Abstract
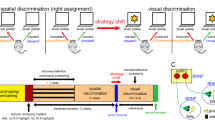
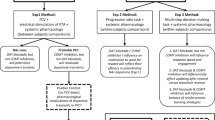
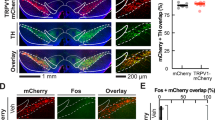
The catecholamine neuromodulators dopamine and norepinephrine are implicated in motor function, motivation, and cognition. Although roles for striatal dopamine in these aspects of behavior are well established, the specific roles for cortical catecholamines in regulating striatal dopamine dynamics and behavior are less clear. We recently showed that elevating cortical dopamine but not norepinephrine suppresses hyperactivity in dopamine transporter knockout (DAT-KO) mice, which have elevated striatal dopamine levels. In contrast, norepinephrine transporter knockout (NET-KO) mice have a phenotype distinct from DAT-KO mice, as they show elevated extracellular cortical catecholamines but reduced baseline striatal dopamine levels. Here we evaluated the consequences of altered catecholamine levels in NET-KO mice on cognitive flexibility and striatal dopamine dynamics. In a probabilistic reversal learning task, NET-KO mice showed enhanced reversal learning, which was consistent with larger phasic dopamine transients (dLight) in the dorsomedial striatum (DMS) during reward delivery and reward omission, compared to WT controls. Selective depletion of dorsal medial prefrontal cortex (mPFC) norepinephrine in WT mice did not alter performance on the reversal learning task but reduced nestlet shredding. Surprisingly, NET-KO mice did not show altered breakpoints in a progressive ratio task, suggesting intact food motivation. Collectively, these studies show novel roles of cortical catecholamines in the regulation of tonic and phasic striatal dopamine dynamics and cognitive flexibility, updating our current views on dopamine regulation and informing future therapeutic strategies to counter multiple psychiatric disorders.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 13 print issues and online access
$259.00 per year
only $19.92 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Data availability
All data related to this manuscript are stored on a University of Florida OneDrive or Dropbox account and will be made available upon request.
References
Schultz W. Getting formal with dopamine and reward. Neuron. 2002;36:241–63.
Palmiter RD. Dopamine signaling in the dorsal striatum is essential for motivated behaviors: lessons from dopamine-deficient mice. Ann N Y Acad Sci. 2008;1129:35–46. https://doi.org/10.1196/annals.1417.003.
Cox J, Witten IB. Striatal circuits for reward learning and decision-making. Nat Rev Neurosci. 2019;20:482–94. https://doi.org/10.1038/s41583-019-0189-2.
Aarts E, van Holstein M, Cools R. Striatal Dopamine and the Interface between Motivation and Cognition. Front Psychol. 2011;2:163 https://doi.org/10.3389/fpsyg.2011.00163.
Sulzer D. How addictive drugs disrupt presynaptic dopamine neurotransmission. Neuron. 2011;69:628–49. https://doi.org/10.1016/j.neuron.2011.02.010.
Abi-Dargham A. From “bedside” to “bench” and back: A translational approach to studying dopamine dysfunction in schizophrenia. Neurosci Biobehav Rev. 2018. https://doi.org/10.1016/j.neubiorev.2018.12.003.
Bernheimer H, Birkmayer W, Hornykiewicz O, Jellinger K, Seitelberger F. Brain dopamine and the syndromes of Parkinson and Huntington. Clinical, morphological and neurochemical correlations. J Neurol Sci. 1973;20:415–55.
Hitchcott PK, Quinn JJ, Taylor JR. Bidirectional modulation of goal-directed actions by prefrontal cortical dopamine. Cereb Cortex. 2007;17:2820–7. https://doi.org/10.1093/cercor/bhm010.
Barker JM, Torregrossa MM, Taylor JR. Bidirectional modulation of infralimbic dopamine D1 and D2 receptor activity regulates flexible reward seeking. Front Neurosci. 2013;7:126 https://doi.org/10.3389/fnins.2013.00126.
Natsheh JY, Shiflett MW. Dopaminergic Modulation of Goal-Directed Behavior in a Rodent Model of Attention-Deficit/Hyperactivity Disorder. Front Integr Neurosci. 2018;12:45 https://doi.org/10.3389/fnint.2018.00045.
Ahmari SE, Spellman T, Douglass NL, Kheirbek MA, Simpson HB, Deisseroth K, et al. Repeated cortico-striatal stimulation generates persistent OCD-like behavior. Science. 2013;340:1234–9. https://doi.org/10.1126/science.1234733.
Burguiere E, Monteiro P, Feng G, Graybiel AM. Optogenetic stimulation of lateral orbitofronto-striatal pathway suppresses compulsive behaviors. Science. 2013;340:1243–6. https://doi.org/10.1126/science.1232380.
Dalton GL, Wang NY, Phillips AG, Floresco SB. Multifaceted Contributions by Different Regions of the Orbitofrontal and Medial Prefrontal Cortex to Probabilistic Reversal Learning. J Neurosci. 2016;36:1996–2006. https://doi.org/10.1523/JNEUROSCI.3366-15.2016.
Gremel CM, Costa RM. Orbitofrontal and striatal circuits dynamically encode the shift between goal-directed and habitual actions. Nat Commun. 2013;4:2264 https://doi.org/10.1038/ncomms3264.
Sesack SR, Hawrylak VA, Matus C, Guido MA, Levey AI. Dopamine axon varicosities in the prelimbic division of the rat prefrontal cortex exhibit sparse immunoreactivity for the dopamine transporter. J Neurosci. 1998;18:2697–708.
Ciliax BJ, Heilman C, Demchyshyn LL, Pristupa ZB, Ince E, Hersch SM, et al. The dopamine transporter: immunochemical characterization and localization in brain. J Neurosci. 1995;15:1714–23.
Lammel S, Hetzel A, Häckel O, Jones I, Liss B, Roeper J. Unique properties of mesoprefrontal neurons within a dual mesocorticolimbic dopamine system. Neuron. 2008;57:760–73. https://doi.org/10.1016/j.neuron.2008.01.022.
Moron JA, Brockington A, Wise RA, Rocha BA, Hope BT. Dopamine uptake through the norepinephrine transporter in brain regions with low levels of the dopamine transporter: evidence from knock-out mouse lines. J Neurosci. 2002;22:389–95.
Xu F, Gainetdinov RR, Wetsel WC, Jones SR, Bohn LM, Miller GW, et al. Mice lacking the norepinephrine transporter are supersensitive to psychostimulants. Nat Neurosci. 2000;3:465–71. https://doi.org/10.1038/74839.
Tanda G, Carboni E, Frau R, Di Chiara G. Increase of extracellular dopamine in the prefrontal cortex: a trait of drugs with antidepressant potential? Psychopharmacology. 1994;115:285–8. https://doi.org/10.1007/BF02244785.
Harris SS, Green SM, Kumar M, Urs NM. A role for cortical dopamine in the paradoxical calming effects of psychostimulants. Sci Rep. 2022;12:3129 https://doi.org/10.1038/s41598-022-07029-2.
Markou A, Kosten TR, Koob GF. Neurobiological similarities in depression and drug dependence: a self-medication hypothesis. Neuropsychopharmacology. 1998;18:135–74. https://doi.org/10.1016/S0893-133X(97)00113-9.
Amara SG, Kuhar MJ. Neurotransmitter transporters: recent progress. Annu Rev Neurosci. 1993;16:73–93. https://doi.org/10.1146/annurev.ne.16.030193.000445.
Shang CY, Lin HY, Gau SS. The norepinephrine transporter gene modulates intrinsic brain activity, visual memory, and visual attention in children with attention-deficit/hyperactivity disorder. Mol Psychiatry. 2021;26:4026–35. https://doi.org/10.1038/s41380-019-0545-7.
Aggarwal S, Mortensen OV. Overview of Monoamine Transporters. Curr Protoc Pharm. 2017;79:12 16 11–12 16 17. https://doi.org/10.1002/cpph.32.
Gainetdinov RR, Sotnikova TD, Caron MG. Monoamine transporter pharmacology and mutant mice. Trends Pharmacol Sci. 2002;23:367–73.
Torres GE, Gainetdinov RR, Caron MG. Plasma membrane monoamine transporters: structure, regulation and function. Nat Rev Neurosci. 2003;4:13–25. https://doi.org/10.1038/nrn1008.
Wang YM, Xu F, Gainetdinov RR, Caron MG. Genetic approaches to studying norepinephrine function: knockout of the mouse norepinephrine transporter gene. Biol Psychiatry. 1999;46:1124–30. https://doi.org/10.1016/s0006-3223(99)00245-0.
Belfer I, Phillips G, Taubman J, Hipp H, Lipsky RH, Enoch MA, et al. Haplotype architecture of the norepinephrine transporter gene SLC6A2 in four populations. J Hum Genet. 2004;49:232–45. https://doi.org/10.1007/s10038-004-0140-9.
Klimek V, Stockmeier C, Overholser J, Meltzer HY, Kalka S, Dilley G, et al. Reduced levels of norepinephrine transporters in the locus coeruleus in major depression. J Neurosci. 1997;17:8451–8. https://doi.org/10.1523/JNEUROSCI.17-21-08451.1997.
Pramod AB, Foster J, Carvelli L, Henry LK. SLC6 transporters: structure, function, regulation, disease association and therapeutics. Mol Asp Med. 2013;34:197–219. https://doi.org/10.1016/j.mam.2012.07.002.
Buttenschøn HN, Kristensen AS, Buch HN, Andersen JH, Bonde JP, Grynderup M, et al. The norepinephrine transporter gene is a candidate gene for panic disorder. J Neural Transm. 2011;118:969–76. https://doi.org/10.1007/s00702-011-0624-7.
Nemoda Z, Angyal N, Tarnok Z, Birkas E, Bognar E, Sasvari-Szekely M, et al. Differential Genetic Effect of the Norepinephrine Transporter Promoter Polymorphisms on Attention Problems in Clinical and Non-clinical Samples. Front Neurosci. 2018;12:1051 https://doi.org/10.3389/fnins.2018.01051.
Seu E, Jentsch JD. Effect of acute and repeated treatment with desipramine or methylphenidate on serial reversal learning in rats. Neuropharmacology. 2009;57:665–72. https://doi.org/10.1016/j.neuropharm.2009.08.007.
Altidor LK, Bruner MM, Deslauriers JF, Garman TS, Ramirez S, Dirr EW, et al. Acute vagus nerve stimulation enhances reversal learning in rats. Neurobiol Learn Mem. 2021;184:107498 https://doi.org/10.1016/j.nlm.2021.107498.
Seu E, Lang A, Rivera RJ, Jentsch JD. Inhibition of the norepinephrine transporter improves behavioral flexibility in rats and monkeys. Psychopharmacology. 2009;202:505–19. https://doi.org/10.1007/s00213-008-1250-4.
Lapiz MD, Bondi CO, Morilak DA. Chronic treatment with desipramine improves cognitive performance of rats in an attentional set-shifting test. Neuropsychopharmacology. 2007;32:1000–10. https://doi.org/10.1038/sj.npp.1301235.
Tait DS, Brown VJ, Farovik A, Theobald DE, Dalley JW, Robbins TW. Lesions of the dorsal noradrenergic bundle impair attentional set-shifting in the rat. Eur J Neurosci. 2007;25:3719–24. https://doi.org/10.1111/j.1460-9568.2007.05612.x.
McGaughy J, Ross RS, Eichenbaum H. Noradrenergic, but not cholinergic, deafferentation of prefrontal cortex impairs attentional set-shifting. Neuroscience. 2008;153:63–71. https://doi.org/10.1016/j.neuroscience.2008.01.064.
Cerpa JC, Piccin A, Dehove M, Lavigne M, Kremer EJ, Wolff M et al. Inhibition of noradrenergic signalling in rodent orbitofrontal cortex impairs the updating of goal-directed actions. Elife. 2023;12. https://doi.org/10.7554/eLife.81623.
Sadacca BF, Wikenheiser AM, Schoenbaum G. Toward a theoretical role for tonic norepinephrine in the orbitofrontal cortex in facilitating flexible learning. Neuroscience. 2017;345:124–9. https://doi.org/10.1016/j.neuroscience.2016.04.017.
Bissonette GB, Powell EM. Reversal learning and attentional set-shifting in mice. Neuropharmacology. 2012;62:1168–74. https://doi.org/10.1016/j.neuropharm.2011.03.011.
Izquierdo A, Wiedholz LM, Millstein RA, Yang RJ, Bussey TJ, Saksida LM, et al. Genetic and dopaminergic modulation of reversal learning in a touchscreen-based operant procedure for mice. Behav Brain Res. 2006;171:181–8. https://doi.org/10.1016/j.bbr.2006.03.029.
Floresco SB. Prefrontal dopamine and behavioral flexibility: shifting from an “inverted-U” toward a family of functions. Front Neurosci. 2013;7:62 https://doi.org/10.3389/fnins.2013.00062.
Bissonette GB, Roesch MR. Neurophysiology of rule switching in the corticostriatal circuit. Neuroscience. 2017;345:64–76. https://doi.org/10.1016/j.neuroscience.2016.01.062.
Clarke HF, Hill GJ, Robbins TW, Roberts AC. Dopamine, but not serotonin, regulates reversal learning in the marmoset caudate nucleus. J Neurosci. 2011;31:4290–7. https://doi.org/10.1523/JNEUROSCI.5066-10.2011.
Ventura R, Alcaro A, Cabib S, Conversi D, Mandolesi L, Puglisi-Allegra S. Dopamine in the medial prefrontal cortex controls genotype-dependent effects of amphetamine on mesoaccumbens dopamine release and locomotion. Neuropsychopharmacology. 2004;29:72–80. https://doi.org/10.1038/sj.npp.1300300.
Ventura R, Alcaro A, Mandolesi L, Puglisi-Allegra S. In vivo evidence that genetic background controls impulse-dependent dopamine release induced by amphetamine in the nucleus accumbens. J Neurochem. 2004;89:494–502. https://doi.org/10.1111/j.1471-4159.2004.02342.x.
Sokolowski JD, Salamone JD. Effects of dopamine depletions in the medial prefrontal cortex on DRL performance and motor activity in the rat. Brain Res. 1994;642:20–28. https://doi.org/10.1016/0006-8993(94)90901-6.
Pycock CJ, Kerwin RW, Carter CJ. Effect of lesion of cortical dopamine terminals on subcortical dopamine receptors in rats. Nature. 1980;286:74–76.
Bari A, Theobald DE, Caprioli D, Mar AC, Aidoo-Micah A, Dalley JW, et al. Serotonin modulates sensitivity to reward and negative feedback in a probabilistic reversal learning task in rats. Neuropsychopharmacology. 2010;35:1290–301. https://doi.org/10.1038/npp.2009.233.
Bruno CA, O'Brien C, Bryant S, Mejaes JI, Estrin DJ, Pizzano C, et al. pMAT: An open-source software suite for the analysis of fiber photometry data. Pharm Biochem Behav. 2021;201:173093 https://doi.org/10.1016/j.pbb.2020.173093.
Fischbach-Weiss S, Reese RM, Janak PH. Inhibiting Mesolimbic Dopamine Neurons Reduces the Initiation and Maintenance of Instrumental Responding. Neuroscience. 2018;372:306–15. https://doi.org/10.1016/j.neuroscience.2017.12.003.
Garman TS, Setlow B, Orsini CA. Effects of a high-fat diet on impulsive choice in rats. Physiol Behav. 2021;229:113260 https://doi.org/10.1016/j.physbeh.2020.113260.
Lustberg D, Iannitelli AF, Tillage RP, Pruitt M, Liles LC, Weinshenker D. Central norepinephrine transmission is required for stress-induced repetitive behavior in two rodent models of obsessive-compulsive disorder. Psychopharmacology. 2020;237:1973–87. https://doi.org/10.1007/s00213-020-05512-0.
Urs NM, Daigle TL, Caron MG. A Dopamine D1 Receptor-Dependent beta-Arrestin Signaling Complex Potentially Regulates Morphine-Induced Psychomotor Activation but not Reward in Mice. Neuropsychopharmacology. 2011;36:551–8. https://doi.org/10.1038/Npp.2010.186.
Dalton GL, Phillips AG, Floresco SB. Preferential involvement by nucleus accumbens shell in mediating probabilistic learning and reversal shifts. J Neurosci. 2014;34:4618–26. https://doi.org/10.1523/JNEUROSCI.5058-13.2014.
Bymaster FP, Katner JS, Nelson DL, Hemrick-Luecke SK, Threlkeld PG, Heiligenstein JH, et al. Atomoxetine increases extracellular levels of norepinephrine and dopamine in prefrontal cortex of rat: a potential mechanism for efficacy in attention deficit/hyperactivity disorder. Neuropsychopharmacology. 2002;27:699–711. https://doi.org/10.1016/S0893-133X(02)00346-9.
Gresch PJ, Sved AF, Zigmond MJ, Finlay JM. Local influence of endogenous norepinephrine on extracellular dopamine in rat medial prefrontal cortex. J Neurochem. 1995;65:111–6.
Berke JD. What does dopamine mean? Nat Neurosci. 2018;21:787–93. https://doi.org/10.1038/s41593-018-0152-y.
Yin HH, Ostlund SB, Knowlton BJ, Balleine BW. The role of the dorsomedial striatum in instrumental conditioning. Eur J Neurosci. 2005;22:513–23. https://doi.org/10.1111/j.1460-9568.2005.04218.x.
Grospe GM, Baker PM, Ragozzino ME. Cognitive Flexibility Deficits Following 6-OHDA Lesions of the Rat Dorsomedial Striatum. Neuroscience. 2018;374:80–90. https://doi.org/10.1016/j.neuroscience.2018.01.032.
Shiflett MW, Balleine BW. Molecular substrates of action control in cortico-striatal circuits. Prog Neurobiol. 2011;95:1–13. https://doi.org/10.1016/j.pneurobio.2011.05.007.
Giustino TF, Maren S. Noradrenergic Modulation of Fear Conditioning and Extinction. Front Behav Neurosci. 2018;12:43 https://doi.org/10.3389/fnbeh.2018.00043.
Aston-Jones G, Cohen JD. An integrative theory of locus coeruleus-norepinephrine function: adaptive gain and optimal performance. Annu Rev Neurosci. 2005;28:403–50. https://doi.org/10.1146/annurev.neuro.28.061604.135709.
Dziedzicka-Wasylewska M, Faron-Górecka A, Kuśmider M, Drozdowska E, Rogóz Z, Siwanowicz J, et al. Effect of antidepressant drugs in mice lacking the norepinephrine transporter. Neuropsychopharmacology. 2006;31:2424–32. https://doi.org/10.1038/sj.npp.1301064.
Moret C, Briley M. The importance of norepinephrine in depression. Neuropsychiatr Dis Treat. 2011;7:9–13,. https://doi.org/10.2147/NDT.S19619.
Dorninger F, Zeitler G, Berger J. Nestlet Shredding and Nest Building Tests to Assess Features of Psychiatric Disorders in Mice. Bio Protoc. 2020;10. https://doi.org/10.21769/BioProtoc.3863.
Spear DJ, Katz JL. Cocaine and food as reinforcers: effects of reinforcer magnitude and response requirement under second-order fixed-ratio and progressive-ratio schedules. J Exp Anal Behav. 1991;56:261–75. https://doi.org/10.1901/jeab.1991.56-261.
Balleine BW, O’Doherty JP. Human and rodent homologies in action control: corticostriatal determinants of goal-directed and habitual action. Neuropsychopharmacology. 2010;35:48–69. https://doi.org/10.1038/npp.2009.131.
Arnsten AF, Pliszka SR. Catecholamine influences on prefrontal cortical function: relevance to treatment of attention deficit/hyperactivity disorder and related disorders. Pharm Biochem Behav. 2011;99:211–6. https://doi.org/10.1016/j.pbb.2011.01.020.
Arnsten AF, Dudley AG. Methylphenidate improves prefrontal cortical cognitive function through alpha2 adrenoceptor and dopamine D1 receptor actions: Relevance to therapeutic effects in Attention Deficit Hyperactivity Disorder. Behav Brain Funct. 2005;1:2 https://doi.org/10.1186/1744-9081-1-2.
Goldman-Rakic PS. The cortical dopamine system: role in memory and cognition. Adv Pharm. 1998;42:707–11. https://doi.org/10.1016/s1054-3589(08)60846-7.
Brennan BP, Tkachenko O, Schwab ZJ, Juelich RJ, Ryan EM, Athey AJ, et al. An Examination of Rostral Anterior Cingulate Cortex Function and Neurochemistry in Obsessive-Compulsive Disorder. Neuropsychopharmacology. 2015;40:1866–76. https://doi.org/10.1038/npp.2015.36.
Riffkin J, Yücel M, Maruff P, Wood SJ, Soulsby B, Olver J, et al. A manual and automated MRI study of anterior cingulate and orbito-frontal cortices, and caudate nucleus in obsessive-compulsive disorder: comparison with healthy controls and patients with schizophrenia. Psychiatry Res. 2005;138:99–113. https://doi.org/10.1016/j.pscychresns.2004.11.007.
Tsai HC, Zhang F, Adamantidis A, Stuber GD, Bonci A, de Lecea L, et al. Phasic firing in dopaminergic neurons is sufficient for behavioral conditioning. Science. 2009;324:1080–4. https://doi.org/10.1126/science.1168878.
Starkweather CK, Uchida N. Dopamine signals as temporal difference errors: recent advances. Curr Opin Neurobiol. 2021;67:95–105. https://doi.org/10.1016/j.conb.2020.08.014.
Wang Y, Toyoshima O, Kunimatsu J, Yamada H, Matsumoto M. Tonic firing mode of midbrain dopamine neurons continuously tracks reward values changing moment-by-moment. Elife. 2021;10. https://doi.org/10.7554/eLife.63166.
Solich J, Faron-Gorecka A, Kusmider M, Palach P, Gaska M, Dziedzicka-Wasylewska M. Norepinephrine transporter (NET) knock-out upregulates dopamine and serotonin transporters in the mouse brain. Neurochem Int. 2011;59:185–91. https://doi.org/10.1016/j.neuint.2011.04.012.
Richardson BD, Saha K, Krout D, Cabrera E, Felts B, Henry LK, et al. Membrane potential shapes regulation of dopamine transporter trafficking at the plasma membrane. Nat Commun. 2016;7:10423 https://doi.org/10.1038/ncomms10423.
Cagniard B, Balsam PD, Brunner D, Zhuang X. Mice with chronically elevated dopamine exhibit enhanced motivation, but not learning, for a food reward. Neuropsychopharmacology. 2006;31:1362–70. https://doi.org/10.1038/sj.npp.1300966.
Sommer S, Danysz W, Russ H, Valastro B, Flik G, Hauber W. The dopamine reuptake inhibitor MRZ-9547 increases progressive ratio responding in rats. Int J Neuropsychopharmacol. 2014;17:2045–56. https://doi.org/10.1017/S1461145714000996.
Seiler JL, Cosme CV, Sherathiya VN, Schaid MD, Bianco JM, Bridgemohan AS, et al. Dopamine signaling in the dorsomedial striatum promotes compulsive behavior. Curr Biol. 2022;32:1175–88.e1175. https://doi.org/10.1016/j.cub.2022.01.055.
Acknowledgements
We would like to thank Dr. Marc Caron for providing us with monoamine transporter knockout mice. We would also like to thank Dr. Stan Floresco (U of British Columbia) for the Med-PC code for the reversal learning task and related advice.
Funding
This work was supported by a NIMH R21 (MH127377) and R01 (MH130778) grant (NMU) and NARSAD/BBRF Young Investigator grant (NMU).
Author information
Authors and Affiliations
Contributions
Jena Delaney –performed experiments, analyzed data. Sanya Nathani - performed experiments. Victor Tan - performed experiments. Alex Orr - performed experiments. Carson Chavez - performed experiments. Joon Paek – Analyzed data. Mojdeh Faraji – analyzed data. Barry Setlow - conceptualized experiments, wrote manuscript. Nikhil Urs - conceptualized experiments, performed experiments, analyzed data, wrote manuscript
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Delaney, J., Nathani, S., Tan, V. et al. Enhanced cognitive flexibility and phasic striatal dopamine dynamics in a mouse model of low striatal tonic dopamine. Neuropsychopharmacol. (2024). https://doi.org/10.1038/s41386-024-01868-5
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41386-024-01868-5