Abstract
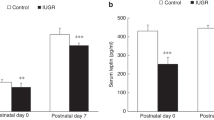
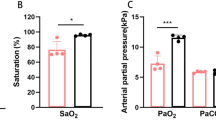
ABSTRACT: Evidence suggests that IGF and their binding proteins play a role in fetal growth, but more knowledge concerning their regulation is essential. We examined the expression of IGF and their binding proteins in experimental intrauterine growth-retarded (lUGR) rat fetuses of hypoxic dams (13–14% oxygen, d 14–21 of gestation). The mean body weight of the fetuses (d 21 of gestation, n = 72) of the six hypoxic dams was 24% lower (p < 0.0001) than the mean weight of the fetuses of six control dams (n = 82). Wet liver weights demonstrated a 20% decrease (p < 0.0001) and placentas a 10% decrease (p < 0.01) compared with control fetuses. The mean serum concentrations of immunoreactive IGF-I in both groups were low but did not differ significantly. The mean serum concentrations of immunoreactive IGF-II, however, were higher in IUGR fetuses. As assessed by Northern blot analysis, there was a 4-fold increase in insulin-like growth factor binding protein-1 (IGFBP-1) mRNA expression in the livers of the IUGR fetuses compared with controls. IGFBP-2 mRNA expression was 6-fold increased in IUGR fetal livers. No difference was found in IGFBP-4 mRNA. An increase in IGFBP-1, −2, and −4 concentrations could be seen by Western ligand blotting in the serum of growth-retarded fetuses compared with control fetuses. This finding was verified by immunoprecipitation with specific antibodies, which demonstrated increases in IGFBP-1 and IGFBP-2. Our results validate the use of maternal hypoxia as an experimental model of intrauterine growth retardation and indicate that increased IGFBP-1 and −2 expression may be of importance in the etiology of fetal growth retardation caused by maternal hypoxia.
Similar content being viewed by others
Article PDF
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Tapanainen, P., Bang, P., Wilson, K. et al. Maternal Hypoxia as a Model for Intrauterine Growth Retardation: Effects on Insulin-Like Growth Factors and Their Binding Proteins. Pediatr Res 36, 152–158 (1994). https://doi.org/10.1203/00006450-199408000-00004
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1203/00006450-199408000-00004
This article is cited by
-
Human adaptation to high altitude: a review of convergence between genomic and proteomic signatures
Human Genomics (2022)
-
Molecular mechanisms governing offspring metabolic programming in rodent models of in utero stress
Cellular and Molecular Life Sciences (2020)
-
Dexamethasone-induced Intra-Uterine Growth Restriction impacts NOSTRIN and its downstream effector genes in the rat mesometrial uterus
Scientific Reports (2018)
-
The role and regulation of IGFBP-1 phosphorylation in fetal growth restriction
Journal of Cell Communication and Signaling (2015)
-
Sleep duration and disorders in pregnancy: implications for glucose metabolism and pregnancy outcomes
International Journal of Obesity (2013)