosmo C, Sellari-Franceschini S, et al. Thyroglobulin in orbital tissues from patients with thyroid-associated ophthalmopathy: predominant localization in fibroadipose tissue. Thyroid. 2002;12:351–60.
Resende de Paiva C, Grønhøj C, Feldt-Rasmussen U, von Buchwald C. Association between Hashimoto’s thyroiditis and thyroid cancer in 64,628 patients. Front Oncol. 2017;7:53.
Bahn RS. Current insights into the pathogenesis of Graves’ ophthalmopathy. Horm Metab Res. 2015;47:773–8.
Hamada N, Okamoto Y, Yoshida H, Tsumura K, Nakamura Y, Noh JY. Sympathetic overactivity in the development of eyelid retraction in a patient with euthyroid Graves’ disease evaluated by accommodation. Endocr J. 2000;47:623–8.
Rogers PB, Gupta N, Rose GE, Plowman PN. Thyroid eye disease associated with athyria Br J Ophthalmol. 2000;84:439.
Antonelli A, Fallahi P, Tolari S, Ferrari SM, Ferrannini E. Thyroid-associated ophthalmopathy and TSH receptor autoantibodies in nonmetastatic thyroid cancer after total thyroidectomy. Am J Med Sci. 2008;336:288-90.
Funding
This research was supported by the National Research Foundation of Korea (NRF) grant funded by the Korean government (MSIT) (NRF-2021R1A2C1011351) and the Chung-Ang University Research Grants in 2023. The funding organisation had no role in the design or conduct of this research.
Author information
Authors and Affiliations
Contributions
MK was responsible for writing the article. JL was responsible for designing the protocol and analysing the data. YBH contributed to data extraction and data analysis. JK contributed to data extraction. HYA was responsible for designing the protocol and interpreting the results. JKL was responsible for designing the protocol, interpreting results, and revising the article.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Kim, M., Lee, J., Hwang, Y.B. et al. Graves’ orbitopathy development in thyroid cancer patients: a 16-year nationwide cohort study in South Korea. Eye (2024). https://doi.org/10.1038/s41433-024-03197-9
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41433-024-03197-9