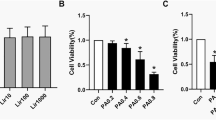
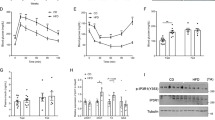
Abstract
Objective
Ectopic lipid deposition is closely associated with type 2 diabetes (T2D). Accumulating evidence shows that GLP-1 receptor agonists (GLP-1 RAs) improve obesity and liver steatosis. However, it remains unknown whether and how they ameliorate lipid deposition in skeletal muscle. This study aimed to investigate the effect of exenatide (a GLP-1 RA) on intramyocellular lipid deposition in the skeletal muscle of T2D models and its dependence on weight loss.
Methods
Ob/ob mice and diet-induced obese (DIO) mice were treated with exenatide (24 nmol/kg), leptin (1 mg/kg), or saline control intraperitoneally once daily for 4 weeks. Phenotypic evaluations were performed during and after the intervention. PA-induced myoblast C2C12 cells were used as an in vitro model. The expression of key enzymes involved in lipid metabolism was assessed in the skeletal muscle of ob/ob mice and DIO mice.
Results
In ob/ob mice, 4-week exenatide treatment did not improve the body weight and fat mass, but modestly ameliorated intramyocellular lipid deposition and lipid profiles. In DIO mice, it remarkably alleviated the body weight, lipid profiles, and intramyocellular lipid deposition. In the skeletal muscle of these two models, exenatide treatment activated the AMP-activated protein kinase (AMPK) signaling pathway, stimulated lipid oxidation enzymes, and upregulated the insulin signaling pathway. In vitro, exendin-4 activated the AMPK signaling pathway and stimulated lipid metabolism to improve lipid accumulation in palmitate-induced myoblast C2C12 cells.
Conclusions
Exenatide ameliorated intramyocellular lipid deposition without body weight reduction in ob/ob mice, but alleviated body weight and intramyocellular lipid deposition in DIO mice. The underlying mechanism included the activation of AMPK signaling pathway and improvement in insulin sensitivity, independent of weight loss in ob/ob mice.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Drucker DJ, Nauck MA. The incretin system: glucagon-like peptide-1 receptor agonists and dipeptidyl peptidase-4 inhibitors in type 2 diabetes. Lancet. 2006;368:1696–705.
Gallwitz B, Guzman J, Dotta F, Guerci B, Simo R, Basson BR, et al. Exenatide twice daily versus glimepiride for prevention of glycaemic deterioration in patients with type 2 diabetes with metformin failure (EUREXA): an open-label, randomised controlled trial. Lancet. 2012;379:2270–8.
Campbell JE, Drucker DJ. Pharmacology, physiology, and mechanisms of incretin hormone action. Cell Metab. 2013;17:819–37.
Bunck MC, Corner A, Eliasson B, Heine RJ, Shaginian RM, Wu Y, et al. One-year treatment with exenatide vs. insulin glargine: effects on postprandial glycemia, lipid profiles, and oxidative stress. Atherosclerosis. 2010;212:223–9.
Schwartz EA, Koska J, Mullin MP, Syoufi I, Schwenke DC, Reaven PD. Exenatide suppresses postprandial elevations in lipids and lipoproteins in individuals with impaired glucose tolerance and recent onset type 2 diabetes mellitus. Atherosclerosis. 2010;212:217–22.
Xu W, Y. Bi Z, Sun J, Li L, Guo T, Yang T. et al. Comparison of the effects on glycaemic control and beta-cell function in newly diagnosed type 2 diabetes patients of treatment with exenatide, insulin or pioglitazone: a multicentre randomized parallel-group trial (the CONFIDENCE study). J Intern Med. 2015;277:137–50.
Iepsen EW, Zhang J, Thomsen HS, Hansen EL, Hollensted M, Madsbad S, et al., Patients with obesity caused by melanocortin-4 receptor mutations can be treated with a glucagon-like peptide-1 receptor agonist. Cell Metab. 2018:28:23–32 e3.
Ding X, Saxena NK, Lin S, Gupta NA, Anania FA. Exendin-4, a glucagon-like protein-1 (GLP-1) receptor agonist, reverses hepatic steatosis in ob/ob mice. Hepatology. 2006;43:173–81.
Xu F, Li Z, Zheng X, Liu H, Liang H, Xu H, et al. SIRT1 mediates the effect of GLP-1 receptor agonist exenatide on ameliorating hepatic steatosis. Diabetes. 2014;63:3637–46.
Armstrong MJ, Hull D, Guo K, Barton D, Hazlehurst JM, Gathercole LL, et al. Glucagon-like peptide 1 decreases lipotoxicity in non-alcoholic steatohepatitis. J Hepatol. 2016;64:399–408.
Armstrong MJ, Gaunt P, Aithal GP, Barton D, Hull D, Parker R, et al. Liraglutide safety and efficacy in patients with non-alcoholic steatohepatitis (LEAN): a multicentre, double-blind, randomised, placebo-controlled phase 2 study. Lancet. 2016;387:679–90.
Coen PM, Goodpaster BH. Role of intramyocelluar lipids in human health. Trends Endocrinol Metab. 2012;23:391–8.
Pan DA, Lillioja S, Milner MR, Kriketos AD, Baur LA, Bogardus C, et al. Skeletal muscle membrane lipid composition is related to adiposity and insulin action. J Clin Investig. 1995;96:2802–8.
Jacob S, Machann J, Rett K, Brechtel K, Volk A, Renn W, et al. Association of increased intramyocellular lipid content with insulin resistance in lean nondiabetic offspring of type 2 diabetic subjects. Diabetes. 1999;48:1113–9.
Perseghin G, Scifo P, De Cobelli F, Pagliato E, Battezzati A, Arcelloni C, et al. Intramyocellular triglyceride content is a determinant of in vivo insulin resistance in humans: a 1H-13C nuclear magnetic resonance spectroscopy assessment in offspring of type 2 diabetic parents. Diabetes. 1999;48:1600–6.
Goodpaster BH, Theriault R, Watkins SC, Kelley DE. Intramuscular lipid content is increased in obesity and decreased by weight loss. Metabolism. 2000;49:467–72.
Bachmann OP, Dahl DB, Brechtel K, Machann J, Haap M, Maier T, et al. Effects of intravenous and dietary lipid challenge on intramyocellular lipid content and the relation with insulin sensitivity in humans. Diabetes. 2001;50:2579–84.
Toledo FG, Menshikova EV, Azuma K, Radikova Z, Kelley CA, Ritov VB, et al. Mitochondrial capacity in skeletal muscle is not stimulated by weight loss despite increases in insulin action and decreases in intramyocellular lipid content. Diabetes. 2008;57:987–94.
Muoio DM, Dohm GL, Fiedorek FT Jr., Tapscott EB, Coleman RA. Leptin directly alters lipid partitioning in skeletal muscle. Diabetes. 1997;46:1360–3.
Minokoshi Y, Kim YB, Peroni OD, Fryer LG, Muller C, Carling D, et al. Leptin stimulates fatty-acid oxidation by activating AMP-activated protein kinase. Nature. 2002;415:339–43.
Minokoshi Y, Kahn BB. Role of AMP-activated protein kinase in leptin-induced fatty acid oxidation in muscle. Biochem Soc Trans. 2003;31:196–201.
Akasaka Y, Tsunoda M, Ide T, Murakami K. Chronic leptin treatment stimulates lipid oxidation in immortalized and primary mouse skeletal muscle cells. Biochim Biophys Acta. 2009;1791:103–9.
Akasaka Y, Tsunoda M, Ogata T, Ide T, Murakami K. Direct evidence for leptin-induced lipid oxidation independent of long-form leptin receptor. Biochim Biophys Acta. 2010;1801:1115–22.
Wolsk E, Mygind H, Grondahl TS, Pedersen BK, van Hall G. The role of leptin in human lipid and glucose metabolism: the effects of acute recombinant human leptin infusion in young healthy males. Am J Clin Nutr. 2011;94:1533–44.
Choung JS, Lee YS, Jun HS. Exendin-4 increases oxygen consumption and thermogenic gene expression in muscle cells. J Mol Endocrinol. 2017;58:79–90.
A Rodriguez SB, Mendez-Gimenez L, Ramirez B, Sainz N, Catalan V, Gomez-Ambrosi J, Fruhbeck G. Leptin administration activates irisin-induced myogenesis via nitric oxide-dependent mechanisms, but reduces its effect on subcutaneous fat browning in mice. Int J Obesity. 2015;39:397–407.
Xu F, Burk D, Gao Z, Yin J, Zhang X, Weng J, et al. Angiogenic deficiency and adipose tissue dysfunction are associated with macrophage malfunction in SIRT1–/– mice. Endocrinology. 2012;153:1706–16.
Bi Y, Wu W, Shi J, Liang H, Yin W, Chen Y, et al. Role for sterol regulatory element binding protein-1c activation in mediating skeletal muscle insulin resistance via repression of rat insulin receptor substrate-1 transcription. Diabetologia. 2014;57:592–602.
Best JH, Lavillotti K, DeYoung MB, Garrison LP. The effects of exenatide bid on metabolic control, medication use and hospitalization in patients with type 2 diabetes mellitus in clinical practice: a systematic review. Diabetes Obes Metab. 2012;14:387–98.
Kim S, Jung J, Kim H, Heo RW, Yi C-o, Lee JE, et al. Exendin-4 improves nonalcoholic fatty liver disease by regulating glucose transporter 4 expression in ob/ob mice. Korean J Physiol Pharmacol. 2014;18:333.
Zheng X, Xu F, Liang H, Cao H, Cai M, Xu W, et al. SIRT1/HSF1/HSP pathway is essential for exenatide-alleviated, lipid-induced hepatic endoplasmic reticulum stress. Hepatology. 2017;66:809–24.
Cusi K. The role of adipose tissue and lipotoxicity in the pathogenesis of type 2 diabetes. Curr Diab Rep. 2010;10:306–15.
Lara-Castro C, Garvey WT. Intracellular lipid accumulation in liver and muscle and the insulin resistance syndrome. Endocrinol Metab Clin N Am. 2008;37:841–56.
Wang C, Liu F, Yuan Y, Wu J, Wang H, Zhang L, et al. Metformin suppresses lipid accumulation in skeletal muscle by promoting fatty acid oxidation. Clin Lab. 2014;60:887–96.
Williams DL, Baskin DG, Schwartz MW. Leptin regulation of the anorexic response to glucagon-like peptide-1 receptor stimulation. Diabetes. 2006;55:3387–93.
Xu F, Lin B, Zheng X, Chen Z, Cao H, Xu H, et al. GLP-1 receptor agonist promotes brown remodelling in mouse white adipose tissue through SIRT1. Diabetologia. 2016;59:1059–69.
Lin S, Thomas TC, Storlien LH, Huang XF. Development of high fat diet-induced obesity and leptin resistance in C57Bl/6J mice. Int J Obes Relat Metab Disord. 2000;24:639–46.
Enriori PJ, Evans AE, Sinnayah P, Cowley MA. Leptin resistance and obesity. Obesity. 2006;14 Suppl 5:254S–258S.
Kim S, Jung J, Kim H, Heo RW, Yi CO, Lee JE, et al. Exendin-4 improves nonalcoholic fatty liver disease by regulating glucose transporter 4 expression in ob/ob mice. Korean J Physiol Pharmacol. 2014;18:333–9.
Acknowledgements
This study was supported by the National Natural Science Foundation of China Grant (81670782 and 81970741 FX), the Guangdong High-Level Talents Special Support Program (2016TQ03R590 FX), the Local Innovative and Research Teams Projects of Guangdong Pearl River Talents Program (2017BT 01S131 to JW and FX), the Pearl River S&T Nova Program of Guangzhou (201610010175 FX), and the NSFC-CIHR (81261120565 JW). FX was the guarantor of this study, had full access to all the data in the study, and took responsibility for the integrity of the data and the accuracy of the data analysis.
Author information
Authors and Affiliations
Contributions
FX contributed to the study design, acquisition of data, data interpretation, manuscript writing and revision. HC researched data, performed animal studies and cell culture, and analyzed data and revised the manuscript. ZC researched data, performed animal studies and analyzed data. HG contributed to the study design and data analysis. WG contributed to data analysis and manuscript revision. BL contributed to animal studies. JW contributed to the study design, data analysis, revision of the manuscript, and approval of the version to be submitted.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
About this article
Cite this article
Xu, F., Cao, H., Chen, Z. et al. Short-term GLP-1 receptor agonist exenatide ameliorates intramyocellular lipid deposition without weight loss in ob/ob mice. Int J Obes 44, 937–947 (2020). https://doi.org/10.1038/s41366-019-0513-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41366-019-0513-y
This article is cited by
-
Exenatide regulates Th17/Treg balance via PI3K/Akt/FoxO1 pathway in db/db mice
Molecular Medicine (2022)
-
Incretin-induced changes in the transcriptome of skeletal muscles of fa/fa Zucker rat (ZFR) with obesity, without diabetes
International Journal of Obesity (2022)
-
Exendin-4 may improve type 2 diabetes by modulating the epigenetic modifications of pancreatic histone H3 in STZ-induced diabetic C57BL/6 J mice
Journal of Physiology and Biochemistry (2022)