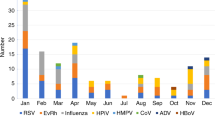
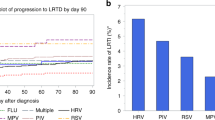
Abstract
We examined associations between specific antibiotic exposures and progression to lower respiratory tract disease (LRTD) following individual respiratory viral infections (RVIs) after hematopoietic cell transplantation (HCT). We analyzed allogeneic HCT recipients of all ages with their first RVI during the first 100 days post-HCT. For the 21 days before RVI onset, we recorded any receipt of specific groups of antibiotics, and the cumulative sum of the number of antibiotics received for each day (antibiotic-days). We used Cox proportional hazards models to assess the relationship between antibiotic exposure and progression to LRTD. Among 469 patients with RVI, 124 progressed to LRTD. Compared to no antibiotics, use of antibiotics with broad anaerobic activity in the prior 21 days was associated with progression to LRTD after adjusting for age, virus type, hypoalbuminemia, neutropenia, steroid use, and monocytopenia (HR 2.2, 95% CI 1.1–4.1). Greater use of those antibiotics (≥7 antibiotic days) was also associated with LRTD in adjusted models (HR 2.2, 95% CI 1.1–4.3). Results were similar after adjusting for lymphopenia instead of monocytopenia. Antibiotic use is associated with LRTD after RVI across different viruses in HCT recipients. Prospective studies using anaerobe-sparing antibiotics should be explored to assess impact on LRTD in patients undergoing HCT.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
Data availability
Data supporting the findings from this study are available from the corresponding author upon reasonable request.
References
Pinana JL, Gomez MD, Perez A, Madrid S, Balaguer-Rosello A, Gimenez E, et al. Community-acquired respiratory virus lower respiratory tract disease in allogeneic stem cell transplantation recipient: Risk factors and mortality from pulmonary virus-bacterial mixed infections. Transpl Infect Dis. 2018;20:e12926.
Khanna N, Steffen I, Studt JD, Schreiber A, Lehmann T, Weisser M, et al. Outcome of influenza infections in outpatients after allogeneic hematopoietic stem cell transplantation. Transpl Infect Dis. 2009;11:100–5.
Khanna N, Widmer AF, Decker M, Steffen I, Halter J, Heim D, et al. Respiratory syncytial virus infection in patients with hematological diseases: single-center study and review of the literature. Clin Infect Dis. 2008;46:402–12.
Spahr Y, Tschudin-Sutter S, Baettig V, Compagno F, Tamm M, Halter J, et al. Community-Acquired Respiratory Paramyxovirus Infection After Allogeneic Hematopoietic Cell Transplantation: A Single-Center Experience. Open Forum Infect Dis. 2018;5:ofy077.
Vakil E, Sheshadri A, Faiz SA, Shah DP, Zhu Y, Li L, et al. Risk factors for mortality after respiratory syncytial virus lower respiratory tract infection in adults with hematologic malignancies. Transpl Infect Dis. 2018;20:e12994.
Ogimi C, Krantz EM, Golob JL, Waghmare A, Liu C, Leisenring WM, et al. Antibiotic Exposure Prior to Respiratory Viral Infection Is Associated with Progression to Lower Respiratory Tract Disease in Allogeneic Hematopoietic Cell Transplant Recipients. Biol Blood Marrow Transpl. 2018;24:2293–301.
Bergeron A, Chevret S, Granata A, Chevallier P, Vincent L, Huynh A, et al. Effect of Azithromycin on Airflow Decline-Free Survival After Allogeneic Hematopoietic Stem Cell Transplant: The ALLOZITHRO Randomized Clinical Trial. JAMA. 2017;318:557–66.
Bradley KC, Finsterbusch K, Schnepf D, Crotta S, Llorian M, Davidson S, et al. Microbiota-Driven Tonic Interferon Signals in Lung Stromal Cells Protect from Influenza Virus Infection. Cell Rep. 2019;28:245–56.e244.
Steed AL, Christophi GP, Kaiko GE, Sun L, Goodwin VM, Jain U, et al. The microbial metabolite desaminotyrosine protects from influenza through type I interferon. Science. 2017;357:498–502.
Ichinohe T, Pang IK, Kumamoto Y, Peaper DR, Ho JH, Murray TS, et al. Microbiota regulates immune defense against respiratory tract influenza A virus infection. Proc Natl Acad Sci USA 2011;108:5354–9.
Vissers M, de Groot R, Ferwerda G. Severe viral respiratory infections: are bugs bugging? Mucosal Immunol. 2014;7(Mar):227–38.
Wu S, Jiang ZY, Sun YF, Yu B, Chen J, Dai CQ, et al. Microbiota regulates the TLR7 signaling pathway against respiratory tract influenza A virus infection. Curr Microbiol. 2013;67:414–22.
Abt MC, Osborne LC, Monticelli LA, Doering TA, Alenghat T, Sonnenberg GF, et al. Commensal bacteria calibrate the activation threshold of innate antiviral immunity. Immunity. 2012;37:158–70.
Lee JR, Huang J, Magruder M, Zhang LT, Gong C, Sholi AN, et al. Butyrate-producing gut bacteria and viral infections in kidney transplant recipients: A pilot study. Transpl Infect Dis. 2019;21:e13180.
Haak BW, Littmann ER, Chaubard JL, Pickard AJ, Fontana E, Adhi F, et al. Impact of gut colonization with butyrate-producing microbiota on respiratory viral infection following allo-HCT. Blood. 2018;131:2978–86.
Holler E, Butzhammer P, Schmid K, Hundsrucker C, Koestler J, Peter K, et al. Metagenomic analysis of the stool microbiome in patients receiving allogeneic stem cell transplantation: loss of diversity is associated with use of systemic antibiotics and more pronounced in gastrointestinal graft-versus-host disease. Biol Blood Marrow Transpl. 2014;20:640–5.
Shono Y, Docampo MD, Peled JU, Perobelli SM, Velardi E, Tsai JJ, et al. Increased GVHD-related mortality with broad-spectrum antibiotic use after allogeneic hematopoietic stem cell transplantation in human patients and mice. Sci Transl Med. 2016;8:339ra371.
Taur Y, Pamer EG. The intestinal microbiota and susceptibility to infection in immunocompromised patients. Curr Opin Infect Dis. 2013;26:332–7.
Shono Y. Gut microbiota and graft-versus-host disease: broad-spectrum antibiotic use increases post-allogeneic hematopoietic stem cell transplant graft-versus-host disease-related mortality. Rinsho Ketsueki. 2017;58:835–42.
Routy B, Letendre C, Enot D, Chenard-Poirier M, Mehraj V, Seguin NC, et al. The influence of gut-decontamination prophylactic antibiotics on acute graft-versus-host disease and survival following allogeneic hematopoietic stem cell transplantation. Oncoimmunology. 2017;6:e1258506.
Golob JL, Pergam SA, Srinivasan S, Fiedler TL, Liu C, Garcia K, et al. Stool Microbiota at Neutrophil Recovery Is Predictive for Severe Acute Graft vs Host Disease After Hematopoietic Cell Transplantation. Clin Infect Dis. 2017;65:1984–91.
Romick-Rosendale LE, Haslam DB, Lane A, Denson L, Lake K, Wilkey A, et al. Antibiotic Exposure and Reduced Short Chain Fatty Acid Production after Hematopoietic Stem Cell Transplant. Biol Blood Marrow Transpl. 2018;24:2418–24.
Brook I. Treatment of anaerobic infection. Expert Rev Anti-Infective Ther. 2007;5:991–1006.
Zimmermann P, Curtis N. The effect of antibiotics on the composition of the intestinal microbiota - a systematic review. J Infect. 2019;79:471–89.
Weber D, Jenq RR, Peled JU, Taur Y, Hiergeist A, Koestler J, et al. Microbiota Disruption Induced by Early Use of Broad-Spectrum Antibiotics Is an Independent Risk Factor of Outcome after Allogeneic Stem Cell Transplantation. Biol Blood Marrow Transpl. 2017;23:845–52.
Hirsch HH, Martino R, Ward KN, Boeckh M, Einsele H, Ljungman P. Fourth European Conference on Infections in Leukaemia (ECIL-4): guidelines for diagnosis and treatment of human respiratory syncytial virus, parainfluenza virus, metapneumovirus, rhinovirus, and coronavirus. Clin Infect Dis. 2013;56:258–66.
Seo S, Waghmare A, Scott EM, Xie H, Kuypers JM, Hackman RC, et al. Human rhinovirus detection in the lower respiratory tract of hematopoietic cell transplant recipients: association with mortality. Haematologica. 2017;102:1120–30.
Campbell AP, Guthrie KA, Englund JA, Farney RM, Minerich EL, Kuypers J, et al. Clinical outcomes associated with respiratory virus detection before allogeneic hematopoietic stem cell transplant. Clin Infect Dis. 2015;61:192–202.
Kuypers J, Wright N, Corey L, Morrow R. Detection and quantification of human metapneumovirus in pediatric specimens by real-time RT-PCR. J Clin Virol. 2005;33:299–305.
Kuypers J, Wright N, Morrow R. Evaluation of quantitative and type-specific real-time RT-PCR assays for detection of respiratory syncytial virus in respiratory specimens from children. J Clin Virol. 2004;31:123–9.
Kim Y, Waghmare A, Xie H, Holmberg LA, Pergam SA, Jerome KR, et al. Respiratory viruses in hematopoietic cell transplant candidates: impact of preexisting lower tract disease on outcomes. Blood Adv. 2022. https://doi.org/10.1182/bloodadvances.2021004915.
Weber D, Oefner PJ, Hiergeist A, Koestler J, Gessner A, Weber M, et al. Low urinary indoxyl sulfate levels early after transplantation reflect a disrupted microbiome and are associated with poor outcome. Blood. 2015;126:1723–8.
Taur Y, Xavier JB, Lipuma L, Ubeda C, Goldberg J, Gobourne A, et al. Intestinal domination and the risk of bacteremia in patients undergoing allogeneic hematopoietic stem cell transplantation. Clin Infect Dis. 2012;55:905–14.
Koh AY. The microbiome in hematopoietic stem cell transplant recipients and cancer patients: Opportunities for clinical advances that reduce infection. PLoS Pathog. 2017;13:e1006342.
Whangbo J, Ritz J, Bhatt A. Antibiotic-mediated modification of the intestinal microbiome in allogeneic hematopoietic stem cell transplantation. Bone Marrow Transpl. 2017;52:183–90.
Chemaly RF, Shah DP, Boeckh MJ. Management of respiratory viral infections in hematopoietic cell transplant recipients and patients with hematologic malignancies. Clin Infect Dis. 2014;59:S344–351.
Shahani L, Ariza-Heredia EJ, Chemaly RF. Antiviral therapy for respiratory viral infections in immunocompromised patients. Expert Rev Anti-Infective Ther. 2017;15:401–15.
Seo S, Xie H, Campbell AP, Kuypers JM, Leisenring WM, Englund JA, et al. Parainfluenza virus lower respiratory tract disease after hematopoietic cell transplant: viral detection in the lung predicts outcome. Clin Infect Dis. 2014;58:1357–68.
Kim YJ, Guthrie KA, Waghmare A, Walsh EE, Falsey AR, Kuypers J, et al. Respiratory syncytial virus in hematopoietic cell transplant recipients: factors determining progression to lower respiratory tract disease. J Infect Dis. 2014;209:1195–204.
Seo S, Gooley TA, Kuypers JM, Stednick Z, Jerome KR, Englund JA, et al. Human Metapneumovirus Infections Following Hematopoietic Cell Transplantation: Factors Associated With Disease Progression. Clin Infect Dis. 2016;63:178–85.
Shah DP, Shah PK, Azzi JM, Chemaly RF. Parainfluenza virus infections in hematopoietic cell transplant recipients and hematologic malignancy patients: A systematic review. Cancer Lett. 2016;370:358–64.
Waghmare A, Xie H, Kuypers J, Sorror ML, Jerome KR, Englund JA, et al. Human Rhinovirus Infections in Hematopoietic Cell Transplant Recipients: Risk Score for Progression to Lower Respiratory Tract Infection. Biol Blood Marrow Transplant. 2019;25:1011–21.
Ogimi C, Xie H, Waghmare A, Jerome KR, Leisenring WM, Milano F, et al. Correlation of initial upper respiratory tract viral burden with progression to lower tract disease in adult allogeneic hematopoietic cell transplant recipients. J Clin Virol. 2022;150–1:105152.
Eichenberger EM, Soave R, Zappetti D, Small CB, Shore T, van Besien K, et al. Incidence, significance, and persistence of human coronavirus infection in hematopoietic stem cell transplant recipients. Bone Marrow Transpl. 2019;54:1058–66.
Thackray LB, Handley SA, Gorman MJ, Poddar S, Bagadia P, Briseno CG, et al. Oral Antibiotic Treatment of Mice Exacerbates the Disease Severity of Multiple Flavivirus Infections. Cell Rep. 2018;22:3440–53.e3446.
Doan T, Arzika AM, Ray KJ, Cotter SY, Kim J, Maliki R, et al. Gut Microbial Diversity in Antibiotic-Naive Children After Systemic Antibiotic Exposure: A Randomized Controlled Trial. Clin Infect Dis. 2017;64:1147–53.
Parker EPK, Praharaj I, John J, Kaliappan SP, Kampmann B, Kang G, et al. Changes in the intestinal microbiota following the administration of azithromycin in a randomised placebo-controlled trial among infants in south India. Sci Rep. 2017;7:9168.
Waghmare A, Xie H, Kimball L, Yi J, Ozkok S, Leisenring W, et al. Supplemental Oxygen-Free Days in Hematopoietic Cell Transplant Recipients With Respiratory Syncytial Virus. J Infect Dis. 2017;216:1235–44.
Sorror ML, Maris MB, Storb R, Baron F, Sandmaier BM, Maloney DG, et al. Hematopoietic cell transplantation (HCT)-specific comorbidity index: a new tool for risk assessment before allogeneic HCT. Blood. 2005;106:2912–9.
Sorror ML, Logan BR, Zhu X, Rizzo JD, Cooke KR, McCarthy PL, et al. Prospective Validation of the Predictive Power of the Hematopoietic Cell Transplantation Comorbidity Index: A Center for International Blood and Marrow Transplant Research Study. Biol Blood Marrow Transpl. 2015;21:1479–87.
Acknowledgements
We thank Chris Davis, Madeline Kesten and Ryan Basom for database services.
Funding
This work was supported by the National Institutes of Health (grant numbers K23AI139385 to CO, R01HL081595 and K24HL093294 to MB, CA18029 to WML [clinical database]); the Fred Hutchinson Cancer Center Vaccine and Infectious Disease Division (biorepository); Seattle Children’s Research Institute Clinical Research Scholars Program Award to CO.
Author information
Authors and Affiliations
Contributions
Authorship contribution: CO designed this study, assisted in analysis, interpreted results, and wrote the manuscript; EMK and WML performed the statistical analysis and wrote the manuscript; JLG, CL, AW, SAP and DNF provided clinical input, interpretation of results, and reviewed the manuscript; KRJ provided technical oversight for laboratory, and reviewed the manuscript; JAE and MB provided oversight, designed this study, interpreted results, and reviewed the manuscript.
Corresponding authors
Ethics declarations
Competing interests
AW reports grant support from Ansun Biopharma, Allovir, VB Tech, Amazon, Inc, GlaxoSmithKline and Pfizer, and is an Advisory Board Member for Kyorin Pharmaceutical. JAE reports grant support from AstraZeneca, Merck, Pfizer, GlaxoSmithKline and Novavax, an Advisory Board Member and consultant for Sanofi Pasteur, Meissa Vaccines, Teva Pharmaceuticals and Astra Zeneca. MB reports grant support from Amazon, GSK, Janssen, Gilead and VirBio, and a consultant for Allovir, Merck, Janssen, Gilead, Kyorin Pharmaceuticals, and VirBio, and an Advisory Board Member for Evrys Bio. All other authors report no potential conflicts. Conflicts that the editors consider relevant to the content of the manuscript have been disclosed.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Ogimi, C., Krantz, E.M., Golob, J.L. et al. Exposure to antibiotics with anaerobic activity before respiratory viral infection is associated with respiratory disease progression after hematopoietic cell transplant. Bone Marrow Transplant 57, 1765–1773 (2022). https://doi.org/10.1038/s41409-022-01790-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41409-022-01790-8