Abstract
Background
Hypertension and proteinuria are common bevacizumab-induced toxicities. No validated biomarkers are available for identifying patients at risk of these toxicities.
Methods
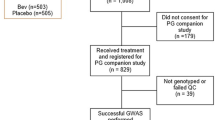
A genome-wide association study (GWAS) meta-analysis was performed in 1039 bevacizumab-treated patients of European ancestry in four clinical trials (CALGB 40502, 40503, 80303, 90401). Grade ≥2 hypertension and proteinuria were recorded (CTCAE v.3.0). Single-nucleotide polymorphism (SNP)-toxicity associations were determined using a cause-specific Cox model adjusting for age and sex.
Results
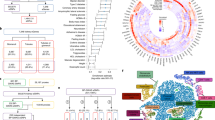
The most significant SNP associated with hypertension with concordant effect in three out of the four studies (p-value <0.05 for each study) was rs6770663 (A > G) in KCNAB1, with the G allele increasing the risk of hypertension (p-value = 4.16 × 10−6). The effect of the G allele was replicated in ECOG-ACRIN E5103 in 582 patients (p-value = 0.005). The meta-analysis of all five studies for rs6770663 led to p-value = 7.73 × 10−8, close to genome-wide significance. The most significant SNP associated with proteinuria was rs339947 (C > A, between DNAH5 and TRIO), with the A allele increasing the risk of proteinuria (p-value = 1.58 × 10−7).
Conclusions
The results from the largest study of bevacizumab toxicity provide new markers of drug safety for further evaluations. SNP in KCNAB1 validated in an independent dataset provides evidence toward its clinical applicability to predict bevacizumab-induced hypertension.
ClinicalTrials.gov Identifier: NCT00785291 (CALGB 40502); NCT00601900 (CALGB 40503); NCT00088894 (CALGB 80303) and NCT00110214 (CALGB 90401).
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 24 print issues and online access
$259.00 per year
only $10.79 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
Data availability
The datasets generated during and/or analysed during the current study are available in the NHGRI-EBI GWAS Catalog under study accession numbers GCST90026609, GCST90026610 and GCST90026611.
Change history
01 December 2021
A Correction to this paper has been published: https://doi.org/10.1038/s41416-021-01617-1
References
Ferrara N, Adamis AP. Ten years of anti-vascular endothelial growth factor therapy. Nat Rev Drug Disov. 2016;15:385–403.
FDA AVASTIN® Prescribing Information. 2020. https://www.accessdata.fda.gov/drugsatfda_docs/label/2020/125085s332lbl.pdf. Accessed on 05 Oct 2020.
Ellis LM, Kirkpatrick P. Bevacizumab. Nat Rev Drug Discov. 2005;3:995–996.
Zhu X, Wu S, Dahut WL, Parikh CR. Risks of proteinuria and hypertension with bevacizumab, an antibody against vascular endothelial growth factor: systematic review and meta-analysis. Am J Kidney Dis. 2007;49:186–93.
Izzedine H, Ederhy S, Goldwasser F, Soria JC, Milano G, Cohen A, et al. Management of hypertension in angiogenesis inhibitor-treated patients. Ann Oncol. 2009;20:807–15.
Mir O, Coriat R, Cabanes L, Ropert S, Billemont B, Alexandre J, et al. An observational study of bevacizumab-induced hypertension as a clinical biomarker of antitumor activity. Oncologist. 2011;16:1325–32.
Morita S, Uehara K, Nakayama G, Shibata T, Oguri T, Inada-Inoue M, et al. Association between bevacizumab-related hypertension and vascular endothelial growth factor (VEGF) gene polymorphisms in Japanese patients with metastatic colorectal cancer. Cancer Chemother Pharmacol. 2013;71:405–11.
Schneider BP, Wang MM, Radovich M, Sledge GW, Badve S, Thor A, et al. Association of vascular endothelial growth factor and vascular endothelial growth factor receptor-2 genetic polymorphisms with outcome in a trial of paclitaxel compared with paclitaxel plus bevacizumab in advanced breast cancer: ECOG 2100. J Clin Oncol. 2008;28:4672–4678.
Schneider BP, Li L, Shen F, Miller KD, Radovich M, O’Neill A, et al. Genetic variant predicts bevacizumab-induced hypertension in ECOG-5103 and ECOG-2100. Br J Cancer. 2014;111:1241–1248.
Kindler HL, Niedzwiecki D, Hollis D, Sutherland S, Schrag D, Hurwitz H, et al. Gemcitabine plus bevacizumab compared with gemcitabine plus placebo in patients with advanced pancreatic cancer: Phase III trial of the Cancer and Leukemia Group B (CALGB 80303). J Clin Oncol. 2010;28:3617–22.
Dickler MN, Barry WT, Cirrincione CT, Ellis MJ, Moynahan ME, Innocenti F, et al. Phase III trial evaluating letrozole as first-line endocrine therapy with or without bevacizumab for the treatment of postmenopausal women with hormone receptor-positive advanced-stage breast cancer: CALGB 40503 (Alliance). J Clin Oncol. 2016;34:2602–2609.
Kelly WK, Halabi S, Carducci M, George D, Mahoney JF, Stadler WM, et al. Randomized, double-blind, placebo-controlled phase III trial comparing docetaxel and prednisone with or without bevacizumab in men with metastatic castration-resistant prostate cancer: CALGB 90401. J Clin Oncol. 2012;30:1534–40.
Rugo HS, Barry WT, Moreno-Aspitia A, Lyss AP, Cirrincione C, Leung E, et al. Randomized phase III trial of paclitaxel once per week compared with nanoparticle albumin-bound nab-paclitaxel once per week or ixabepilone with bevacizumab as first-line chemotherapy for locally recurrent or metastatic breast cancer: CALGB 40502/NCCTG N0. J Clin Oncol. 2015;33:2361–2369.
National Institute of Cancer (NCI) Guidelines for Investigators. 2021. https://ctep.cancer.gov/protocoldevelopment/electronic_applications/docs/aeguidelines.pdf. Accessed on 08 Jan 2021.
Innocenti F, Jiang C, Sibley AB, Denning S, Etheridge AS, Watson D, et al. An initial genetic analysis of gemcitabine-induced high-grade neutropenia in pancreatic cancer patients in CALGB 80303 (Alliance). Pharmacogenet Genomics. 2019;29:123–31.
Innocenti F, Owzar K, Cox NL, Evans P, Kubo M, Zembutsu H, et al. A genome-wide association study of overall survival in pancreatic cancer patients treated with gemcitabine in CALGB 80303. Clin Cancer Res. 2012;18:577–84.
Rashkin SR, Chua KC, Ho C, Mulkey F, Jiang C, Mushiroda T, et al. A pharmacogenetic prediction model of progression-free survival in breast cancer using genome-wide genotyping data from CALGB 40502 (Alliance). Clin Pharmacol Ther. 2019;105:738–45.
Hertz DL, Owzar K, Lessans S, Wing C, Jiang C, Kelly WK, et al. Pharmacogenetic discovery in CALGB (alliance) 90401 and mechanistic validation of a VAC14 polymorphism that increases risk of docetaxel-induced neuropathy. Clin Cancer Res. 2016;22:4890–4900.
Price AL, Patterson NJ, Plenge RM, Weinblatt ME, Shadick NA, Reich D. Principal components analysis corrects for stratification in genome-wide association studies. Nat Genet. 2006;38:904–909.
Zhang W, et al. SCAN database: Facilitating integrative analyses of cytosine modification and expression QTL. Database. 2015;27:bav025.
Machiela MJ, Chanock SJ. LDlink: A web-based application for exploring population-specific haplotype structure and linking correlated alleles of possible functional variants. Bioinformatics. 2015;31:3555–3557.
James Kent W, Sugnet CW, Furey TS, Roskin KM, Pringle TH, Zahler AM, et al. The human genome browser at UCSC. Genome Res. 2002;12:996–1006.
Boyle AP, Hong EL, Hariharan M, Cheng Y, Schaub MA, Kasowski M, et al. Annotation of functional variation in personal genomes using RegulomeDB. Genome Res. 2012;22:1790–1797.
Ward LD, Kellis M. HaploReg: A resource for exploring chromatin states, conservation, and regulatory motif alterations within sets of genetically linked variants. Nucleic Acids Res. 2012;40:D930–D934.
GTEx Consortium. The Genotype-Tissue Expression (GTEx) pilot analysis: multitissue gene regulation in humans. Science. 2015;348:648–660.
Gillies CE, Putler R, Menon R, Otto E, Yasutake K, Nair V, et al. An eQTL landscape of kidney tissue in human nephrotic syndrome. Am J Hum Genet. 2018;103:232–444.
Thul PJ, Akesson L, Wiking M, Mahdessian D, Geladaki A, Ait Blal H, et al. A subcellular map of the human proteome. Science. 2017;356:eaal3321.
González C, Baez-Nieto D, Valencia I, Oyarzún I, Rojas P, Naranjo D, et al. K+ channels: Function-structural overview. Compr Physiol. 2012;2:2087–2149.
Tipparaju SM, Liu SQ, Barski OA, Bhatnagar A. NADPH binding to β-subunit regulates inactivation of voltage-gated K+ channels. Biochem Biophys Res Commun. 2007;359:269–276.
Sobey CG. Potassium channel function in vascular disease. Arterioscler Thromb Vasc Biol. 2001;21:28–38.
Martens JR, Gelband CH. Alterations in rat interlobar artery membrane potential and K+ channels in genetic and nongenetic hypertension. Circ Res. 1996;79:295–301.
Banerjee B, Peiris DN, Koo SH, Chui P, Lee EJD, Hande MP. Genomic imbalances in key ion channel genes and telomere shortening in sudden cardiac death victims. Cytogenet Genome Res. 2009;122:350–355.
Tur J, Chapalamadugu KC, Padawer T, Badole SL, Kilfoil PJ, Bhatnagar A, et al. Deletion of Kvβ1.1 subunit leads to electrical and haemodynamic changes causing cardiac hypertrophy in female murine hearts. Exp Physiol. 2016;101:494–508.
Kulakovskiy IV, Vorontsov IE, Yevshin IS, Sharipov RN, Fedorova AD, Rumynskiy EI, et al. HOCOMOCO: Towards a complete collection of transcription factor binding models for human and mouse via large-scale ChIP-Seq analysis. Nucleic Acids Res. 2018;46:D252–D259.
Niu Z, Li A, Zhang SX, Schwartz RJ. Serum response factor micromanaging cardiogenesis. Curr Opin Cell Biol. 2007;19:618–627.
Rouillard AD, Gundersen GW, Fernandez NF, Wang Z, Monteiro CD, McDermott MG, et al. The harmonizome: a collection of processed datasets gathered to serve and mine knowledge about genes and proteins. Database (Oxford). 2016;2016:baw100.
Sherry ST, Ward MH, Kholodov M, Baker J, Phan L, Smigielski EM, et al. dbSNP: the NCBI database of genetic variation. Nucleic Acids Res. 2001;29:308–311.
Leigh MW, Pittman JE, Carson JL, Ferkol TW, Dell SD, Davis SD, et al. Clinical and genetic aspects of primary ciliary dyskinesia/kartagener syndrome. Genet Med. 2009;11:473–487.
Maier M, Baldwin C, Aoudjit L, Takano T. The role of trio, a rho guanine nucleotide exchange factor, in glomerular podocytes. Int J Mol Sci. 2018;19:E479.
Brahmer JR, Dahlberg SE, Gray RJ, Schiller JH, Perry MC, Sandler A, et al. Sex differences in outcome with bevacizumab therapy: analysis of patients with advanced-stage non-small cell lung cancer treated with or without bevacizumab in combination with paclitaxel and carboplatin in the eastern cooperative oncology group trial 4599. J Thorac Oncol. 2011;6:1031–1038.
Schneider BP, Li L, Radovich M, Shen F, Miller KD, Flockhart DA, et al. Genome-wide association studies for taxane-induced peripheral neuropathy in ECOG-5103 and ECOG-1199. Clin Cancer Res. 2015;21:5082–5091.
Baldwin RM, Owzar K, Zembutsu H, Chhibber A, Kubo M, Jiang C, et al. A genome-wide association study identifies novel loci for paclitaxel-induced sensory peripheral neuropathy in CALGB 40101. Clin Cancer Res. 2012;18:5099–5109.
Schirmer MA, Lüske CM, Roppel S, Schaudinn A, Zimmer C, Pflüger R, et al. Relevance of Sp binding site polymorphism in WWOX for treatment outcome in pancreatic cancer. J Natl Cancer Inst. 2016;108:djv387.
Acknowledgements
We acknowledge the PI of ECOG-ACRIN E5103, Kathy Miller, MD.
Funding
This work was supported by the National Cancer Institute of the National Institutes of Health under Award Numbers U10CA180821 and U10CA180882 (to the Alliance for Clinical Trials in Oncology), U24CA196171, UG1CA233253, UG1CA233327 and UG1CA233373. JCFQ was supported by the São Paulo Research Foundation-FAPESP (2018/04491-2). Also supported in part by funds from Abraxis BioScience, Bristol Meyers Squibb, Celgene and Genentech. The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Institutes of Health. https://acknowledgments.alliancefound.org
Author information
Authors and Affiliations
Contributions
JCFQ, ASE and FI wrote the manuscript; FI designed the research; JCFQ, JW, ABS, CJ, ASE, FS, FM, JNP, DLH, ECD, HLM, MB, HR, HLK, WKK, MJR, DLK, KO, BS, DL and F.I. performed the research; JCFQ, JW, ABS, CJ, ASE, GJ, KO and FI analyzed the data; JW, ABS, CJ, ASE and DL contributed new reagents/analytical tools.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
All participants provided written informed consent for sample collection and pharmacogenetic analysis, and all trials were conducted in accordance with recognised ethical guidelines. The study was performed in accordance with the Declaration of Helsinki and was approved by the local IRB.
Consent for publication
Not applicable.
Competing interests
JCFQ, JW, DL, KO and FI are coinventors of a patent application, serial number 16/932,002. FI is an advisor for Emerald Lake Safety. These relationships have been disclosed to and are under management by UNC-Chapel Hill.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
The original online version of this article was revised: Due to an error in an affiliation.
Supplementary information
Rights and permissions
About this article
Cite this article
Quintanilha, J.C.F., Wang, J., Sibley, A.B. et al. Bevacizumab-induced hypertension and proteinuria: a genome-wide study of more than 1000 patients. Br J Cancer 126, 265–274 (2022). https://doi.org/10.1038/s41416-021-01557-w
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41416-021-01557-w
This article is cited by
-
Safety of first-line systemic therapy in patients with metastatic colorectal cancer: a network meta-analysis of randomized controlled trials
BMC Cancer (2024)
-
Contribution of plasma levels of VEGF-A and angiopoietin-2 in addition to a genetic variant in KCNAB1 to predict the risk of bevacizumab-induced hypertension
The Pharmacogenomics Journal (2024)
-
Efficacy and safety of ramucirumab for gastric or gastro-esophageal junction adenocarcinoma: a systematic review and meta-analysis
European Journal of Clinical Pharmacology (2024)
-
PIK3R5 genetic predictors of hypertension induced by VEGF-pathway inhibitors
The Pharmacogenomics Journal (2022)
-
Combination of Bevacizumab and Osimertinib in Patients with EGFR T790M-Mutated Non-small Cell Lung Cancer
Clinical Drug Investigation (2022)