Abstract
Background
Among patients with nephroblastoma, those with bilateral disease are a unique population where maximising tumour control must be balanced with preserving renal parenchyma.
Methods
The SIOP 2001 protocol recommended surgery after neoadjuvant cycle(s) of Dactinomycin and Vincristine (AV) with response-adapted intensification, if needed. Adjuvant treatment was given based on the lesion with the worst histology.
Results
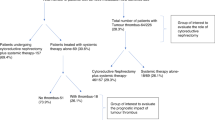
Three hundred and twenty seven patients with stage V disease were evaluable: 174 had bilateral Wilms tumour (BWT), 101 unilateral WT and contralateral nephroblastomatosis (NB) and 52 bilateral nephroblastomatosis. In these three groups, the estimated 5y-EFS was 76.1%, 84.6%, and 74.9%, respectively. AV chemotherapy alone was the successful chemotherapy for 58.7% of all the patients and 65.6% of the non-metastatic patients. Among the 174 patients with BWT, 149 (88.2%) had at least one nephron-sparing surgery. Twenty of 61 bilateral stage I patients were treated with four-week AV postoperatively achieving 94.4% 5y-EFS. At last follow-up, 87% of patients had normal renal function.
Conclusions
This study demonstrates that AV without anthracyclines is sufficient to achieve NSS and good survival in the majority of patients. For patients with bilateral stage I WT and intermediate risk histology, only four weeks adjuvant AV seems to be sufficient.
Clinical Trial Registration
NCT00047138
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 24 print issues and online access
$259.00 per year
only $10.79 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Data availability
All data generated or analysed during this study are included in this published article [and its supplementary information files].
References
Breslow N, Olshan A, Beckwith JB, Green DM. Epidemiology of Wilms tumour. Med Pediatr Oncol. 1993;21:172–81.
Charlton J, Irtan S, Bergeron C, Pritchard-Jones K. Bilateral Wilms tumour: a review of clinical and molecular features. Expert Rev Mol Med. 2017;19:e8.
Coppes MJ, de Kraker J, van Dijken PJ, Perry HJ, Delemarre JF, Tournade MF, et al. Bilateral Wilms’ Tumour: long-term survival and some epidemiological features. J Clin Oncol. 1989;7:310–5.
Tournade MF, Com-Nougué C, De Kraker J, Ludwig R, Rey A, Burgers JMB, et al. Optimal duration of preoperative therapy in unilateral and nonmetastatic Wilms’ Tumour in children older than 6 months: Results of the Ninth International Society of Pediatric Oncology Wilms’ Tumour Trial and Study. J Clin Oncol. 2001;19:488–500.
Furtwängler R, Schmolze M, Graber S, Leuschner I, Amann G, Schenk JP, et al. Pretreatment for bilateral Nephroblastomatosis is an Independent risk factor for progressive disease in patients with Stage V Nephroblastoma. Klin Padiatr. 2014;226:175–81.
Sudour H, Audry G, Schleimacher G, Patte C, Dussart S, Bergeron C. Bilateral Wilms Tumours (WT) treated with the SIOP 93 protocol in France: epidemiological survey and patient outcome. Pediatr Blood Cancer. 2012;59:57–61.
Ehrlich P, Chi YY, Chintagumpala MM, Hoffer FA, Perlman EJ, Kalapurakal JA, et al. Results of the first prospective Multi-institutional Treatment Study in Children With Bilateral Wilms Tumour (AREN0534): A report from the Children’s Oncology Group. Ann Surg. 2017;266:470–8.
Indolfi P, Jenkner A, Terenziani M, Crocoli A, Serra A, Collini P, et al. Synchronous bilateral Wilms Tumour: a report from the Associazione Italiana Ematologia Oncologia Pediatrica (AIEOP). Cancer. 2013;119:1586–92.
Wilde JC, Aronson DC, Sznajder B, Van Tinteren H, Powis M, Okoye B, et al. Nephron sparing surgery (NSS) for unilateral wilms Tumour (UWT): the SIOP 2001 experience. Pediatr Blood Cancer. 2014;61:2175–9.
Welter N, Wagner A, Furtwangler R, Melchior P, Kager L, Vokuhl C, et al. Characteristics of Nephroblastoma/Nephroblastomatosis in children with a clinically reported underlying malformation or cancer predisposition syndrome. Cancers. 2021;13:5016.
Kubiak R, Gundeti M, Duffy PG, Ransley PG, Wilcox DT. Renal function and outcome following salvage surgery for bilateral Wilms’ Tumour. J Pediatr Surg. 2004;39:1667–72.
Murphy AJ, Davidoff AM. Bilateral Wilms tumour: A surgical perspective. Children. 2018;5:134.
Pritchard-Jones K, Bergeron C, de Camargo B, van den Heuvel-Eibrink MM, Acha T, Godzinski J, et al. Omission of doxorubicin from the treatment of stage II-III, intermediate-risk Wilms’ tumour (SIOP WT 2001): an open-label, non-inferiority, randomised controlled trial. Lancet. 2015;386:1156–64.
Van Den Heuvel-Eibrink MM, Van Tinteren H, Bergeron C, Coulomb-L’Hermine A, De Camargo B, Leuschner I, et al. Outcome of localised blastemal-type Wilms tumour patients treated according to intensified treatment in the SIOP WT 2001 protocol, a report of the SIOP Renal Tumour Study Group (SIOP-RTSG). Eur J Cancer. 2015;51:498–506.
Furtwängler R, Gooskens SL, van Tinteren H, de Kraker J, Schleiermacher G, Bergeron C, et al. Clear cell sarcomas of the kidney registered on International Society of Pediatric Oncology (SIOP) 93-01 and SIOP 2001 protocols: a report of the SIOP Renal Tumour Study Group. Eur J Cancer. 2013;49:3497–506.
Hamilton TE, Ritchey ML, Haase GM, Argani P, Peterson SM, Anderson JR, et al. The management of synchronous bilateral wilms Tumour: A report from the National Wilms Tumour Study Group. Ann Surg. 2011;253:1004–10.
Rapley EA, Barfoot R, Bonaïti-Pellié C, Chompret A, Foulkes W, Perusinghe N, et al. Evidence for susceptibility genes to familial Wilms tumour in addition to WT1, FWT1 and FWT2. BJC. 2000;82:177–83.
Ehrlich PF. Bilateral Wilms’ Tumour: the need to improve outcomes. Expert Rev Anticancer Ther. 2009;9:963–73.
Weirich A, Ludwig R, Graf N, Abel U, Leuschner I, Vujanic GM, et al. Survival in nephroblastoma treated according to the trial and study SIOP-9/GPOH with respect to relapse and morbidity. Ann Oncol. 2004;15:808.
Perlman EJ, Faria P, Soares A, Hoffer F, Sredni S, Ritchey M, et al. Hyperplastic perilobar nephroblastomatosis: Long-term survival of 52 patients. Pediatr Blood Cancer. 2006;46:203–21.
Shamberger RC, Haase GM, Argani P, Perlman EJ, Cotton CA, Takashima J, et al. Bilateral Wilms’ Tumours with progressive or nonresponsive disease. J Pediatr Surg. 2006;41:652–7.
Verschuur AC, Vujanic GM, Van Tinteren H, Jones KP, de Kraker J, Sandstedt B. Stromal and epithelial predominant Wilms tumours have an excellent outcome: the SIOP 93 01 experience. Pediatr Blood cancer. 2010;55:233–8.
Vujanic GM, Gessler M, Ooms A, Collini P, Coulomb-l’Hermine A, D’Hooghe E, et al. The UMBRELLA SIOP-RTSG 2016 Wilms tumour pathology and molecular biology protocol. Nat Rev Urol. 2018;15:693–701.
Treger TD, Chagtai T, Butcher R, Cresswell GD, Al-Saadi R, Brok J, et al. Somatic TP53 mutations are detectable in circulating tumour DNA from children with anaplastic Wilms tumours. Transl Oncol. 2018;11:1301–6.
Littooij AS, Nikkels PG, Hulsbergen-van de Kaa CA, van de Ven CP, van den Heuvel-Eibrink MM, Olsen ØE. Apparent diffusion coefficient as it relates to histopathology finding in post chemotherapy nephroblastoma: a feasibility study. Pediatr Radiol. 2017;47:1608–14.
Van den Heuvel-Eibrink MM, Hol JA, Pritchard-Jones K, van Tinteren H, Furtwangler R, Verschuur AC, et al. Position paper: Rationale for the treatment of Wilms tumour in the UMBRELLA SIOP-RTSG 2016 protocol. Nat Rev Urol. 2017;14:743–52.
Fialkowski E, Sudour-Bonnange H, Vujanic GM, Shamberger RC, Chowdhury T, Aldrink JH, et al. The varied spectrum of nephroblastomatosis, nephrogenic rests, and Wilms Tumours: Review of current definitions and challenges of the field. Pediatr Blood Cancer. 2023;70:e30162.
Ortiz MV, Fernandez-Ledon S, Ramaswamy K, Forlenza CJ, Shukla NN, Kobos R, et al. Maintenance chemotherapy to reduce the risk of a metachronous Wilms Tumour in children with bilateral nephroblastomatosis. Pediatr Blood Cancer. 2019;66:e27500.
Jagt CT, Zuckermann M, Ten Kate F, Taminiau JA, Dijkgraaf MG, Heij H, et al. Veno-occlusive disease as a complication of preoperative chemotherapy for Wilms Tumour: A clinico-pathological analysis. Pediatr Blood Cancer. 2009;53:1211–5.
Vanden Berg RN, Bierman EN, Noord MV, Rice HE, Routh JC. Nephron-sparing surgery for Wilms Tumour: A systematic review. Urol Oncol. 2016;34:24–32.
Fuchs J, Schunn MC, Schäfer JF, Ebinger M, Graf N, Furtwängler R, et al. Redo nephron-sparing surgery in stage V pediatric renal Tumours - A report from the SIOP/GPOH study group for renal Tumours. Eur J Surg Oncol. 2024;50:107265. Jan
Godzinski J, Graf N, Audry G. Current concepts in surgery for Wilms Tumour-the risk and function-adapted strategy. Eur J Pediatr Surg. 2014;24:457–60.
Breslow NE, Collins AJ, Ritchey ML, Grigoriev YA, Peterson SM, Green DM. End stage renal disease in patients with Wilms Tumour: Results from the National Wilms Tumour Study Group and the United States Renal Data System. J Urol. 2005;174:1972–5.
Aronson DC, Slaar A, Heinen RC, de Kraker J, Heij HA. Long-term outcome of bilateral Wilms Tumours (BWT). Pediatr Blood Cancer. 2011;56:1110–3.
Hubertus J, Günther B, Becker K, Graf N, Furtwängler R, Ferrari R, et al. Development of hypertension is less frequent after bilateral nephron sparing surgery for bilateral Wilms Tumour in a long-term survey. J Urol. 2015;193:262–6.
Kieran K, Williams MA, Dome JS, McGregor LM, Krasin MJ, Davidoff AM. Margin status and Tumour recurrence after nephron-sparing surgery for bilateral Wilms Tumour. J Pediatr Surg. 2013;48:1481–5.
Spiegl HR, Murphy AJ, Yanishevski D, Brennan RC, Li C, Lu Z, et al. Complications following Nephron-sparing surgery for Wilms tumour. J Pediatr Surg. 2020;55:126–9.
Beckwith JB, Kiviat NB, Bonadio JF. Nephrogenic rests, nephroblastomatosis, and the pathogenesis of Wilms’ Tumour. J Pediatr Surg. 2020;55:126–9.
Cozzi F, Schiavetti A, Cozzi DA, Morini F, Uccini S, Pierani P, et al. Conservative management of hyperplastic and multicentric nephroblastomatosis. J Urol. 2004;172:1069–70.
Acknowledgements
We acknowledge the enormous efforts made by more than 1000 clinicians working at the 251 childhood cancer treatment centres from 26 countries who enroled and followed up with patients in this study, and the patients and their families for their participation. The SIOP Renal Tumours Study Group and all authors thank the members of the independent data monitoring committee: Max Coppes (British Columbia Cancer Agency, Canada), Otilia Dalesio (Netherlands Cancer Institute, Amsterdam), and Dietmar Schmidt (Mannheim, Germany). We also acknowledge Lucy Wilkie and Aidan McManus for editorial assistance with the journal style and formatting of the final draft.
Special Dedication We address a special memorial to Dr. Christophe Bergeron, dead in December 2023 who has initiated this work in the spectrum of BWT. He has played an important role in numerous initiatives and projects within SIOP and SFCE, including the SIOP Renal Tumour Study Group. He strongly contributed to our collaborative efforts in the field of paediatric renal tumours over many decades.
Funding
The SIOP WT 2001 study was funded by Cancer Research UK (grant C1188/A8687), the UK National Cancer Research Network and Children’s Cancer and Leukaemia Group (CCLG) (who supported the UK section), Société Française des Cancers de l’Enfant and Association Leon Berard Enfant Cancéreux and Enfant et Santé (who supported the French section), Gesellschaft fü r Pädiatrische Onkologie und Hämatologie and Deutsche Krebschilfe (grant 50-2709-Gr2, who supported the German section), Grupo Cooperativo Brasileiro para o Tratamento do Tumour de Wilms and Sociedade Brasileira de Oncologia Pediátrica (who supported the Brazilian section), the Spanish Society of Paediatric Haematology and Oncology and the Spanish Association Against Cancer (who supported the Spanish section) and SIOP-NL. K P-J is partly supported by the National Institute for Health Research Biomedical Research Centre Funding Scheme
Author information
Authors and Affiliations
Contributions
Hélène Sudour-Bonnange: Conceptualisation, Investigation, Writing – Original Draft, Writing – Review & Editing., Harm van Tinteren: Methodology, Formal analysis, Writing – Review & Editing, Gema L Ramírez-Villar: Investigation, Resources, Writing – Review & Editing, Jan Godzinski: Investigation, Resources, Writing – Review & Editing, Sabine Irtan: Investigation, Resources, Writing – Review & Editing, Manfred Gessler: Investigation, Resources, Writing – Review & Editing, Tanzina Chowdhury: Investigation, Resources, Writing – Review & Editing, Georges Audry: Investigation, Resources, Writing – Review & Editing, Joerg Fuchs: Investigation, Resources, Writing – Review & Editing, Mark Powis: Investigation, Resources, Writing – Review & Editing, Cornelis P van de Ven: Investigation, Resources, Writing – Review & Editing, Bruce Okoye: Investigation, Resources, Writing – Review & Editing, Naima Smeulders: Investigation, Resources, Writing – Review & Editing, Gordan M Vujanic: Investigation, Resources, Writing – Review & Editing, Arnaud Verschuur: Investigation, Resources, Writing – Review & Editing, Aurore L’Herminé-Coulomb: Investigation, Resources, Writing – Review & Editing, Beatriz de Camargo: Investigation, Resources, Writing – Review & Editing, Joaquim Caetano de Aguirre Neto: Investigation, Resources, Writing – Review & Editing, Jens Peter Schenk: Investigation, Resources, Writing – Review & Editing, Mary M van den Heuvel-Eibrink: Investigation, Resources, Writing – Review & Editing, Kathy Pritchard-Jones: Investigation, Resources, Writing – Review & Editing, Norbert Graf: Investigation, Resources, Writing – Review & Editing, Christophe Bergeron: Conceptualisation, Investigation, and Rhoikos Furtwängler: Conceptualisation, Investigation, Writing – Review & Editing, Supervision.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethics approval and consent to participate
Ethics approval and consent to participate: national and local regulatory and ethical approvals were obtained according to national regulations.
Consent for publication
The parents or guardians signed a written informed consent for enrolment and treatment of their child in the SIOP 2001 study.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Sudour-Bonnange, H., van Tinteren, H., Ramírez-Villar, G.L. et al. Characteristics and outcome of synchronous bilateral Wilms tumour in the SIOP WT 2001 Study: Report from the SIOP Renal Tumour Study Group (SIOP-RTSG). Br J Cancer 131, 972–981 (2024). https://doi.org/10.1038/s41416-024-02799-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41416-024-02799-0
This article is cited by
-
Pursuit of the optimal therapeutic approach and intensity for children with bilateral Wilms tumour
British Journal of Cancer (2024)