Abstract
Background
Various prognostic factors are associated with overall survival (OS) after resection of distal cholangiocarcinoma (dCCA). The objective of this study was to develop and validate a prediction model for 3-year OS after pancreatoduodenectomy for dCCA.
Methods
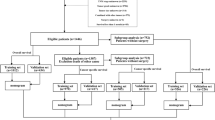
The derivation cohort consisted of all patients who underwent pancreatoduodenectomy for dCCA in the Netherlands (2009–2016). Clinically relevant variables were selected based on the Akaike information criterion using a multivariate Cox proportional hazards regression model, with model performance being assessed by concordance index (C-index) and calibration plots. External validation was performed using patients from the Belgium Cancer Registry (2008–2016), and patients from two university hospitals of Southampton (U.K.) and Verona (Italy).
Results
Independent prognostic factors for OS in the derivation cohort of 454 patients after pancreatoduodenectomy for dCCA were age (HR 1.02, 95% CI 1.01–1.03), pT (HR 1.43, 95% CI 1.07–1.90) and pN category (pN1: HR 1.78, 95% CI 1.37–2.32; pN2: HR 2.21, 95% CI 1.63–3.01), resection margin status (HR 1.79, 95% CI 1.39–2.29) and tumour differentiation (HR 2.02, 95% CI 1.62–2.53). The prediction model was based on these prognostic factors. The optimism-adjusted C-indices were similar in the derivation cohort (0.69), and in the Belgian (0.66) and Southampton-Verona (0.68) validation cohorts. Calibration was accurate in the Belgian validation cohort (slope = 0.93, intercept = 0.12), but slightly less optimal in the Southampton-Verona validation cohort (slope = 0.88, intercept = 0.32). Based on this model, three risk groups with different prognoses were identified (3-year OS of 65.4%, 33.2% and 11.8%).
Conclusions
The prediction model for 3-year OS after resection of dCCA had reasonable performance in both the derivation and geographically external validation cohort. Calibration slightly differed between validation cohorts. The model is readily available via www. pancreascalculator.com to inform patients from Western European countries on their prognosis, and may be used to stratify patients for clinical trials.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 24 print issues and online access
$259.00 per year
only $10.79 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
Data availability
Anonymous individual data could be requested from the corresponding author.
References
Rizvi S, Khan SA, Hallemeier CL, Kelley RK, Gores GJ. Cholangiocarcinoma—evolving concepts and therapeutic strategies. Nat Rev Clin Oncol. 2018;15:95–111.
Razumilava N, Gores GJ. Classification, diagnosis, and management of cholangiocarcinoma. Clin Gastroenterol Hepatol. 2013;11:13–21 e11.
Roos E, Strijker M, Franken LC, Busch OR, van Hooft JE, Klumpen HJ, et al. Comparison of short- and long-term outcomes between anatomical subtypes of resected biliary tract cancer in a Western high-volume center. HPB (Oxf). 2020;22:405–14.
Zhou Y, Liu S, Wu L, Wan T. Survival after surgical resection of distal cholangiocarcinoma: a systematic review and meta-analysis of prognostic factors. Asian J Surg. 2017;40:129–38.
Byrling J, Andersson R, Sasor A, Lindell G, Ansari D, Nilsson J, et al. Outcome and evaluation of prognostic factors after pancreaticoduodenectomy for distal cholangiocarcinoma. Ann Gastroenterol. 2017;30:571–7.
Wellner UF, Shen Y, Keck T, Jin W, Xu Z. The survival outcome and prognostic factors for distal cholangiocarcinoma following surgical resection: a meta-analysis for the 5-year survival. Surg Today. 2017;47:271–9.
Belkouz A, Wilmink JW, Haj Mohammad N, Hagendoorn J, de Vos-Geelen J, Dejong CHC, et al. Advances in adjuvant therapy of biliary tract cancer: an overview of current clinical evidence based on phase II and III trials. Crit Rev Oncol Hematol. 2020;151:102975.
Shroff RT, Kennedy EB, Bachini M, Bekaii-Saab T, Crane C, Edeline J, et al. Adjuvant therapy for resected biliary tract cancer: ASCO Clinical Practice Guideline. J Clin Oncol. 2019;37:1015–27.
Primrose JN, Fox RP, Palmer DH, Malik HZ, Prasad R, Mirza D, et al. Capecitabine compared with observation in resected biliary tract cancer (BILCAP): a randomised, controlled, multicentre, phase 3 study. Lancet Oncol. 2019;20:663–73.
Groot Koerkamp B, Wiggers JK, Gonen M, Doussot A, Allen PJ, Besselink MGH, et al. Survival after resection of perihilar cholangiocarcinoma-development and external validation of a prognostic nomogram. Ann Oncol. 2015;26:1930–5.
Hyder O, Marques H, Pulitano C, Marsh JW, Alexandrescu S, Bauer TW, et al. A nomogram to predict long-term survival after resection for intrahepatic cholangiocarcinoma: an Eastern and Western experience. JAMA Surg. 2014;149:432–8.
Moons KGM, Wolff RF, Riley RD, Whiting PF, Westwood M, Collins GS, et al. PROBAST: A tool to assess risk of bias and applicability of prediction model studies: explanation and elaboration. Ann Intern Med. 2019;170:W1–W33.
Collins GS, Reitsma JB, Altman DG, Moons KG. Transparent Reporting of a multivariable prediction model for Individual Prognosis or Diagnosis (TRIPOD): the TRIPOD statement. Ann Intern Med. 2015;162:55–63.
Strijker M, Belkouz A, van der Geest LG, van Gulik TM, van Hooft JE, de Meijer VE, et al. Treatment and survival of resected and unresected distal cholangiocarcinoma: a nationwide study. Acta Oncol. 2019;58:1048–55.
Frosio F, Mocchegiani F, Conte G, Bona ED, Vecchi A, Nicolini D, et al. Neoadjuvant therapy in the treatment of hilar cholangiocarcinoma: review of the literature. World J Gastrointest Surg. 2019;11:279–86.
Seita K, Ebata T, Mizuno T, Maeda A, Yamaguchi R, Kurumiya Y, et al. Phase 2 trial of adjuvant chemotherapy with S - 1 for node-positive biliary tract cancer (N-SOG 09). Ann Surg Oncol. 2020;27:2348–56.
Ebata T, Hirano S, Konishi M, Uesaka K, Tsuchiya Y, Ohtsuka M, et al. Randomized clinical trial of adjuvant gemcitabine chemotherapy versus observation in resected bile duct cancer. Br J Surg. 2018;105:192–202.
Edeline J, Benabdelghani M, Bertaut A, Watelet J, Hammel P, Joly JP, et al. Gemcitabine and oxaliplatin chemotherapy or surveillance in resected biliary tract cancer (PRODIGE 12-ACCORD 18-UNICANCER GI): a randomized phase III study. J Clin Oncol. 2019;37:658–67.
Nakachi K, Konishi M, Ikeda M, Mizusawa J, Eba J, Okusaka T, et al. A randomized Phase III trial of adjuvant S-1 therapy vs. observation alone in resected biliary tract cancer: Japan Clinical Oncology Group Study (JCOG1202, ASCOT). Jpn J Clin Oncol. 2018;48:392–5.
Stein A, Arnold D, Bridgewater J, Goldstein D, Jensen LH, Klumpen HJ, et al. Adjuvant chemotherapy with gemcitabine and cisplatin compared to observation after curative intent resection of cholangiocarcinoma and muscle invasive gallbladder carcinoma (ACTICCA-1 trial)—a randomized, multidisciplinary, multinational phase III trial. BMC Cancer. 2015;15:564.
Moon A, Choi DW, Choi SH, Heo JS, Jang KT. Validation of T stage according to depth of invasion and N stage subclassification based on number of metastatic lymph nodes for distal extrahepatic bile duct (EBD) carcinoma. Medicine (Baltim). 2015;94:e2064.
Postlewait LM, Ethun CG, Le N, Pawlik TM, Buettner S, Poultsides G, et al. Proposal for a new T-stage classification system for distal cholangiocarcinoma: a 10-institution study from the U.S. Extrahepatic Biliary Malignancy Consortium. HPB (Oxf). 2016;18:793–9.
Li X, Lin H, Sun Y, Gong J, Feng H, Tu J. Prognostic significance of the lymph node ratio in surgical patients with distal cholangiocarcinoma. J Surg Res. 2019;236:2–11.
Gottlieb-Vedi E, Mattsson F, Lagergren P, Lagergren J. Annual hospital volume of surgery for gastrointestinal cancer in relation to prognosis. Eur J Surg Oncol. 2019;45:1839–46.
Komaya K, Ebata T, Shirai K, Ohira S, Morofuji N, Akutagawa A, et al. Recurrence after resection with curative intent for distal cholangiocarcinoma. Br J Surg. 2017;104:426–33.
Strijker M, Chen JW, Mungroop TH, Jamieson NB, van Eijck CH, Steyerberg EW, et al. Systematic review of clinical prediction models for survival after surgery for resectable pancreatic cancer. Br J Surg. 2019;106:342–54.
Acknowledgements
This study was conducted on behalf of the Dutch Pancreatic Cancer Group (DPCG).
Funding
This research did not receive any specific grant from funding agencies in the public, commercial or not-for-profit sectors.
Author information
Authors and Affiliations
Consortia
Contributions
AB, SR, IB, CV, CP, MB and HK designed the study. AB, SR, CD, LD, A. Balduzzi, A.B.C. and a trained registry administrator from the BCR collected and updated the necessary data. A.B. performed the statistical analyses with support from S.R. and M.O. which were interpreted by all authors. A.B. drafted the manuscript and all authors revised the manuscript and approved the final manuscript for publication.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethical approval and consent to participate
This study was approved by the Dutch Pancreatic Cancer Group. We received a waiver for ethical approval of this study by the Medical Ethics Review Committee at Amsterdam UMC, location Academic Medical Center (W19_191 # 19.230).
Consent to publish
This manuscript does not contain any individual person’s data in any form.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
About this article
Cite this article
Belkouz, A., Van Roessel, S., Strijker, M. et al. Development and external validation of a prediction model for overall survival after resection of distal cholangiocarcinoma. Br J Cancer 126, 1280–1288 (2022). https://doi.org/10.1038/s41416-021-01687-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41416-021-01687-1
This article is cited by
-
Laparoscopic Versus Open Pancreatoduodenectomy for Periampullary Tumors: A Systematic Review and Meta-Analysis of Randomized Controlled Trials
Journal of Gastrointestinal Cancer (2024)
-
The road to tailored adjuvant chemotherapy for all four non-pancreatic periampullary cancers: An international multimethod cohort study
British Journal of Cancer (2024)
-
The clinical implication of minimally invasive versus open pancreatoduodenectomy for non-pancreatic periampullary cancer: a systematic review and individual patient data meta-analysis
Langenbeck's Archives of Surgery (2023)