Abstract
Telomerase is associated with cellular aging, and its presence limits cellular lifespan. Telomerase by preventing telomere shortening can extend the number of cell divisions for cancer cells. In adult pancreatic cells, telomeres gradually shorten, while in precancerous lesions of cancer, telomeres in cells are usually significantly shortened. At this time, telomerase is still in an inactive state, and it is not until before and after the onset of cancer that telomerase is reactivated, causing cancer cells to proliferate. Methylation of the telomerase reverse transcriptase (TERT) promoter and regulation of telomerase by lactate dehydrogenase B (LDHB) is the mechanism of telomerase reactivation in pancreatic cancer. Understanding the role of telomeres and telomerase in pancreatic cancer will help to diagnose and initiate targeted therapy as early as possible. This article reviews the role of telomeres and telomerase as biomarkers in the development of pancreatic cancer and the progress of research on telomeres and telomerase as targets for therapeutic intervention.
Similar content being viewed by others
Introduction
Pancreatic cancer is currently the twelfth most common cancer and the seventh most common cause of cancer-related death worldwide [1]. According to statistics in recent years, the number of deaths and the incidence of pancreatic cancer in the world are very close, indicating that the prognosis of the disease is very poor [1]. Pancreatic cancer is usually considered resectable if there is minimal contact with major vessels [2]. If a patient has advanced pancreatic cancer, gemcitabine and albumin combined with paclitaxel or modified FOLFIRINOX are often used as first-line treatment [3, 4]. However, by the time the disease is diagnosed, patients are often in a late stage disease and treatment response is less than ideal [2]. The five-year survival rate is less than 10% [5]. Therefore, researchers have initiated multidisciplinary and interdisciplinary research on the treatment and disease management of pancreatic cancer to help patients improve their quality of life [6]. The pathogenesis of pancreatic cancer still needs further exploration, especially in the fields related to other pancreatic diseases. Many pancreas-related diseases increase the risk of pancreatic cancer, such as hereditary pancreatitis, chronic pancreatitis, diabetes, etc [7,8,9]. These chronic diseases will lead to cell aging and promote pancreatic cancer through some mechanisms that are not yet fully understood.
It has been known for decades that cancer is usually the result of changes in somatic chromosomes [10,11,12,13]. Using next-generation RNA sequencing, researchers found that on average there are 63 mutated genes per tumour. Most of these mutations are point mutations and affect the core set of 12 cell signalling pathways in most tumours, such as apoptosis, DNA damage control, regulation of G1/S phase transition, hedgehog signaling, or other pathways that produce effects such as homophilic cell ashesion, etc [14]. The study showed that the role of the mutated genes varies greatly in different tumours, making targeted personalised therapy essential. A review of 39 studies found that telomerase activity can help doctors differentiate between pancreatitis and pancreatic cancer, and that telomerase plays a key role in malignant cell immortality [15]. Telomeres and telomerase can maintain cell and human vitality, but in cancer cells, they can be detrimental to human health. This means that it can assist tumour cells to proliferate and, because of the nature of tumour cells, their ability to proliferate exceeds that of normal cells, leading to cancer and even death (Fig. 1). In this review, we describe the changes in telomeres and telomerase in different stages of pancreatic cancer. We also introduce the biomarkers associated with telomeres, telomerase and pancreatic cancer, and how telomerase may be targeted for the treatment of pancreatic cancer in the future.
The development of telomerase expression and telomere length are displayed in the context of health and disease. Telomeres are the DNA Protein structure at the end of chromosomes. Telomeres are synthesized by telomerase, and the activity of telomerase in the infant’s pancreas is strong, which promotes the proliferation of healthy pancreatic cells. In adulthood or after illness, exposure to adverse sleep habits, diseases, and other risk factors can induce inflammation, oxidative stress, and cell death [155]. Subsequently, the pancreas maintains its function through cell renewal, but this process can lead to telomere shortening. The shortening of telomeres to a certain length may be the fundamental reason for cell division reaching the Hayflick limit, during which cells undergo replication aging, leading to DNA damage response (DDR) and cell senescence. Before and after pancreatic cancer, telomerase is reactivated, and telomeres are maintained at a certain length, which allows pancreatic cancer cells to escape from replicative senescence and make pancreatic cancer continue to develop.
Function and structure of telomere and telomerase
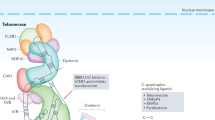
In mammalian cells, including humans, telomere DNA is composed of double-stranded tandem repeats containing TTAGGG, comprising thousands to tens of thousands of bases [16]. These double strands form a loop structure, called a T loop, with a 3’G-rich single-stranded overhang at the end that inserts into the double-stranded DNA and returns to the telomere to form the so-called D loop [17] (Fig. 2). Telomeres are associated with 6 protein subunits, TRF1, TRF2, TPP1, POT1, TIN2 and RAP1, which form a complex called the shelterin complex [18] (Fig. 2). This structure protects telomeres and inhibits telomere damage, while telomeres can protect DNA and prevent DNA from being damaged [18]. In addition to protecting DNA, telomeres can also regulate the pluripotency of stem cells and maintain their homeostasis [19]. All replicating or dividing human somatic cells shorten their telomeres due to the end replication problem (ERC) due to the semiconservative mechanism of DNA replication [20]. Once the telomeres in a cell have shortened to a certain length, the cell begins to undergo replicative senescence or programmed death [21]. All this suggests that the structure and function of telomeres are closely linked to disease, aging and other life processes. By 1999, researchers had established a link between cancer and telomeres [22].
The telomere has a T-loop and D-loop structure, which is divided into 3 ‘and 5’ ends at the end. There is a Shelterin complex on the telomeres, which helps maintain stability. POT1 protection of telomeres 1. RAP1 repressor/activator protein 1. TIN2 TERF1-interacting nuclear factor 2. TPP1 telomere protection protein 1. TRF1 telomeric repeat binding factor 1. TRF2 telomeric repeat binding factor 2.
Human telomerase is mainly composed of two parts, one is called telomerase RNA (TER), and the other is called TERT [23] (Fig. 3). The activity of telomerase mainly depends on the expression of TERT [24]. The human TERT is called hTERT. The conservative domain of TERT has four parts, which are arranged in a linear manner and maintain its biological function. One end is the telomerase essential N-terminal (TEN) domain, the other end is the C-terminal extension (CTE) domain, and the TER binding domain (TRBD), the reverse transcriptase (RT) domain between them [25]. In addition to TERT and TER, the composition of cell telomerase holoenzyme is more complex. The telomerase holoenzyme also includes some cofactors, which have an important impact on the function of telomerase [26,27,28]. The main function of telomerase is to synthesize telomeres, which can maintain a stable length due to the existence of telomerase [29, 30].
The composition of human telomerase is divided into three parts: TERT, TEN, and Cofactor. They combine together to form telomerase, maintaining the length of telomeres. GAR1 nucleolar protein family A. member 1. NHP2 nucleolar protein family A, member 2. NOP10 nucleolar protein 10. TCAB1 telomerase Cajal body protein 1.
Telomeres and precancerous lesions of pancreatic cancer
Pancreatic intraepithelial neoplasia (PanIN) is the most common precursor lesion of pancreatic cancer [31]. In addition, there are two common precursor lesions, namely mucinous cystic neoplasm (MCN) and intraluminal papillary mucinous neoplasm (IPMN) [32]. In PanIN and IPMN, there are many changes in the epithelial cells affected by the disease, one of which is the presence of telomere shortening [33, 34]. Other studies have shown that the telomere-centromere ratio (TRC) decreases as the pathological grade of IPMN increases [35]. This suggests that telomeres become progressively shorter as the disease progresses. Telomere shortening is the most common early genetic abnormality in current pancreatic cancer models, but this telomere shortening is not the direct cause of cancer. The direct cause is likely to be that telomere shortening leads to chromosomal abnormalities, such as fusion of chromosomes, or deletion and insertion of a particular part of the chromosome [36]. The presence of unbalanced chromosomal rearrangements is a necessary condition for most human epithelial cancers [36]. Some studies have shown that many chromosomes in pancreatic cancer often have various abnormalities [37, 38]. Many other studies have linked telomere dynamics to aging, demonstrating that inflammation, epigenetic dysregulation, loss of proteostasis, cellular senescence, stem cell exhaustion, mitochondrial dysfunction caused by telomere dysfunction are almost all signs of aging that will lead to the onset or acceleration of aging [39]. Age-related morphological changes of the pancreas are closely related to the occurrence of pancreatic cancer [40]. Therefore, the development of pancreatic cancer is likely to be caused by age-related telomere shortening and subsequent telomere dysfunction, leading to chromosomal instability (CIN), chromosomal abnormalities and consequently the development of pancreatic cancer [41,42,43]. Some studies suggest that cancer stem cells originate from differentiated tumor cells [44,45,46]. Some researchers have proposed other pathogenic models, suggesting that the onset of cancer occurs due to abnormalities in the self-renewal of normal stem cells, leading to the emergence of tumor stem cells that exhibit increased resistance to drug therapy and apoptosis [47]. This phenomenon partly explains the ineffectiveness of standard chemotherapy and radiotherapy in early-stage pancreatic cancer. It is speculated that this phenomenon may be associated with telomeres and telomerase in pancreatic cancer stem cells (PCSCs). A study reveals that pancreatic cancer stem cells exhibit an elevated telomerase activity in comparison to non-stem pancreatic tumor cells, concurrent with the elongated telomere lengths [48]. The initiation of telomerase function within PCSCs seems intrinsically connected to the aberrant expression of core pluripotency factors, such as SOX2, OCT3/4, KLF4, and NANOG, where the overexpression of any single constituent among these can instigate a collective upregulation of the remaining factors, thereby amplifying telomerase activity within these specific cell type [48]. Therefore, cancer stem cells may maintain their stemness through telomere and telomerase-related mechanisms, facilitating the progression from pancreatic cancer precursors to pancreatic cancer. This continuous proliferation may drive the transition of early-stage pancreatic cancer to advanced-stage pancreatic cancer.
Special mechanism of telomerase reactivation in pancreatic cancer
Chromosome changes
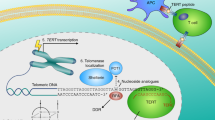
The TERT promoter is altered in many cancers, including melanoma, liposarcoma, hepatocellular carcinoma, hepatocellular carcinoma and urothelial carcinoma. However, some studies have found that mutations in the TERT promoter are rare in pancreatic cancer cases [49,50,51]. Different from other cancers, pancreatic cancer may originate from chromosome changes. In 2021, The research team of Yagyu et al. found that a gene in the 3p21.3 chromosome region inhibits TERT by inhibiting the activity of the TERT promoter, thereby inhibiting the invasion and proliferation of pancreatic cancer cells, thus achieving the effect of inhibiting cancer [24] (Fig. 4). The probability of detecting loss of heterozygosity in this region in other cancers has reached 75%, but it has not been established that this alteration is associated with the development of pancreatic cancer [24]. Therefore, at present, the development of pancreatic cancer may be due to a different mechanism, namely methylation of the TERT promoter. In 2009, an Indian study found that methylation of hTERT promoter and p16 promoter was positively correlated with hTERT expression and telomerase activity by comparing pancreatic cancer tissue with adjacent normal tissue [52]. The researchers in this study also found that the use of 5-azacytidine, a demethylating agent, in pancreatic tissue resulted in a decrease in hTERT gene expression, leading to a decrease in telomerase activity. Demethylation of 5-azacytidine leads to hTERT inhibition, which can reduce telomerase activity to 37–49% of the control group [53]. Therefore, it is likely that hTERT promoter methylation regulates the expression of hTERT and then regulates telomerase activity, thereby affecting the occurrence and development of pancreatic cancer.
Current research indicates that in a 433 bp genomic region known as the TERT hypermethylated tumor region (THOR), located at the 52 CpG sites upstream of the TERT core promoter. It has been observed that unmethylated THOR exerts an inhibitory effect on TERT promoter activity, whereas extensive methylation of THOR counteracts this inhibitory function [54]. Some researchers speculate that the THOR sequence may contain multiple binding sites for transcription inhibitors, such as WT1 and MZF2 binding sites [55]. In highly methylated THOR, these sites may be unable to inhibit factor binding, potentially leading to the abnormal upregulation of TERT [56]. Further research is needed to elucidate the precise mechanisms underlying this phenomenon.
The presence of LDHB
The sustained proliferation of cancer cells is related to telomere length, which is usually associated with telomerase activity. The maintenance of telomerase activity requires certain conditions. In 2022, a cellular study conducted by Wang et al. showed that regardless of its metabolic pathway, LDHB is essential for maintaining telomere length and promoting telomerase activity [57] (Fig. 4). Knocking out LDHB can inhibit tumour cell proliferation, telomerase activity and reduce telomere length in both in vivo and in vitro experiments [57]. The team also found that LDHB was negatively correlated with the disease-free survival (DFS) of patients by detecting pancreatic cancer tissue and adjacent positive tissue [57]. This is somewhat different from the research of Cui et al. In 2015, Cui et al. showed that LDHB in pancreatic cancer tissues was higher than that in normal adjacent tissues, and inhibiting the expression of LDHB could promote the growth and development of pancreatic cancer [58]. This growth promotion may be caused by an increase in glycolysis, and LDHB is an important enzyme in this pathway. Previous studies have shown that LDHB is an important enzyme in the glycolytic pathway. LDHB can also activate tumor cell lysosomes and perform autophagy through lysosomes, thereby regulating metabolism when tumor cells are under nutrition [59]. However, Cui et al’s study found that the mechanism by which LDHB prolongs telomeres is mainly due to the direct interaction between LDHB and TERT to regulate telomerase, and no association has been found between LDHB and its role in glycolysis [57].
The Interaction between GOLPH3 and STIP1
Golgi phosphoprotein 3 (GOLPH3) is a highly conserved cytosolic trans-Golgi associated protein [60]. The study by Sun et al. found that overexpression of GOLPH3 significantly increased the expression of β‐catenin, c‐Myc, and cyclin‐D1 [61]. However, this study was found in ovarian cancer cells, so it still needs to be further verified in pancreatic cancer cells. Stress-inducible protein-1 (STIP1) is a novel GOLPH3 binding partner [62]. STIP1 can promote the proliferation of pancreatic cancer cells and accelerate the cell cycle [62]. Telomerase activity is regulated by hTERT, which is activated by c-Myc through the E-box [63] (Fig. 4). Studies have shown that inhibition of hTERT can inhibit cyclin D1 signalling in cancer cells and also inhibit the cell cycle [64, 65]. When GOLPH3 and STIP1 are knocked down, they can not only inhibit the proliferation of pancreatic cancer cells, but also inhibit the expression of cyclin D1 in cancer cell [62]. This study also showed that the overexpression of GOLPH3 was positively correlated with the expression of STIP1 in pancreatic cancer tissue, but the knockout of GOLPH3 did not affect the level of STIP1 [62].
Telomere and telomerase as biomarkers
Biological markers, also known as biomarkers, are objectively measurable and evaluable indicators of certain biological states in normal and pathogenic processes, or possible pharmacologic responses to therapeutics [66]. More importantly, they can not only predict the occurrence or progression of diseases at the expression level, but also potentially indicate dynamic changes in biological processes or state [67].
Due to the critical role of telomeres and telomerase in the onset and development of cancer, an increasing number of studies have targeted telomeres and telomerase as biomarkers, resulting in the acquisition of many meaningful methods for detecting biomarker. These methods can be examined through blood, faeces, urine and other means without invasive testing [68] (Table 1). This is very easy to implement for many doctors and patients. These biomarkers can help doctors distinguish pancreatic cancer from other pancreatic diseases, and predict the incidence rate and prognosis of pancreatic cancer through telomere length and telomerase activity.
In 1996, Mizumoto et al. compared human pancreatic cancer duct samples and the pancreatic cancer cell line MIA with normal pancreatic ducts and found that telomerase activity may be a specific marker for pancreatic duct cancer [69]. A year later, Tsutsumi et al. found an increase in telomerase activity in 32 out of 38 pancreatic ductal cancer samples compared to the surrounding tissue of the tumour [70]. However, no such change was found in the tissue surrounding the tumour. In 1998, Suehara et al. found that telomerase activity increased in the pancreatic juice of a patient, but no tumour was found in various examinations. After 19 months, the patient developed pancreatic cancer. This led the research team to suggest that telomerase activity in pancreatic juice could be a marker for the early diagnosis of cancer [71]. Subsequently, in 1999, researchers used telomerase activity as a biological indicator in patients with malignant and precancerous pancreatic cystic tumours, benign tumours and pseudocysts and found that its specificity was 67%, sensitivity was 100% and overall accuracy was 86% [72]. In the same year, cell experiments and clinical trials found that the higher the telomerase activity, the more invasive pancreatic cancer could be [73, 74]. As research deepened, in 2004 researchers found that the serum of pancreatic cancer patients could also be used for related tests to help diagnose pancreatic cancer [75]. In a study of 17 pancreatic cancer patients and 12 chronic pancreatitis patients, telomerase was found to be elevated in 88% of pancreatic cancer patients and 17% of chronic pancreatitis patients [76]. At present, it is not possible to distinguish whether elevated telomerase is caused by pancreatic cancer or chronic pancreatitis.
In addition to telomerase activity, the alteration of TERT also demonstrates its value as a biomarker. Research has shown that the hypermethylation region within the hTERT promoter is associated with upregulation of hTERT in cancers expressing hTERT, hence this region is called the TERT hypermethylation tumour region (THOR) [77]. A study in 2021 found that patients with higher levels of THOR methylation had shorter overall survival (OS) and recurrence-free survival (RFS) compared to patients with lower levels of THOR methylation, based on analysis of data from The Cancer Genome Atlas (TCGA) database [78]. In 2022, researchers also found that hTERT is phosphorylated at threonine 249 (Thr249) by the serine/threonine kinase CDK1 [79]. This phosphorylation is a molecular switch for hTERT to display RNA-dependent RNA polymerase (RdRP) activity, and the activity of hTERT RdRP can prevent the expression of some tumour suppressor genes, such as Forkhead box O4 (FOXO4), which can lead to pancreatic and liver cancer [79]. Through a study of 1523 cases of lung cancer, colon cancer, stomach cancer, pancreatic cancer, liver cancer, breast cancer and kidney cancer, it is concluded that the phosphorylation of hTERT at threonine 249(p-hTERT), which promotes the activity of RdRP, is a strong risk factor independent of TNM(Tumor Node Metastasis) staging and can be used as a biomarker for prognostic stratification of pancreatic cancer, lung cancer and other cancers [79].
In terms of telomere research, a study of 331 pathologies in 10 European countries showed that cell telomere length could not predict the incidence of pancreatic cancer [80]. In the study by Campa et al, 116 out of 26540 samples had been diagnosed with pancreatic cancer during an average follow-up of 12.8 years [81]. According to statistical analysis, longer leukocyte telomeres were significantly associated with the risk of pancreatic cancer [81]. An American study of 642 pancreatic cancer samples found that the telomere length of lymphocytes in white blood cells was negatively correlated with age at diagnosis [82]. The shorter telomeres, the shorter the survival, and the shorter the telomere, the higher the mortality [82]. Although telomere length is closely related to pancreatic cancer, statistical studies have shown that through gene discovery in 1500 pancreatic cancer patients and 1500 non-cancer patients, the genes regulating telomere length of leukocytes between the two groups cannot currently predict the incidence of pancreatic cancer, indicating that expression of genes regulating telomere length is related to many complex factors and needs further research [83]. Because first-line chemotherapy treatment affects the length of leukocyte telomeres, the research team also looked at the relationship between the length of leukocyte telomeres and the risk of pancreatic cancer in untreated patients [84]. The results showed that short leukocyte telomeres in untreated patients were associated with a high risk of pancreatic cancer [84]. These results suggest that when using leukocyte telomere length (LTL) as a biomarker, other confounding factors such as body size, diet, age and other stratified comparisons should be considered, which may help to provide more accurate predictive methods. One study suggested that the risk of pancreatic cancer could be assessed using the telomere score of lymphocytes. This study first looked at 11 genes that could affect telomere length and how many genes were associated with longer telomeres. A weighted calculation produces a number between 0 and 20. The higher the value, the longer the telomere, while the lower the value, the shorter the telomere. Finally, 2374 patients and 4326 controls showed that the higher the telomere score, the lower the risk of cancer [85]. Chinese scientists used telomere length variation (TLV) as an indicator to predict pancreatic cancer, which represents the heterogeneous telomere length at the ends of all chromosomes [86]. The white blood cells in the peripheral blood of 900 pancreatic cancer cases and controls were measured. It was found that an increase in TLV was associated with an increased risk of pancreatic cancer [86]. Another study predicted the occurrence of pancreatic cancer by detecting telomere fusion. This study suggested that telomere shortening would lead to telomere fusion and then telomerase activation, allowing pancreatic cancer cells to replicate indefinitely [87].
Telomere and telomerase therapies
As research into the relationship between telomeres, telomerase and various cancers gradually deepens, researchers suggest that treating cancer through telomerase inhibition is a promising research approach. Starting from this pathway may help us to understand and treat various cancers [88,89,90,91]. There are two main types of treatment using telomerase as a pathway: immunotherapy and telomerase inhibitor therapy [92,93,94] (Fig. 5). The drugs used for immunotherapy are the TERT peptide vaccine and oncolytic virus [92, 95]. Drugs used for direct inhibition of telomerase include oligonucleotide inhibitors, small molecule inhibitors and natural products [93, 96, 97]. Indirect inhibitors of telomerase are mainly divided into G-quadruplex stabilisers, nucleoside analogues and telomere uncapping agents [94, 98, 99].
A Telomerase vaccines can recognize TERT peptides on the surface of cells with high TERT levels, thereby promoting the killing of CD4 + T and CD8 + T cells. The oncolytic virus can selectively replicate and lyse cells in cells activated by telomerase. B Small molecule inhibitor BIBR1532 can bind to TERT or TER, prevent the binding between TERT and TER, and inhibit the formation of telomerase. C The oligonucleotide inhibitor imetelstat can compete with telomeres and bind to TER, thereby preventing the binding between telomeres and telomerase. D CDDO Me can weaken hTERT mRNA, basal hTERT, phosphorylated hTERT, and methylation of hTERT promoter, thereby reducing telomerase activity. E G-quadruplex stabilizers can stabilize the G-quadruplex structure of telomeres, prevent DNA helicase from degrading them, and block the telomere elongation process.
Immunotherapy
In immunotherapy, one of the TERT vaccines currently in clinical research for pancreatic cancer is called GV1001. This treatment targets cells with activated telomerase. As telomerase is often activated in pancreatic cancer cells, this treatment is likely to be one of the most important ways of treating pancreatic cancer in the future. In a study of 38 patients with inoperable pancreatic cancer in Norway, researchers used GV1001 and granulocyte macrophage colony-stimulating factor in patients, and the survival of 24 patients who responded was significantly prolonged, showing the drug’s therapeutic potential [100]. The efficacy of GV1001 is likely due to its ability to induce CD4 + T cells and CD8 + T cells in the immune system to mount an appropriate immune response, leading to a strong inflammatory response against tumours or tumour-draining lymph nodes, thereby achieving the effect of inhibiting tumours and prolonging patient survival [100]. Another UK study showed that the combination of GV1001 and chemotherapy did not improve the survival rate of locally advanced or metastatic pancreatic cancer patients, suggesting that the clinical potential of GV1001 needs further investigation [101]. At the same time, researchers have also begun to study the therapeutic strategy of combining telomerase vaccines with chemotherapy. In addition to the treatment of pancreatic cancer, GV1001 has also been developed for the treatment of other cancers, including melanoma, small cell lung cancer, prostate cancer, colorectal cancer, renal cell cancer, etc [102,103,104,105,106]. Its specific anticancer mechanisms include inhibition of HIF-1α、regulation of VEGF/VEGFR-2 signalling pathway that inhibits angiogenesis and selectively stimulates the Gαs/cAMP pathway that inhibits cancer cell proliferation, and so on. However, these cannot fully explain its mechanism and researchers need to do more research.
The other two vaccines used in pancreatic cancer research are INO-1400 and INO-1401. The difference between the two vaccines is that the former is modified to have 99% similarity to human TERT and the latter is further modified to have 95% similarity to human TERT than the former [107]. This modification may enhance the efficacy of the vaccine and help improve tolerance. The researchers treated pancreatic cancer with or without INO-9012. INO-1401 and INO-1401 are synthetic DNA plasmids encoding modified human telomerase protein, while INO-9012 consists of DNA plasmids encoding synthetic human IL-12 (p35 and p40 subunits) [107]. Following treatment, researchers observed an upregulation of CD38 on hTERT-specific CD4+ and CD8 + T cells in patients, as well as a survival benefit in patients [107].
In oncolytic virus research, there is a type of oncolytic virus called OBP-502 [108]. When telomerase is activated, its hTERT is often also in an activated state, and the activation of hTERT drives the expression of oncolytic virus E1A and E1B genes, allowing the oncolytic virus to selectively replicate in tumor cells, ultimately leading to tumor cell death. The virus then continues to replicate in surrounding cells, eliminating tumour cells. In cellular and animal studies, OBP-502 induced the release of ICD molecules from CT26 and PAN02 cells, leading to recruitment of CD8+ lymphocytes and inhibition of Foxp3+ lymphocyte infiltration into tumors [108]. Although its variant OBP-301 has entered clinical research, it may be some time before OBP-502 enters clinical practice.
Among the small molecule inhibitors, there is a non-competitive small molecule inhibitor called BIBR1532 [109]. It can disrupt the binding of TERT and TER, thereby reducing telomerase activity. In vitro experiments show that BIBR1532 can not only cause DNA damage to pancreas cancer samples and promote cancer stem cells(CSC) apoptosis, but also reduce the expression of TERT, prevent the number of CSCs in organoid culture medium and thus prevent them from forming spheres [48]. The augmented telomerase activity in CSCs is highly likely associated with the activation and mutation of TERT promoter, whereupon its activation or mutation leads to an increase in TERT activity, thus culminating in heightened telomerase function [49]. When BIBR1532 interacts with TERT within CSCs, it serves to inhibit telomerase activity, a mechanism that ultimately depletes the CSC [48]. Given the poor pharmacokinetic properties of this drug, it is difficult to say whether it can be used in clinical practice. However, in the future, new drugs may be developed and applied in clinical practice based on the mechanism of their pharmacological effects [95].
Oligonucleotide inhibitor
In oligonucleotide inhibitor research, current research shows that ten strains of pancreatic cancer cells (L3.6pl, MiaPaCa2, HPAF, AsPC1, CD18, Panc1, Hs766T, CFPAC1, CAPAN1, CAPAN2) all respond to the oligonucleotide inhibitorimetelstat (GRN163L) [110]. Its mechanism of action is mainly to block the binding between telomeres and telomerase, thereby inhibiting the action of telomerase on telomeres [111]. However, for unknown reasons, sensitivity toimetelstat varies between different cell lines, resulting in varying degrees of telomerase inhibition. And when the drug was discontinued after it had worked, it was found that the drug continued to work for up to three weeks after discontinuation. This suggests that in future clinical practice, patients may be able to avoid taking the drug for a period of time after it has taken effect to reduce the occurrence of side effects [110]. Three weeks later, telomerase inhibition disappeared, and telomeres of pancreatic cancer cells were prolonged, γ- H2AX decreased again to the level when it was not administered, which confirmed the relationship among these factors in cancer. In addition, this experiment also showed the research potential ofimetelstat in the treatment of pancreatic cancer [110]. Research by Joseph et al. shows thatimetelstat can reduce the number of CSCs in both pancreatic and breast cancer cells, thereby inhibiting tumour cell growth and preventing tumours from progressing [112]. In the study of oesophageal cancer, long-term use of imetelstat therapy also inhibits telomerase activity, which not only reduces the proliferation of oesophageal cancer cells, but also induces increased DNA double-strand breaks (DSBs). after irradiation, which is helpful in the delivery of radiotherapy [113]. In studies of non-small cell lung cancer, imetelstat has an effect on different genetic characteristics and cell lines, and cells with shorter telomeres respond more quickly to imetelstat [114]. In clinical trials involving non-small cell lung cancer (NSCLC) patients treated with imetelstat, only individuals with shorter telomeres exhibited a propensity towards enhanced progression-free survival (PFS) and OS. Conversely, patients with longer telomeres did not exhibit statistically significant improvements in these clinical endpoints following imetelstat treatment. This suggests that there is still a long way to go in the clinical development of imetelstat [115].
Inhibitors of natural compounds
In a study of natural compound, Deeb et al. found that the natural compound derivative methyl-2-cyano-3,12-dioxoene-1,9 (11) - dien-28-ester (CDDO-Me) can inhibit the proliferation of pancreatic cancer cells and promote their apoptosis at very low concentrations [116]. In in vitro experiments, CDDO-Me can attenuate hTERT mRNA, basal hTERT and phosphorylated hTERT, as well as telomerase activity, and inhibit hTERT promoter methylation [116]. The specific mechanism may be related to the core promoter of hTERT. In the experiment, it was found that CDDO-Me inhibited the activity of Sp1, c-Myc, and NF-κB in Panc-1 and MiaPaCa-2 cells [116]. These are binding sites of the core promoter of hTERT [117,118,119]. In the study by Deeb et al., it was also found that CDDO Me inhibits the protein levels of DNA methyltransferases DNMT1 and DNMT3a, leading to hypomethylation of the hTERT promoter [116]. Other studies have shown that the inhibition of telomerase activity by CDDO me can be mediated through ROS dependent pathways [120]. Gao et al.‘s study showed that CDDO-Me can inhibit tumor growth and prolong survival after tumor resection in animal experiments [121]. All this suggests that CDDO-Me is a potential drug for the treatment of pancreatic cancer in future, but clinical trials are still needed to verify this.
G-quadruplex
G-quadruplex is a quadruplex consisting of four guanine and hydrogen bonds on DNA and RNA [122]. DNA helicase is required to break this structure prior to telomere elongation [123]. This structural change can upregulate or inhibit telomerase binding, and current research suggests that the stability of the G-quadruplex may help inhibit telomerase activity [124]. The 3’ end of telomeres binds to a protein known as POT, which plays a crucial role in preserving the stability of G-quadruplex and preventing its interaction with hTR [125]. This ultimately leads to the inhibition of telomerase activity [125]. Therefore, researchers in related fields are attempting to develop a ligand to make the G-quadruplex more stable. One of the G-quadruplex ligand drugs, BMSG-SH-3, has shown good therapeutic potential in animal studies of pancreatic cancer [126]. The mice did not lose weight during treatment, but tumour growth was reduced by 50% and telomerase activity was reduced by about 50%. And this drug is concentrated in tumours and the pancreas, and is less present in other organs [126]. This drug can either replace POT1 at the end of the telomere and binds to the single-stranded telomere DNA overhang, thereby inhibiting the telomerase activity [126, 127]. In addition, some G-quadruplex stabilisers such as naphthalene diimide, MM41, porphyrin-1 (cobalt-containing) and porphyrin-2 (palladium-containing) have also shown good experimental effects and clinical prospects [128,129,130].
Potential issues
Although anti-telomerase cancer therapies have demonstrated encouraging research prospects, several potential concerns deserve careful consideration. Research findings suggest that the action of telomerase inhibitors on cancer cells may require a certain time frame to effectively shorten telomeres to a critical length, necessitating adequate periods of drug exposure for efficacious treatment [131]. However, during this interval, patients could potentially succumb to the progression of their cancer [132]. Consequently, there remains a compelling need to investigate more rapidly acting medications that can expedite the reduction in drug exposure times, thereby improving patient outcomes. Given that telomerase is typically not expressed in normal human somatic cells, and the fact that telomeres in normal stem cells are generally longer than those found in cancer cells, the anticipated impact of telomerase inhibitors on healthy cells is expected to be minimal [133]. Nonetheless, we cannot ignore the potential detrimental effects that telomerase inhibition may exert on certain cells, such as stem cells, which rely on telomerase activity during their proliferation process [134]. Particularly as the duration of treatment extends, the potential risks associated with telomerase inhibitors on normal cells may escalate concomitantly [135]. Additionally, telomeres serve as protective caps that maintain genomic stability in both normal and neoplastic cells. The suppression of telomerase activity in tumor cells results in telomere erosion, which could precipitate genomic instability within these malignantly transformed cells. This instability may further contribute to their carcinogenic progression. As such, the use of telomerase inhibitors necessitates careful and thorough assessment of the disease state to mitigate the risk of inadvertently promoting detrimental consequences. Although the disabling of telomerase activity leads to a brief slowdown in tumor growth. The therapeutic approach targeting telomerase inhibition may not be comprehensive, as telomere elongation is not solely dependent on the activation of telomerase. Telomeres can also be maintained through an alternative lengthening mechanism known as alternative lengthening of telomeres (ALT), which could potentially allow cancer cells to bypass the effects of telomerase inhibitors and sustain their telomere length. A subsequent resurgence in tumor proliferation occurred as these neoplasms adapted by adopting the ALT mechanism and aberrant transcriptional networks centering on mitochondrial biology and oxidative defense [136]. However, at present, the activation of ALT pathways in cancers appears relatively infrequent; the majority of malignancies primarily rely on modulating telomerase activity for telomere maintenance, with this ALT phenotype being observed in only approximately 10–15% of tumors [137]. Consequently, the preponderance of scientific attention has been directed towards the development and investigation of telomerase inhibitors as a therapeutic strategy.
Conclusion
Telomeres and telomerase are closely linked to the occurrence and development of pancreatic cancer. The detection of telomerase can differentiate pancreatic cancer from other pancreatic diseases. Telomere length can also predict patient prognosis. Mutations in related genes can cause changes in telomeres and telomerase, reducing the body’s inhibition of cancer cells and promoting the proliferation and metastasis of pancreatic cancer cells. Many related physiological and pathological changes still need to be elucidated. The efficacy of telomerase inhibitor-based treatments also needs to be further investigated and their mechanisms of action explored. We can discover more accurate biomarkers based on research results, help researchers better design prospective studies, and pave the way for research into new diagnoses and treatments. This will allow medical researchers to develop better telomerase inhibitors and treat pancreatic cancer as safely as possible. In addition, research into telomeres and telomerase may not only help shorten the lifespan of tumours in humans, but also potentially extend the lifespan of patients with other diseases. This will have huge benefits for human health.
References
Sung H, Ferlay J, Siegel RL, Laversanne M, Soerjomataram I, Jemal A, et al. Global cancer statistics 2020: globocan estimates of incidence and mortality worldwide for 36 cancers in 185 countries. Ca: A Cancer J Clin. 2021;71:209–49. https://doi.org/10.3322/caac.21660.
Park W, Chawla A, O’Reilly EM. Pancreatic cancer: a review. JAMA. 2021;326:851–62. https://doi.org/10.1001/jama.2021.13027.
Von Hoff DD, Ervin T, Arena FP, Chiorean EG, Infante J, Moore M, et al. Increased survival in pancreatic cancer with nab-paclitaxel plus gemcitabine. N Engl J Med. 2013;369:1691–703.
Vaccaro V, Sperduti I, Milella M. Folfirinox versus gemcitabine for metastatic pancreatic cancer. N Engl J Med. 2011;365:768–9. https://doi.org/10.1056/NEJMc1107627.
Siegel RL, Miller KD, Fuchs HE, Jemal A. Cancer statistics, 2021. Ca: A Cancer J Clin. 2021;71:7–33. https://doi.org/10.3322/caac.21654.
Moffat GT, Epstein AS, O’Reilly EM. Pancreatic cancer-a disease in need: optimizing and integrating supportive care. Cancer. 2019;125:3927–35. https://doi.org/10.1002/cncr.32423.
Canto MI, Almario JA, Schulick RD, Yeo CJ, Klein A, Blackford A, et al. Risk of neoplastic progression in individuals at high risk for pancreatic cancer undergoing long-term surveillance. Gastroenterology. 2018;155:740–51.
Vasen HF, Wasser M, Van Mil A, Tollenaar RA, Konstantinovski M, Gruis NA, et al. Magnetic resonance imaging surveillance detects early-stage pancreatic cancer in carriers of a p16-leiden mutation. Gastroenterology. 2011;140:850–6.
Canto MI, Harinck F, Hruban RH, Offerhaus GJ, Poley J, Kamel I, et al. International cancer of the pancreas screening (caps) consortium summit on the management of patients with increased risk for familial pancreatic cancer. Gut. 2013;62:339–47.
Rowley JD, Golomb HM, Dougherty C. 15/17 translocation, a consistent chromosomal change in acute promyelocytic leukaemia. Lancet (Lond, Engl). 1977;1:549–50. https://doi.org/10.1016/s0140-6736(77)91415-5.
Rowley JD, Golomb HM, Vardiman J, Fukuhara S, Dougherty C, Potter D. Further evidence for a non-random chromosomal abnormality in acute promyelocytic leukemia. Int J Cancer. 2010;20:869–72.
Sundaresan V, Heppell-Parton A, Coleman N, Miozzo M, Sozzi G, Ball R, et al. Somatic genetic changes in lung cancer and precancerous lesions. Ann Oncol: Off J Eur Soc Med Oncol. 1995;27-31:31–2. https://doi.org/10.1093/annonc/6.suppl_1.s27.
Tirkkonen M, Kainu T, Loman N, Jóhannsson OT, Olsson H, Barkardóttir RB, et al. Somatic genetic alterations in brca2-associated and sporadic male breast cancer. Genes Chromosomes Cancer. 1999;24:56–61. https://doi.org/10.1002/(sici)1098-2264(199901)24:1<56::aid-gcc8>3.0.co;2-x.
Jones S, Zhang X, Parsons DW, Lin JC, Leary RJ, Angenendt P, et al. Core signaling pathways in human pancreatic cancers revealed by global genomic analyses. Science (N. Y, N. Y). 2008;321:1801–06. https://doi.org/10.1126/science.1164368.
Hata T, Ishida M, Motoi F, Yamaguchi T, Naitoh T, Katayose Y, et al. Telomerase activity in pancreatic juice differentiates pancreatic cancer from chronic pancreatitis: a meta-analysis. Pancreatol: Off J Int Assoc Pancreatol. 2016;16:372–81. https://doi.org/10.1016/j.pan.2016.01.007.
Griffith JD, Comeau L, Rosenfield S, Stansel RM, Bianchi A, Moss H, et al. Mammalian telomeres end in a large duplex loop. Cell. 1999;97:503–14.
Palm W, Lange TD. How shelterin protects mammalian telomeres. Annu Rev Genet. 2008;42:301–34.
Lange TD. Shelterin-mediated telomere protection. Annu Rev Genet. 2018;52:223–47.
Huang J, Wang F, Okuka M, Liu N, Ji G, Ye X, et al. Association of telomere length with authentic pluripotency of es/ips cells. Cell Res. 2011;21:779–92.
Duell EJ. Telomere length and pancreatic cancer risk: breaking down the evidence. Gut. 2017;66:1. https://doi.org/10.1136/gutjnl-2016-313156.
Stewart SA, Weinberg RA. Telomeres: cancer to human aging. Annu Rev Cell Dev Biol. 2006;22:531–57. https://doi.org/10.1146/annurev.cellbio.22.010305.104518.
Hastie ND, Dempster M, Dunlop MG, Thompson AM, Green DK, Allshire RC. Telomere reduction in human colorectal carcinoma and with ageing. Nature. 1990;346:866–8. https://doi.org/10.1038/346866a0.
Lu W, Zhang Y, Liu D, Zhou S, Wan M. Telomeres-structure, function, and regulation. Exp Cell Res. 2013;319:133–41.
Yagyu T, Ohira T, Shimizu R, Morimoto M, Murakami Y, Hanaki T, et al. Human chromosome 3p21.3 carries tert transcriptional regulators in pancreatic cancer. Sci Rep. 2021;11:15355. https://doi.org/10.1038/s41598-021-94711-6.
Nguyen T. Structural biology of human telomerase: progress and prospects. Biochem Soc Trans. 2021;49:1927–39. https://doi.org/10.1042/BST20200042.
Collins KL. The biogenesis and regulation of telomerase holoenzymes. Nat Rev Mol Cell Biol. 2006;7:484–94.
Egan ED, Collins K. Biogenesis of telomerase ribonucleoproteins. Rna-a Publ Rna Soc. 2012;18:1747.
Cech TR, Schmidt JC. Human telomerase: biogenesis, trafficking, recruitment, and activation. Genes Dev a J Devoted Mol Anal Gene Expr Eukaryotes Prokaryotes Viruses. 2015;29:1095–105.
Verdun RE, Karlseder J. Replication and protection of telomeres. Nature. 2007;447:924–31. https://doi.org/10.1038/nature05976.
Collins K, Mitchell JR. Telomerase in the human organism. Oncogene. 2002;21:564–79. https://doi.org/10.1038/sj.onc.1205083.
Søreide K, Marchegiani G. Clinical management of pancreatic premalignant lesions. Gastroenterology. 2022;162:379–84. https://doi.org/10.1053/j.gastro.2021.09.073.
Remmers N, Bailey JM, Mohr AM, Hollingsworth MA. Molecular pathology of early pancreatic cancer. Cancer Biomark: Sect a Dis Markers. 2010;9:421–40. https://doi.org/10.3233/CBM-2011-0168.
Hashimoto Y, Murakami Y, Uemura K, Hayashidani Y, Sudo T, Ohge H, et al. Telomere shortening and telomerase expression during multistage carcinogenesis of intraductal papillary mucinous neoplasms of the pancreas. J Gastrointest Surg: Off J Soc Surg Alimentary Trac. 2008;12:17–28. https://doi.org/10.1007/s11605-007-0383-9.
Singh AP, Moniaux N, Chauhan SC, Meza JL, Batra SK. Inhibition of muc4 expression suppresses pancreatic tumor cell growth and metastasis. Cancer Res. 2004;64:622–30. https://doi.org/10.1158/0008-5472.can-03-2636.
Miki A, Matsuda Y, Aida J, Watanabe J, Sanada Y, Sakuma Y, et al. Telomere attrition in intraductal papillary mucinous neoplasms of the pancreas associated with carcinogenesis and aging. Pancreas. 2022;51:678–83. https://doi.org/10.1097/MPA.0000000000002081.
van Heek NT, Meeker AK, Kern SE, Yeo CJ, Lillemoe KD, Cameron JL, et al. Telomere shortening is nearly universal in pancreatic intraepithelial neoplasia. Am J Pathol. 2002;161:1541–7. https://doi.org/10.1016/S0002-9440(10)64432-X.
Griffin CA, Hruban RH, Morsberger LA, Ellingham T, Long PP, Jaffee EM, et al. Consistent chromosome abnormalities in adenocarcinoma of the pancreas. Cancer Res. 1995;55:2394–9.
Hruban RH, Wilentz RE, Goggins M, Offerhaus GJ, Yeo CJ, Kern SE. Pathology of incipient pancreatic cancer. Ann Oncol: Off J Eur Soc Med Oncol. 1999:9–11. https://doi.org/10.1023/a:1008359929858.
Chakravarti D, Labella KA, Depinho RA. Telomeres: history, health, and hallmarks of aging. Cell. 2021;184:306–22. https://doi.org/10.1016/j.cell.2020.12.028.
Matsuda Y. Age-related morphological changes in the pancreas and their association with pancreatic carcinogenesis. Pathol Int. 2019;69:450–62. https://doi.org/10.1111/pin.12837.
Meeker AK, Gage WR, Hicks JL, Simon I, Coffman JR, Platz EA, et al. Telomere length assessment in human archival tissues: combined telomere fluorescence in situ hybridization and immunostaining. Am J Pathol. 2002;160:1259–68. https://doi.org/10.1016/S0002-9440(10)62553-9.
Artandi SE, Chang S, Lee SL, Alson S, Gottlieb GJ, Chin L, et al. Telomere dysfunction promotes non-reciprocal translocations and epithelial cancers in mice. Nature. 2000;406:641–5. https://doi.org/10.1038/35020592.
Hackett JA, Greider CW. Balancing instability: dual roles for telomerase and telomere dysfunction in tumorigenesis. Oncogene. 2002;21:619–26. https://doi.org/10.1038/sj.onc.1205061.
Santisteban M, Reiman JM, Asiedu MK, Behrens MD, Nassar A, Kalli KR, et al. Immune-induced epithelial to mesenchymal transition in vivo generates breast cancer stem cells. Cancer Res. 2009;69:2887–95. https://doi.org/10.1158/0008-5472.CAN-08-3343.
Mani SA, Guo W, Liao MJ, Eaton EN, Ayyanan A, Zhou AY, et al. The epithelial-mesenchymal transition generates cells with properties of stem cells. Cell. 2008;133:704–15. https://doi.org/10.1016/j.cell.2008.03.027.
Morel AP, Lièvre M, Thomas C, Hinkal G, Ansieau S, Puisieux A, et al. Generation of breast cancer stem cells through epithelial-mesenchymal transition. Plos One. 2008;3:e2888.
Al-Hajj M, Clarke MF. Self-renewal and solid tumor stem cells. Oncogene. 2004;23:7274–82. https://doi.org/10.1038/sj.onc.1207947.
Walter K, Rodriguez-Aznar E, Ferreira M, Frappart PO, Dittrich T, Tiwary K, et al. Telomerase and pluripotency factors jointly regulate stemness in pancreatic cancer stem cells. Cancers (Basel). 2021;13. https://doi.org/10.3390/cancers13133145.
Vinagre J, Almeida A, Pópulo H, Batista R, Lyra J, Pinto V, et al. Frequency of tert promoter mutations in human cancers. Nat Commun. 2013;4:2185.
Killela PJ, Reitman ZJ, Jiao Y, Bettegowda C, Agrawal N, Diaz LA Jr, et al. Tert promoter mutations occur frequently in gliomas and a subset of tumors derived from cells with low rates of self-renewal. Proc Natl Acad Sci. 2013;110:6021–6.
Huang D, Wang Z, He X, Diplas BH, Yang R, Killela PJ, et al. Recurrent tert promoter mutations identified in a large-scale study of multiple tumour types are associated with increased tert expression and telomerase activation. Eur J Cancer. 2015;51:969–76.
Kumari A, Srinivasan R, Vasishta RK, Wig JD. Positive regulation of human telomerase reverse transcriptase gene expression and telomerase activity by dna methylation in pancreatic cancer. Ann Surg Oncol. 2009;16:1051–9. https://doi.org/10.1245/s10434-009-0333-8.
Kumari A, Srinivasan R, Wig JD. Effect of c-myc and e2f1 gene silencing and of 5-azacytidine treatment on telomerase activity in pancreatic cancer-derived cell lines. Pancreatol: Off J Int Assoc Pancreatol. 2009;9:360–8. https://doi.org/10.1159/000212094.
Lee DD, Leão R, Komosa M, Gallo M, Zhang CH, Lipman T, et al. Dna hypermethylation within tert promoter upregulates tert expression in cancer. J Clin Investig. 2019;129:223–9. https://doi.org/10.1172/JCI121303.
Kyo S, Takakura M, Fujiwara T, Inoue M. Understanding and exploiting htert promoter regulation for diagnosis and treatment of human cancers. Cancer Sci. 2008;99:1528–38. https://doi.org/10.1111/j.1349-7006.2008.00878.x.
Lee DD, Komosa M, Nunes NM, Tabori U. Dna methylation of the tert promoter and its impact on human cancer. Curr Opin Genet Dev. 2020;60:17–24. https://doi.org/10.1016/j.gde.2020.02.003.
Wang R, Li J, Zhang C, Guan X, Qin B, Jin R, et al. Lactate dehydrogenase b is required for pancreatic cancer cell immortalization through activation of telomerase activity. Front Oncol. 2022;12:821620. https://doi.org/10.3389/fonc.2022.821620.
Cui J, Quan M, Jiang W, Hu H, Jiao F, Li N, et al. Suppressed expression of ldhb promotes pancreatic cancer progression via inducing glycolytic phenotype. Med Oncol (Northwood, Lond, Engl). 2015;32:143. https://doi.org/10.1007/s12032-015-0589-8.
Brisson L, Bański P, Sboarina M, Dethier C, Danhier P, Fontenille M, et al. Lactate dehydrogenase b controls lysosome activity and autophagy in cancer. Cancer Cell. 2016;30. https://doi.org/10.1016/j.ccell.2016.08.005.
Wu CC, Taylor RS, Lane DR, Ladinsky MS, Weisz JA, Howell KE. Gmx33: a novel family of trans-golgi proteins identified by proteomics. Traffic (Cph, Den). 2000;1:963–75. https://doi.org/10.1111/j.1600-0854.2000.11206.x.
Sun J, Yang X, Zhang R, Liu S, Gan X, Xi X, et al. Golph3 induces epithelial-mesenchymal transition via wnt/β-catenin signaling pathway in epithelial ovarian cancer. Cancer Med. 2017;6:834–44. https://doi.org/10.1002/cam4.1040.
Wang K, Jiang S, Huang A, Gao Y, Peng B, Li Z, et al. Golph3 promotes cancer growth by interacting with stip1 and regulating telomerase activity in pancreatic ductal adenocarcinoma. Front Oncol. 2020;10:575358. https://doi.org/10.3389/fonc.2020.575358.
Shervington A, Cruickshanks N, Wright H, Atkinson-Dell R, Lea R, Roberts G, et al. Glioma: what is the role of c-myc, hsp90 and telomerase? Mol Cell Biochem. 2006;283:1–09. https://doi.org/10.1007/s11010-006-2495-z.
Qian X, Cheng J, Chen A, Wang Y, Tao Y, Cao J, et al. Long-term effects of short hairpin rna-targeted human telomerase reverse transcriptase on suppression of sgc-7901 cell proliferation by inhibition of telomerase activity. Oncol Rep. 2008;19:575–81.
Li J, Huang X, Xie X, Wang J, Duan M. Human telomerase reverse transcriptase regulates cyclin d1 and g1/s phase transition in laryngeal squamous carcinoma. Acta Otolaryngol. 2011;131:546–51. https://doi.org/10.3109/00016489.2011.557393.
Lin Y, Qian F, Shen L, Chen F, Chen J, Shen B. Computer-aided biomarker discovery for precision medicine: data resources, models and applications. Brief Bioinform. 2017;20:952–75. https://doi.org/10.1093/bib/bbx158.
Chen J, Sun M, Shen B. Deciphering oncogenic drivers: from single genes to integrated pathways. Brief Bioinform. 2015;16:413–28. https://doi.org/10.1093/bib/bbu039.
Yuan X, Dai M, Xu D. Telomere-related markers for cancer. Curr Top Med Chem. 2020;20:410–32. https://doi.org/10.2174/1568026620666200106145340.
Mizumoto K, Suehara N, Muta T, Kitajima S, Hamasaki N, Tominaga Y, et al. Semi-quantitative analysis of telomerase in pancreatic ductal adenocarcinoma. J Gastroenterol. 1996;31:894–7. https://doi.org/10.1007/BF02358622.
Tsutsumi M, Tsujiuchi T, Ishikawa O, Majima T, Yoshimoto M, Sasaki Y, et al. Increased telomerase activities in human pancreatic duct adenocarcinomas. Jpn J Cancer Res: Gann. 1997;88:971–6. https://doi.org/10.1111/j.1349-7006.1997.tb00317.x.
Suehara N, Mizumoto K, Kusumoto M, Niiyama H, Ogawa T, Yamaguchi K, et al. Telomerase activity detected in pancreatic juice 19 months before a tumor is detected in a patient with pancreatic cancer. Am J Gastroenterol. 1998;93:1967–71.
Yeh TS, Cheng AJ, Chen TC, Jan YY, Hwang TL, Jeng LB, et al. Telomerase activity is a useful marker to distinguish malignant pancreatic cystic tumors from benign neoplasms and pseudocysts. J Surg Res. 1999;87:171–7. https://doi.org/10.1006/jsre.1999.5699.
Sato N, Maehara N, Mizumoto K, Nagai E, Yasoshima T, Hirata K, et al. Telomerase activity of cultured human pancreatic carcinoma cell lines correlates with their potential for migration and invasion. Cancer. 2001;91:496–504. https://doi.org/10.1002/1097-0142(20010201)91:3<496::aid-cncr1028>3.0.co;2-0.
Balcom JH, Keck T, Warshaw AL, Antoniu B, Graeme-Cook F, Fernández-Del Castillo C. Telomerase activity in periampullary tumors correlates with aggressive malignancy. Ann Surg. 2001;234:344–50. https://doi.org/10.1097/00000658-200109000-00008.
Sawabu N, Watanabe H, Yamaguchi Y, Ohtsubo K, Motoo Y. Serum tumor markers and molecular biological diagnosis in pancreatic cancer. Pancreas. 2004;28:263–7.
Seki K, Suda T, Aoyagi Y, Sugawara S, Natsui M, Motoyama H, et al. Diagnosis of pancreatic adenocarcinoma by detection of human telomerase reverse transcriptase messenger rna in pancreatic juice with sample qualification. Clin Cancer Res: Off J Am Assoc Cancer Res. 2001;7:1976–81.
Apolónio JD, Dias JS, Fernandes MT, Komosa M, Lipman T, Zhang CH, et al. Thor is a targetable epigenetic biomarker with clinical implications in breast cancer. Clin Epigenet. 2022;14:178. https://doi.org/10.1186/s13148-022-01396-3.
Faleiro I, Apolónio JD, Price AJ, De Mello RA, Roberto VP, Tabori U, et al. The tert hypermethylated oncologic region predicts recurrence and survival in pancreatic cancer. Future Oncol (Lond, Engl). 2017;13:2045–51. https://doi.org/10.2217/fon-2017-0167.
Matsuda Y, Yamashita T, Ye J, Yasukawa M, Yamakawa K, Mukai Y, et al. Phosphorylation of htert at threonine 249 is a novel tumor biomarker of aggressive cancer with poor prognosis in multiple organs. J Pathol. 2022;257:172–85. https://doi.org/10.1002/path.5876.
Campa D, Mergarten B, De Vivo I, Boutron-Ruault M, Racine A, Severi G, et al. Leukocyte telomere length in relation to pancreatic cancer risk: a prospective studytelomeres and pancreatic cancer. Cancer Epidemiol Biomark Prev. 2014;23:2447–54.
Luu HN, Huang JY, Wang R, Adams-Haduch J, Jin A, Koh WP, et al. Association between leukocyte telomere length and the risk of pancreatic cancer: findings from a prospective study. Plos One. 2019;14:e221697. https://doi.org/10.1371/journal.pone.0221697.
Antwi SO, Bamlet WR, Cawthon RM, Rabe KG, Druliner BR, Sicotte H, et al. Shorter treatment-naïve leukocyte telomere length is associated with poorer overall survival of patients with pancreatic ductal adenocarcinoma. Cancer Epidemiol Biomark Prev: Publ Am Assoc Cancer Res Cosponsored Am Soc Preventive Oncol. 2021;30:210–16. https://doi.org/10.1158/1055-9965.EPI-20-1279.
Antwi SO, Bamlet WR, Broderick BT, Chaffee KG, Oberg A, Jatoi A, et al. Genetically predicted telomere length is not associated with pancreatic cancer risktelomere-related genotype and pancreatic cancer risk. Cancer Epidemiol Biomark Prev. 2017;26:971–4.
Antwi SO, Bamlet WR, Rabe KG, Cawthon RM, Umudi I, Druliner BR, et al. Leukocyte telomere length and its interaction with germline variation in telomere-related genes in relation to pancreatic adenocarcinoma risk. Cancer Epidemiol Biomark Prev: A Publ Am Assoc Cancer Res Cosponsored Am Soc Preventive Oncol. 2020;29:1492–500. https://doi.org/10.1158/1055-9965.EPI-19-1597.
Campa D, Matarazzi M, Greenhalf W, Bijlsma M, Saum KU, Pasquali C, et al. Genetic determinants of telomere length and risk of pancreatic cancer: a pandora study. Int J Cancer. 2019;144:1275–83. https://doi.org/10.1002/ijc.31928.
Zhang R, Zhao J, Xu J, Liu F. Association of peripheral leukocyte telomere length and its variation with pancreatic cancer and colorectal cancer risk in Chinese population. Oncotarget. 2016;7:38579–85. https://doi.org/10.18632/oncotarget.9536.
Hata T, Dal Molin M, Mcgregor-Das A, Song TJ, Wolfgang C, Eshleman JR, et al. Simple detection of telomere fusions in pancreatic cancer, intraductal papillary mucinous neoplasm, and pancreatic cyst fluid. J Mol Diagnostics: Jmd. 2018;20:46–55. https://doi.org/10.1016/j.jmoldx.2017.09.006.
Maciejowski J, de Lange T. Telomeres in cancer: tumour suppression and genome instability. Nat Rev Mol Cell Biol. 2017;18:175–86. https://doi.org/10.1038/nrm.2016.171.
Okamoto K, Seimiya H. Revisiting telomere shortening in cancer. Cells. 2019;8. https://doi.org/10.3390/cells8020107.
Hannen R, Bartsch JW. Essential roles of telomerase reverse transcriptase htert in cancer stemness and metastasis. Febs Lett. 2018;592:2023–31. https://doi.org/10.1002/1873-3468.13084.
Scardino A, Gross DA, Alves P, Schultze JL, Graff-Dubois S, Faure O, et al. Her-2/neu and htert cryptic epitopes as novel targets for broad spectrum tumor immunotherapy. J Immunol (Baltim, Md: 1950). 2002;168:5900–06. https://doi.org/10.4049/jimmunol.168.11.5900.
Lilleby W, Gaudernack G, Brunsvig PF, Vlatkovic L, Schulz M, Mills K, et al. Phase i/iia clinical trial of a novel htert peptide vaccine in men with metastatic hormone-naive prostate cancer. Cancer Immunol Immunother: Cii. 2017;66:891–901. https://doi.org/10.1007/s00262-017-1994-y.
Asai A, Oshima Y, Yamamoto Y, Uochi TA, Kusaka H, Akinaga S, et al. A novel telomerase template antagonist (grn163) as a potential anticancer agent. Cancer Res. 2003;63:3931–9.
Biffi G, Tannahill D, Mccafferty J, Balasubramanian S. Quantitative visualization of dna g-quadruplex structures in human cells. Nat Chem. 2013;5:182–6. https://doi.org/10.1038/nchem.1548.
Guterres AN, Villanueva J. Targeting telomerase for cancer therapy. Oncogene. 2020;39:5811–24. https://doi.org/10.1038/s41388-020-01405-w.
Damm K, Hemmann U, Garin-Chesa P, Hauel N, Kauffmann I, Priepke H, et al. A highly selective telomerase inhibitor limiting human cancer cell proliferation. Embo J. 2001;20:6958–68. https://doi.org/10.1093/emboj/20.24.6958.
Baell JB. Feeling nature’s pains: natural products, natural product drugs, and pan assay interference compounds (pains). J Nat Prod. 2016;79:616–28. https://doi.org/10.1021/acs.jnatprod.5b00947.
Mender I, Gryaznov S, Dikmen ZG, Wright WE, Shay JW. Induction of telomere dysfunction mediated by the telomerase substrate precursor 6-thio-2’-deoxyguanosine. Cancer Discov. 2015;5:82–95. https://doi.org/10.1158/2159-8290.CD-14-0609.
Mender I, Laranger R, Luitel K, Peyton M, Girard L, Lai TP, et al. Telomerase-mediated strategy for overcoming non-small cell lung cancer targeted therapy and chemotherapy resistance. Neoplasia (N. Y, N. Y). 2018;20:826–37. https://doi.org/10.1016/j.neo.2018.06.002.
Bernhardt SL, Gjertsen MK, Trachsel S, Møller M, Eriksen JA, Meo M, et al. Telomerase peptide vaccination of patients with non-resectable pancreatic cancer: a dose escalating phase i/ii study. Br J Cancer. 2006;95:1474–82. https://doi.org/10.1038/sj.bjc.6603437.
Middleton G, Silcocks P, Cox T, Valle J, Wadsley J, Propper D, et al. Gemcitabine and capecitabine with or without telomerase peptide vaccine gv1001 in patients with locally advanced or metastatic pancreatic cancer (telovac): an open-label, randomised, phase 3 trial. Lancet Oncol. 2014;15:829–40.
Kim S, Kim BJ, Kim I, Kim JH, Kim HK, Ryu H, et al. A phase ii study of chemotherapy in combination with telomerase peptide vaccine (gv1001) as second-line treatment in patients with metastatic colorectal cancer. J Cancer. 2022;13:1363–9. https://doi.org/10.7150/jca.70385.
Kim JW, Yadav DK, Kim SJ, Lee MY, Park JM, Kim BS, et al. Anti-cancer effect of gv1001 for prostate cancer: function as a ligand of gnrhr. Endocr Relat Cancer. 2019;26:147–62. https://doi.org/10.1530/ERC-18-0454.
Kyte JA, Gaudernack G, Dueland S, Trachsel S, Julsrud L, Aamdal S. Telomerase peptide vaccination combined with temozolomide: a clinical trial in stage iv melanoma patients. Clin Cancer Res: Off J Am Assoc Cancer Res. 2011;17:4568–80. https://doi.org/10.1158/1078-0432.CCR-11-0184.
Kim JH, Cho YR, Ahn EK, Kim S, Han S, Kim SJ, et al. A novel telomerase-derived peptide gv1001-mediated inhibition of angiogenesis: regulation of vegf/vegfr-2 signaling pathways. Transl Oncol. 2022;26:101546. https://doi.org/10.1016/j.tranon.2022.101546.
Kim GE, Jung AR, Kim MY, Lee JB, Im JH, Lee KW, et al. Gv1001 induces apoptosis by reducing angiogenesis in renal cell carcinoma cells both in vitro and in vivo. Urology. 2018;113:129–37. https://doi.org/10.1016/j.urology.2017.10.038.
Vonderheide RH, Kraynyak KA, Shields AF, Mcree AJ, Johnson JM, Sun W, et al. Phase 1 study of safety, tolerability and immunogenicity of the human telomerase (htert)-encoded dna plasmids ino-1400 and ino-1401 with or without il-12 dna plasmid ino-9012 in adult patients with solid tumors. J Immunother Cancer. 2021;9. https://doi.org/10.1136/jitc-2021-003019.
Kanaya N, Kuroda S, Kakiuchi Y, Kumon K, Tsumura T, Hashimoto M, et al. Immune modulation by telomerase-specific oncolytic adenovirus synergistically enhances antitumor efficacy with anti-pd1 antibody. Mol Ther: J Am Soc Gene Ther. 2020;28:794–804. https://doi.org/10.1016/j.ymthe.2020.01.003.
Bryan C, Rice C, Hoffman H, Harkisheimer M, Sweeney M, Skordalakes E. Structural basis of telomerase inhibition by the highly specific bibr1532. Struct (Lond, Engl: 1993). 2015;23:1934–42. https://doi.org/10.1016/j.str.2015.08.006.
Burchett KM, Yan Y, Ouellette MM. Telomerase inhibitor imetelstat (grn163l) limits the lifespan of human pancreatic cancer cells. Plos One. 2014;9:e85155. https://doi.org/10.1371/journal.pone.0085155.
Röth A, Harley CB, Baerlocher GM. Imetelstat (grn163l)-telomerase-based cancer therapy. Recent Results Cancer Res Fortschr Der Krebsforsch Prog Dans Les Rech Sur Le Cancer. 2010;184:221–34. https://doi.org/10.1007/978-3-642-01222-8_16.
Joseph I, Tressler R, Bassett E, Harley C, Buseman CM, Pattamatta P, et al. The telomerase inhibitor imetelstat depletes cancer stem cells in breast and pancreatic cancer cell lines. Cancer Res. 2010;70:9494–504. https://doi.org/10.1158/0008-5472.CAN-10-0233.
Wu X, Smavadati S, Nordfjäll K, Karlsson K, Qvarnström F, Simonsson M, et al. Telomerase antagonist imetelstat inhibits esophageal cancer cell growth and increases radiation-induced dna breaks. Biochim Et Biophys Acta. 2012;1823:2130–5. https://doi.org/10.1016/j.bbamcr.2012.08.003.
Frink RE, Peyton M, Schiller JH, Gazdar AF, Shay JW, Minna JD. Telomerase inhibitor imetelstat has preclinical activity across the spectrum of non-small cell lung cancer oncogenotypes in a telomere length dependent manner. Oncotarget. 2016;7:31639.
Chiappori AA, Kolevska T, Spigel DR, Hager S, Rarick M, Gadgeel S, et al. A randomized phase ii study of the telomerase inhibitor imetelstat as maintenance therapy for advanced non-small-cell lung cancer. Ann Oncol: Off J Eur Soc Med Oncol. 2015;26:354–62. https://doi.org/10.1093/annonc/mdu550.
Deeb D, Brigolin C, Gao X, Liu Y, Pindolia KR, Gautam SC. Induction of apoptosis in pancreatic cancer cells by cddo-me involves repression of telomerase through epigenetic pathways. J Carcinogenesis Mutagenesis. 2014;5:177. https://doi.org/10.4172/2157-2518.1000177.
Kimura A, Ohmichi M, Kawagoe J, Kyo S, Mabuchi S, Takahashi T, et al. Induction of htert expression and phosphorylation by estrogen via akt cascade in human ovarian cancer cell lines. Oncogene. 2004;23:4505–15. https://doi.org/10.1038/sj.onc.1207582.
Kyo S, Takakura M, Taira T, Kanaya T, Itoh H, Yutsudo M, et al. Sp1 cooperates with c-myc to activate transcription of the human telomerase reverse transcriptase gene (htert). Nucleic Acids Res. 2000;28:669–77. https://doi.org/10.1093/nar/28.3.669.
Konnikova L, Simeone MC, Kruger MM, Kotecki M, Cochran BH. Signal transducer and activator of transcription 3 (stat3) regulates human telomerase reverse transcriptase (htert) expression in human cancer and primary cells. Cancer Res. 2005;65:6516–20. https://doi.org/10.1158/0008-5472.CAN-05-0924.
Deeb D, Gao X, Liu Y, Varma NR, Arbab AS, Gautam SC. Inhibition of telomerase activity by oleanane triterpenoid cddo-me in pancreatic cancer cells is ros-dependent. Molecules (Basel, Switz). 2013;18:3250–65. https://doi.org/10.3390/molecules18033250.
Gao X, Deeb D, Liu Y, Liu P, Zhang Y, Shaw J, et al. Cddo-me inhibits tumor growth and prevents recurrence of pancreatic ductal adenocarcinoma. Int J Oncol. 2015;47:2100–06. https://doi.org/10.3892/ijo.2015.3212.
Burge S, Parkinson GN, Hazel P, Todd AK, Neidle S. Quadruplex dna: sequence, topology and structure. Nucleic Acids Res. 2006;34:5402–15. https://doi.org/10.1093/nar/gkl655.
Drosopoulos WC, Kosiyatrakul ST, Schildkraut CL. Blm helicase facilitates telomere replication during leading strand synthesis of telomeres. J Cell Biol. 2015;210:191–208. https://doi.org/10.1083/jcb.201410061.
Kosiol N, Juranek S, Brossart P, Heine A, Paeschke K. G-quadruplexes: a promising target for cancer therapy. Mol Cancer. 2021;20:40. https://doi.org/10.1186/s12943-021-01328-4.
Tesmer VM, Brenner KA, Nandakumar J. Human pot1 protects the telomeric ds-ss dna junction by capping the 5’ end of the chromosome. Science (N. Y, N. Y). 2023;381:771–8. https://doi.org/10.1126/science.adi2436.
Gunaratnam M, de la Fuente M, Hampel SM, Todd AK, Reszka AP, Schätzlein A, et al. Targeting pancreatic cancer with a g-quadruplex ligand. Bioorg Med Chem. 2011;19:7151–7. https://doi.org/10.1016/j.bmc.2011.09.055.
Hampel SM, Sidibe A, Gunaratnam M, Riou JF, Neidle S. Tetrasubstituted naphthalene diimide ligands with selectivity for telomeric g-quadruplexes and cancer cells. Bioorg Med Chem Lett. 2010;20:6459–63. https://doi.org/10.1016/j.bmcl.2010.09.066.
Micco M, Collie GW, Dale AG, Ohnmacht SA, Pazitna I, Gunaratnam M, et al. Structure-based design and evaluation of naphthalene diimide g-quadruplex ligands as telomere targeting agents in pancreatic cancer cells. J Med Chem. 2013;56:2959–74. https://doi.org/10.1021/jm301899y.
Cogoi S, Zorzet S, Rapozzi V, Géci I, Pedersen EB, Xodo LE. Maz-binding g4-decoy with locked nucleic acid and twisted intercalating nucleic acid modifications suppresses kras in pancreatic cancer cells and delays tumor growth in mice. Nucleic Acids Res. 2013;41:4049–64. https://doi.org/10.1093/nar/gkt127.
Pattanayak R, Barua A, Das A, Chatterjee T, Pathak A, Choudhury P, et al. Porphyrins to restrict progression of pancreatic cancer by stabilizing kras g-quadruplex: in silico, in vitro and in vivo validation of anticancer strategy. Eur J Pharm Sci: Off J Eur Federation Pharm Sci. 2018;125:39–53. https://doi.org/10.1016/j.ejps.2018.09.011.
Seimiya H, Oh-Hara T, Suzuki T, Naasani I, Shimazaki T, Tsuchiya K, et al. Telomere shortening and growth inhibition of human cancer cells by novel synthetic telomerase inhibitors mst-312, mst-295, and mst-1991. Mol Cancer Ther. 2002;1:657–65.
Salloum R, Hummel TR, Kumar SS, Dorris K, Li S, Lin T, et al. A molecular biology and phase ii study of imetelstat (grn163l) in children with recurrent or refractory central nervous system malignancies: a pediatric brain tumor consortium study. J Neurooncol. 2016;129:443–51. https://doi.org/10.1007/s11060-016-2189-7.
Shay JW, Wright WE. Telomeres and telomerase in normal and cancer stem cells. Febs Lett. 2010;584:3819–25. https://doi.org/10.1016/j.febslet.2010.05.026.
Hahn WC, Counter CM, Lundberg AS, Beijersbergen RL, Brooks MW, Weinberg RA. Creation of human tumour cells with defined genetic elements. Nature. 1999;400:464–8. https://doi.org/10.1038/22780.
Jäger K, Walter M. Therapeutic targeting of telomerase. Genes (Basel). 2016;7. https://doi.org/10.3390/genes7070039.
Hu J, Hwang SS, Liesa M, Gan B, Sahin E, Jaskelioff M, et al. Antitelomerase therapy provokes alt and mitochondrial adaptive mechanisms in cancer. Cell. 2012;148:651–63.
Shay JW, Wright WE. Telomeres and telomerase: three decades of progress. Nat Rev Genet. 2019;20:299–309.
Hiyama E, Kodama T, Shinbara K, Iwao T, Itoh M, Hiyama K, et al. Telomerase activity is detected in pancreatic cancer but not in benign tumors. Cancer Res. 1997;57:326–31.
Uehara H, Nakaizumi A, Tatsuta M, Baba M, Takenaka A, Uedo N, et al. Diagnosis of pancreatic cancer by detecting telomerase activity in pancreatic juice: comparison with k-ras mutations. Am J Gastroenterol. 1999;94:2513–8. https://doi.org/10.1111/j.1572-0241.1999.01386.x.
Myung SJ, Kim MH, Kim YS, Kim HJ, Park ET, Yoo KS, et al. Telomerase activity in pure pancreatic juice for the diagnosis of pancreatic cancer may be complementary to k-ras mutation. Gastrointest Endosc. 2000;51:708–13. https://doi.org/10.1067/mge.2000.104654.
Pearson AS, Chiao P, Zhang L, Zhang W, Larry L, Katz RL, et al. The detection of telomerase activity in patients with adenocarcinoma of the pancreas by fine needle aspiration. Int J Oncol. 2000;17:381–5. https://doi.org/10.3892/ijo.17.2.381.
Büchler P, Conejo-Garcia JR, Lehmann G, Müller M, Emrich T, Reber HA, et al. Real-time quantitative pcr of telomerase mrna is useful for the differentiation of benign and malignant pancreatic disorders. Pancreas. 2001;22:331–40. https://doi.org/10.1097/00006676-200105000-00001.
Zhou GX, Huang JF, Li ZS, Xu GM, Liu F, Zhang H. Detection of k-ras point mutation and telomerase activity during endoscopic retrograde cholangiopancreatography in diagnosis of pancreatic cancer. World J Gastroenterol. 2004;10:1337–40. https://doi.org/10.3748/wjg.v10.i9.1337.
Ohuchida K, Mizumoto K, Ishikawa N, Sato N, Nagai E, Yamaguchi K, et al. A highly sensitive and quantitative telomerase activity assay with pancreatic juice is useful for diagnosis of pancreatic carcinoma without problems due to polymerase chain reaction inhibitors: analysis of 100 samples of pancreatic juice from consecutive patients. Cancer. 2004;101:2309–17. https://doi.org/10.1002/cncr.20649.
Mizumoto K, Tanaka M. Detection of telomerase activity in patients with pancreatic cancer. Methods Mol Med. 2005;103:199–205. https://doi.org/10.1385/1-59259-780-7:199.
Ohuchida K, Mizumoto K, Ogura Y, Ishikawa N, Nagai E, Yamaguchi K, et al. Quantitative assessment of telomerase activity and human telomerase reverse transcriptase messenger rna levels in pancreatic juice samples for the diagnosis of pancreatic cancer. Clin Cancer Res: Off J Am Assoc Cancer Res. 2005;11:2285–92. https://doi.org/10.1158/1078-0432.CCR-04-1581.
Kawahara R, Odo M, Kinoshita H, Shirouzu K, Aoyagi S. Analysis of htert mrna expression in biliary tract and pancreatic cancer. J Hepato-Biliary-Pancreat Surg. 2007;14:189–93. https://doi.org/10.1007/s00534-006-1132-2.
Zhou GX, Huang JF, Zhang H, Chen JP. Diagnosis of pancreatic cancer by cytology and telomerase activity in exfoliated cells obtained by pancreatic duct brushing during endoscopy. Hepatobiliary Pancreat Dis Int: Hbpd Int. 2007;6:308–11.
Uehara H, Nakaizumi A, Iishi H, Takenaka A, Eguchi H, Ohigashi H, et al. In situ telomerase activity in pancreatic juice may discriminate pancreatic cancer from other pancreatic diseases. Pancreas. 2008;36:236–40. https://doi.org/10.1097/MPA.0b013e31815bc1d6.
Skinner HG, Gangnon RE, Litzelman K, Johnson RA, Chari ST, Petersen GM, et al. Telomere length and pancreatic cancer: a case-control study. Cancer Epidemiol, Biomark Prev: A Publ Am Assoc Cancer Res Cosponsored Am Soc Preventive Oncol. 2012;21:2095–100. https://doi.org/10.1158/1055-9965.EPI-12-0671.
Matsuda Y, Ishiwata T, Izumiyama-Shimomura N, Hamayasu H, Fujiwara M, Tomita K, et al. Gradual telomere shortening and increasing chromosomal instability among panin grades and normal ductal epithelia with and without cancer in the pancreas. Plos One. 2015;10:e117575. https://doi.org/10.1371/journal.pone.0117575.
Bao Y, Prescott J, Yuan C, Zhang M, Kraft P, Babic A, et al. Tertleucocyte telomere length, genetic variants at the gene region and risk of pancreatic cancer. Gut. 2017;66:1116–22. https://doi.org/10.1136/gutjnl-2016-312510.
Matsuda Y, Suzuki A, Esaka S, Hamashima Y, Imaizumi M, Kinoshita M, et al. Telomere length determined by the fluorescence in situ hybridisation distinguishes malignant and benign cells in cytological specimens. Cytopathol: Off J Br Soc Clin Cytol. 2018;29:262–6. https://doi.org/10.1111/cyt.12535.
Hamada T, Yuan C, Bao Y, Zhang M, Khalaf N, Babic A, et al. Prediagnostic leukocyte telomere length and pancreatic cancer survival. Cancer Epidemiol Biomark Prev: A Publ Am Assoc Cancer Res Cosponsored Am Soc Preventive Oncol. 2019;28:1868–75. https://doi.org/10.1158/1055-9965.EPI-19-0577.
Nault JC, Ningarhari M, Rebouissou S, Zucman-Rossi J. The role of telomeres and telomerase in cirrhosis and liver cancer. Nat Rev Gastroenterol Hepatol. 2019;16:544–58. https://doi.org/10.1038/s41575-019-0165-3.
Funding
This work was supported by grants from the Central High-level Hospital of Traditional Chinese Medicine Clinical Research and Achievement Transformation Ability Improvement Project (grant number HLCMHPP2023005).
Author information
Authors and Affiliations
Contributions
BP and conceived the study and revised the final manuscript. SS, YL, JC collected relevant references, drafted the manuscript, CL, XZ, and XJ and finished the table. DL, YL, EG, and ZP offered crucial content revision and language polishing. All authors reviewed the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Shou, S., Li, Y., Chen, J. et al. Understanding, diagnosing, and treating pancreatic cancer from the perspective of telomeres and telomerase. Cancer Gene Ther (2024). https://doi.org/10.1038/s41417-024-00768-6
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41417-024-00768-6