Abstract
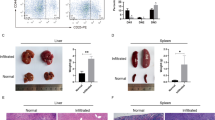
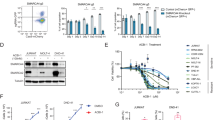
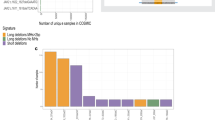
Adult T-cell leukemia/lymphoma (ATL) is a genetically complex hematological malignancy derived from mature T cells. Using an integrative approach, we previously identified genes recurrently associated with super-enhancers in ATL. One of those genes was TP73, a TP53 family gene; however, the roles and function of TP73 and its super-enhancer in ATL pathogenesis are poorly understood. Our study demonstrates that TP73 is highly activated under the control of a super-enhancer in ATL cells but not in normal T cells or other hematological malignancies examined. Full-length TP73 is required for ATL cell maintenance in vitro and in vivo via the regulation of cell proliferation and DNA damage response pathways. Notably, recurrent deletions of TP73 exons 2–3 were observed in a fraction of primary ATL cases that harbored the super-enhancer, while induction of this deletion in cell lines further increased proliferation and mutational burden. Our study suggests that formation of the TP73 intragenic super-enhancer and genetic deletion are likely sequentially acquired in relation to intracellular state of ATL cells, which leads to functional alteration of TP73 that confers additional clonal advantage.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Waldmann TA. Daclizumab (anti-Tac, Zenapax) in the treatment of leukemia/lymphoma. Oncogene. 2007;26:3699–703.
Matsuoka M, Jeang K-T. Human T-cell leukaemia virus type 1 (HTLV-1) infectivity and cellular transformation. Nat Rev Cancer. 2007;7:270–80.
Ishitsuka K, Tamura K. Human T-cell leukaemia virus type I and adult T-cell leukaemia-lymphoma. Lancet Oncol. 2014;15:517–26.
Bangham CR, Ratner L. How does HTLV-1 cause adult T-cell leukaemia/lymphoma (ATL)? Curr Opin Virol. 2015;14:93–100.
Watanabe T. Adult T-cell leukemia: molecular basis for clonal expansion and transformation of HTLV-1-infected T cells. Blood. 2017;129:1071–81.
Wong RWJ, Ngoc PCT, Leong WZ, Yam AWY, Zhang T, Asamitsu K, et al. Enhancer profiling identifies critical cancer genes and characterizes cell identity in adult T-cell leukemia. Blood, J Am Soc Hematol. 2017;130:2326–38.
Wong RWJ, Tan TK, Amanda S, Ngoc PCT, Leong WZ, Tan SH, et al. Feed-forward regulatory loop driven by IRF4 and NF-κB in adult T-cell leukemia/lymphoma. Blood. 2020;135:934–47.
Kataoka K, Nagata Y, Kitanaka A, Shiraishi Y, Shimamura T, Yasunaga J-I, et al. Integrated molecular analysis of adult T cell leukemia/lymphoma. Nat Genet. 2015;47:1304–15.
Kogure Y, Kameda T, Koya J, Yoshimitsu M, Nosaka K, Yasunaga J-I, et al. Whole-genome landscape of adult T-cell leukemia/lymphoma. Blood. 2022;139:967–82.
Yang A, McKeon F. P63 and P73: P53 mimics, menaces and more. Nat Rev Mol cell Biol. 2000;1:199–207.
Melino G, De Laurenzi V, Vousden KH. p73: Friend or foe in tumorigenesis. Nat Rev Cancer. 2002;2:605–15.
Gong J, Costanzo A, Yang H-Q, Melino G, Kaelin WG, Levrero M, et al. The tyrosine kinase c-Abl regulates p73 in apoptotic response to cisplatin-induced DNA damage. Nature. 1999;399:806–9.
Ng S-W, Yiu GK, Liu Y, Huang L-W, Palnati M, Jun SH, et al. Analysis of p73 in human borderline and invasive ovarian tumor. Oncogene. 2000;19:1885–90.
O’nions J, Brooks L, Sullivan A, Bell A, Dunne B, Rozycka M, et al. p73 is over-expressed in vulval cancer principally as the δ 2 isoform. Br J Cancer. 2001;85:1551–6.
Zaika AI, Kovalev S, Marchenko ND, Moll UM. Overexpression of the wild type p73 gene in breast cancer tissues and cell lines. Cancer Res. 1999;59:3257–63.
Grob T, Novak U, Maisse C, Barcaroli D, Lüthi A, Pirnia F, et al. Human ΔNp73 regulates a dominant negative feedback loop for TAp73 and p53. Cell Death Differ. 2001;8:1213–23.
Casciano I, Mazzocco K, Boni L, Pagnan G, Banelli B, Allemanni G, et al. Expression of ΔNp73 is a molecular marker for adverse outcome in neuroblastoma patients. Cell Death Differ. 2002;9:246–51.
Concin N, Becker K, Slade N, Erster S, Mueller-Holzner E, Ulmer H, et al. Transdominant ΔTAp73 isoforms are frequently up-regulated in ovarian cancer. Evidence for their role as epigenetic p53 inhibitors in vivo. Cancer Res. 2004;64:2449–60.
Stiewe T, Tuve S, Peter M, Tannapfel A, Elmaagacli AH, Pützer BM. Quantitative TP73 transcript analysis in hepatocellular carcinomas. Clin Cancer Res. 2004;10:626–33.
Kwiatkowski N, Zhang T, Rahl PB, Abraham BJ, Reddy J, Ficarro SB, et al. Targeting transcription regulation in cancer with a covalent CDK7 inhibitor. Nature. 2014;511:616–20.
Flores ER, Tsai KY, Crowley D, Sengupta S, Yang A, McKeon F, et al. p63 and p73 are required for p53-dependent apoptosis in response to DNA damage. Nature. 2002;416:560–4.
Zaika E, Wei J, Yin D, Andl C, Moll U, El‐Rifai W, et al. p73 protein regulates DNA damage repair. FASEB J. 2011;25:4406–14.
Agami R, Blandino G, Oren M, Shaul Y. Interaction of c-Abl and p73α and their collaboration to induce apoptosis. Nature. 1999;399:809–13.
Nakagawa T, Takahashi M, Ozaki T, Watanabe K-I, Todo S, Mizuguchi H, et al. Autoinhibitory regulation of p73 by ΔNp73 to modulate cell survival and death through a p73-specific target element within the Δ Np73 promoter. Mol Cell Biol. 2002;22:2575–85.
Kinner A, Wu W, Staudt C, Iliakis G. γ-H2AX in recognition and signaling of DNA double-strand breaks in the context of chromatin. Nucleic Acids Res. 2008;36:5678–94.
Bonner WM, Redon CE, Dickey JS, Nakamura AJ, Sedelnikova OA, Solier S, et al. γH2AX and cancer. Nat Rev Cancer. 2008;8:957–67.
Grassmann R, Aboud M, Jeang K-T. Molecular mechanisms of cellular transformation by HTLV-1 tax. Oncogene. 2005;24:5976–85.
Janssens S, Tinel A, Lippens S, Tschopp J. PIDD mediates NF-κB activation in response to DNA damage. Cell. 2005;123:1079–92.
Nakayama T, Hieshima K, Arao T, Jin Z, Nagakubo D, Shirakawa A, et al. Aberrant expression of Fra-2 promotes CCR4 expression and cell proliferation in adult T-cell leukemia. Oncogene. 2008;27:3221–32.
Nakagawa M, Shaffer AL, Ceribelli M, Zhang M, Wright G, Huang DW, et al. Targeting the HTLV-I-regulated BATF3/IRF4 transcriptional network in adult T cell leukemia/lymphoma. Cancer Cell. 2018;34:286–297.e210.
Yang A, Walker N, Bronson R, Kaghad M, Oosterwegel M, Bonnin J, et al. p73-deficient mice have neurological, pheromonal and inflammatory defects but lack spontaneous tumours. Nature. 2000;404:99–103.
Ikawa S, Nakagawara A, Ikawa Y. p53 family genes: structural comparison, expression and mutation. Cell Death Differ. 1999;6:1154–61.
Yang A, Kaghad M, Caput D, McKeon F. On the shoulders of giants: p63, p73 and the rise of p53. TRENDS Genet. 2002;18:90–95.
George J, Lim JS, Jang SJ, Cun Y, Ozretić L, Kong G, et al. Comprehensive genomic profiles of small cell lung cancer. Nature. 2015;524:47–53.
Liu Y, Easton J, Shao Y, Maciaszek J, Wang Z, Wilkinson MR, et al. The genomic landscape of pediatric and young adult T-lineage acute lymphoblastic leukemia. Nat Genet. 2017;49:1211–8.
Kamada N, Sakurai M, Miyamoto K, Sanada I, Sadamori N, Fukuhara S, et al. Chromosome abnormalities in adult T-cell leukemia/lymphoma: a karyotype review committee report. Cancer Res. 1992;52:1481–93.
Tsukasaki K, Krebs J, Nagai K, Tomonaga M, Koeffler HP, Bartram CR, et al. Comparative genomic hybridization analysis in adult T-cell leukemia/lymphoma: correlation with clinical course. Blood. 2001;97:3875–81.
Whyte WA, Orlando DA, Hnisz D, Abraham BJ, Lin CY, Kagey MH, et al. Master transcription factors and mediator establish super-enhancers at key cell identity genes. Cell. 2013;153:307–19.
Hnisz D, Abraham BJ, Lee TI, Lau A, Saint-André V, Sigova AA, et al. Super-enhancers in the control of cell identity and disease. Cell. 2013;155:934–47.
Miyoshi H, Shimizu K, Kozu T, Maseki N, Kaneko Y, Ohki M. t (8; 21) breakpoints on chromosome 21 in acute myeloid leukemia are clustered within a limited region of a single gene, AML1. Proc Natl Acad Sci USA. 1991;88:10431–4.
Ng CE, Yokomizo T, Yamashita N, Cirovic B, Jin H, Wen Z, et al. A Runx1 intronic enhancer marks hemogenic endothelial cells and hematopoietic stem cells. Stem Cells. 2010;28:1869–81.
Chen MJ, Yokomizo T, Zeigler BM, Dzierzak E, Speck NA. Runx1 is required for the endothelial to haematopoietic cell transition but not thereafter. Nature. 2009;457:887–91.
Hiebert SW, Sun W, Davis JN, Golub T, Shurtleff S, Buijs A, et al. The t (12; 21) translocation converts AML-1B from an activator to a repressor of transcription. Mol Cell Biol. 1996;16:1349–55.
Acknowledgements
We thank Nature Publishing Group Language Editing for editing the manuscript. We thank the members of the Sanda laboratory for discussions and critical reviews. We acknowledge support from the Yong Loo Lin School of Medicine BSL-3 Core Facility, National University of Singapore, National University Health System, and from the Singapore Ministry of Health, National Medical Research Council, Center Grant ‘MINE’, Research Core #4 (NMRC/CG/013/2013). This research is supported by the National Medical Research Council of the Singapore Ministry of Health (NMRC/CIRG/1491/2018 and OFLCG18May-0028: TS); the National Research Foundation Singapore and the Singapore Ministry of Education under its Research Centres of Excellence initiative (TS); Japan Society for the Promotion of Science, KAKENHI (18K19960: TS and SI); and the Japan Agency for Medical Research and Development (no.19ae0101074s0401: RU). Illustrations were created with Biorender.
Author information
Authors and Affiliations
Contributions
JZLO and RWJW performed the experiments; RY and TKT conducted the bioinformatic analyses; RU, TI, and SI provided the primary samples; and JZLO and TS wrote the manuscript.
Corresponding author
Ethics declarations
Competing interests
RU reports receiving research funding from Kyowa Kirin, Chugai and Ono Pharmaceutical.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Ong, J.Z.L., Yokomori, R., Wong, R.W.J. et al. Requirement for TP73 and genetic alterations originating from its intragenic super-enhancer in adult T-cell leukemia. Leukemia 36, 2293–2305 (2022). https://doi.org/10.1038/s41375-022-01655-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41375-022-01655-5
This article is cited by
-
Enhancers in T Cell development and malignant lesions
Cell Death Discovery (2024)
-
Clinical landscape of TP73 structural variants in ATL patients
Leukemia (2023)