Abstract
Objective
To evaluate outcomes in opioid exposed neonates (OENs) assessed by the Eat, Sleep, Console (ESC) tool compared to the Finnegan Neonatal Abstinence Scoring System (FNASS).
Methods
Retrospective analysis of a statewide database of OENs from 2017 to 2020 with birthing hospitals classified based on the assessment tool used. Four main outcomes were examined using multivariable and Poisson logistic regression models.
Results
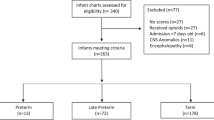
Of 2375 OENs, 42.1% received pharmacotherapy (PT) with a consistent decrease in PT, length of treatment (LOT), and length of stay (LOS) over the study period. There was no change in use of mother’s own milk (MoM). While outcomes were significantly associated with several specific variables, there were no differences in outcomes between assessment methods.
Conclusion
While there was a significant decrease over time in PT, LOT, and LOS, improvements were independent of the assessment tool used and likely related to the increased use of non-pharmacologic care.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout

Similar content being viewed by others
Data availability
The dataset used for this study is not publicly available as it contains some identifiable information, but is available from the corresponding author on reasonable request.
References
https://nida.nih.gov/research-topics/trends-statistics/overdose-death-rates. Accessed 10 Jun 2023.
Keyes KM, Rutherford C, Hamilton A, Barocas JA, Gelberg KH, Mueller PP, et al. What is the prevalence of and trend in opioid use disorder in the United States from 2010 to 2019? Using multiplier approaches to estimate prevalence for an unknown population size. Drug Alcohol Depend Rep. 2022;3:100052.
Hirai AH, Ko JY, Owens PL, Stocks C, Patrick SW. Neonatal abstinence syndrome and maternal opioid-related diagnoses in the US, 2010-2017. JAMA. 2021;325:146–55.
Patrick SW, Barfield WD, Poindexter B, Committee on Fetus and Newborn, Committee on Substance Use and Prevention. Neonatal Opioid Withdrawal Syndrome. Pediatrics. 2020;146:e2020029074.
Finnegan LP, Connaughton JF Jr, Kron RE, Emich JP. Neonatal abstinence syndrome: assessment and management. Addict Dis. 1975;2:141–58.
Devlin LA, Breeze JL, Terrin N, Pomar EG, Bada H, Finnegan LP, et al. Association of a simplified Finnegan neonatal abstinence scoring Method with the need for pharmacologic treatment for neonatal abstinence syndrome. JAMA Netw Open. 2020;3:e202275.
Grossman MR, Lipshaw MJ, Osborn RR, Berkwitt AK. A novel approach to assessing infants with neonatal abstinence syndrome. Hosp Pediatr. 2018;8:1–6.
Wachman EM, Houghton M, Melvin P, Isley BC, Murzycki J, Singh R, et al. A quality improvement initiative to implement the eat, sleep, console neonatal opioid withdrawal syndrome care method in Massachusetts’ PNQIN collaborative. J Perinatol. 2020;40:1560–9.
Young LW, Ounpraseuth ST, Merhar SL, Hu Z, Simon AE, Bremer AA et al. ACT NOW Collaborative. Eat, sleep, console approach or usual care for neonatal opioid withdrawal. N Engl J Med. 2023;388:2326–37.
Singh R, Houghton M, Melvin P, Wachman EM, Diop H, Iverson R, et al. Predictors of pharmacologic therapy for neonatal opioid withdrawal syndrome: a retrospective analysis of a statewide database. J Perinatol. 2021;41:1381–8.
Singh R, Rothstein R, Ricci K, Vistainer P, Shenberger J, Attwood E, et al. Partnering with parents to improve outcomes for substance exposed newborns—a pilot program. J Perinatol. 2020;40:1041–9.
Kilpatrick SJ, Papile LA, Macones GA. Organization of perinatal healthcare. In: Guidelines for Perinatal Care. 8th Edition:1-36. American Academy of Pediatrics and American College of Obstetrics and Gynecology; 2017.
https://matracking.ehs.state.ma.us/eohhs_regions/eohhs_regions.html. Accessed 10 Aug 2023.
Pregnancy to Early Life Longitudinal Data System (PELL). NAS Dashboard New Data v6a - 2023-02-21 - MHT (mass.gov). Accessed 10 Aug 2023 https://www.mass.gov/info-details/neonatal-abstinence-syndrome-nas-data.
Jilani SM, Jordan CJ, Jansson LM, Davis JM. Definitions of neonatal abstinence syndrome in clinical studies of mothers and infants: an expert literature review. J Perinatol. 2021;41:1364–71.
Jilani SM, Jones HE, Davis JM. Implementation of a standardized clinical definition of opioid withdrawal in the neonate: challenges and opportunities. JAMA. 2022;327:1643–4.
Jones HE, Kaltenbach K, Heil SH, Stein S. M., Coyle M. G., Arria A. M. et al. Neonatal abstinence syndrome after methadone or buprenorphine exposure. N. Engl J Med. 2010;363:2320–31.
Singh R, Davis JM. Escaping the Finnegan - Is it time? Semin Fetal Neonatal Med. 2021;26:101218.
Czynski AJ, Davis JM, Dansereau LM, Engelhardt B, Marro P, Bogen DL, et al. Neurodevelopmental outcomes of neonates randomized to morphine or methadone for treatment of neonatal abstinence syndrome. J Pediatr. 2020;219:146–51.e1.
Jones H, Baer G, Davis JM. Neonatal Abstinence Syndrome. In: Boardman JP, Groves A, Ramasethu J editors. Avery and MacDonald’s Neonatology, Pathophysiology and Management of the Newborn, 8th edition, J.B. Lippincott Co., Philadelphia, PA, 2021
Pahl A, Young L, Buss-Frank ME, Marcellus L, Soll R. Non-pharmacological care for opioid withdrawal in newborns. Cochrane Database of Systematic Reviews 2020, Art. No. : CD013217. Accessed Oct 31 2023.
Morrison TM, MacMillan KDL, Melvin P, Singh R, Murzycki J, VanVleet MW, et al. Neonatal opioid withdrawal syndrome: a comparison of as-needed pharmacotherapy. Hosp Pediatr. 2022;12:530–8.
Acknowledgements
The authors gratefully acknowledge the contributions of their subjects, and their subjects’ families, as well as those of their colleagues. PNQIN Perinatal Opioid Project Collaborating Hospitals: Baystate Medical Center, Springfield, MA, USA; Baystate Franklin Medical Center, Greenfield, MA, USA; Berkshire Medical Center, Pittsfield, MA, USA; Beth Israel Deaconess Medical Center, Boston, MA, USA; Beverly Hospital, Beverly, MA, USA; Boston Medical Center, Boston, MA, USA; Brockton Hospital, Brockton, MA, USA; Cape Cod Hospital, Hyannis, MA, USA; Cooley-Dickenson Hospital, Northampton, MA, USA; Lowell General Hospital, Lowell, MA, USA; Massachusetts General Hospital, Boston, MA, USA; MelroseWakefield Hospital, Melrose MA, USA; Mercy Medical Center, Springfield, MA, USA; Southcoast Health, Wareham, MA, USA; South Shore Hospital, South Weymouth, MA, USA; Tufts Medical Center, Boston, MA, USA; UMass Memorial Medical Center, Worcester, MA, USA.
Funding
This work has been supported by programmatic funding from Health Policy Commission of Massachusetts.
Author information
Authors and Affiliations
Consortia
Contributions
RS and JD drafted the original manuscript and conceived and designed the study, contributed to data acquisition and analysis, and drafted the manuscript. PM contributed to data analysis; critically reviewed and revised the manuscript for intellectual content. EW, RR, DS, JM, MVV and MG contributed to data acquisition and analysis; critically reviewed and revised the manuscript for important intellectual content. All authors approved the final manuscript as submitted and agree to be accountable for all aspects of the work.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Singh, R., Melvin, P., Wachman, E.M. et al. Short term outcomes of neonatal opioid withdrawal syndrome: a comparison of two approaches. J Perinatol 44, 1137–1145 (2024). https://doi.org/10.1038/s41372-024-01953-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41372-024-01953-z