Abstract
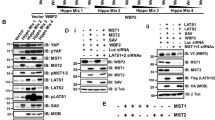
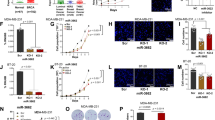
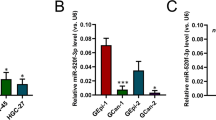
A lack of expression of miR-143 and miR-145 has been demonstrated to be a frequent feature of colorectal tumors. Activating KRAS mutations have been reported in 30–60% of colorectal cancers and an inverse correlation between Kras and miR-143/145 expression has been observed. Previously, we have demonstrated that oncogenic Kras leads to repression of the miR-143/145 cluster in pancreatic cancer and is dependent on the Ras responsive element (RRE) binding protein (RREB1), which negatively regulates miR-143/145 expression. In the present study, we have found that RREB1 is overexpressed in colorectal adenocarcinoma tumors and cell lines, and the expression of the miR-143/145 primary transcript is inversely related to RREB1 expression. In colorectal cancer cell lines, the miR-143/145 cluster is repressed by RREB1 downstream of constitutively active KRAS. RREB1 is activated by the MAPK pathway and negatively represses the miR-143/145 promoter through interaction with two RREs. In addition, overexpression of miR-143 or miR-145 in HCT116 cells abrogates signaling through the MAPK, PI3K and JNK pathways by downregulation of both KRAS and RREB1 in addition to downregulation of a cohort of genes in the MAPK signaling cascade. These results establish a complex network of regulation through which the miR-143/145 cluster is able to modulate KRAS signaling in colorectal cancer.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Ambros V . The functions of animal microRNAs. Nature 2004; 431: 350–355.
Bartel DP . MicroRNAs: genomics, biogenesis, mechanism, and function. Cell 2004; 116: 281–297.
Mendell JT . miRiad roles for the miR-17-92 cluster in development and disease. Cell 2008; 133: 217–222.
Chang TC, Wentzel EA, Kent OA, Ramachandran K, Mullendore M, Lee KH et al. Transactivation of miR-34a by p53 broadly influences gene expression and promotes apoptosis. Mol Cell 2007; 26: 745–752.
He L, He X, Lim LP, de Stanchina E, Xuan Z, Liang Y et al. A microRNA component of the p53 tumour suppressor network. Nature 2007; 447: 1130–1134.
Barbacid M . Ras genes. Annu Rev Biochem 1987; 56: 779–827.
Hingorani SR, Tuveson DA . Ras redux: rethinking how and where Ras acts. Curr Opin Genet Dev 2003; 13: 6–13.
Johnson SM, Grosshans H, Shingara J, Byrom M, Jarvis R, Cheng A et al. RAS is regulated by the let-7 microRNA family. Cell 2005; 120: 635–647.
Hatley ME, Patrick DM, Garcia MR, Richardson JA, Bassel-Duby R, van Rooij E et al. Modulation of K-Ras-dependent lung tumorigenesis by MicroRNA-21. Cancer Cell 2010; 18: 282–293.
Kent OA, Chivukula RR, Mullendore M, Wentzel EA, Feldmann G, Lee KH et al. Repression of the miR-143/145 cluster by oncogenic Ras initiates a tumor-promoting feed-forward pathway. Genes Dev 2010; 24: 2754–2759.
Date S, Nibu Y, Yanai K, Hirata J, Yagami K, Fukamizu A . Finb, a multiple zinc finger protein, represses transcription of the human angiotensinogen gene. Int J Mol Med 2004; 13: 637–642.
Mukhopadhyay NK, Cinar B, Mukhopadhyay L, Lutchman M, Ferdinand AS, Kim J et al. The zinc finger protein ras-responsive element binding protein-1 is a coregulator of the androgen receptor: implications for the role of the Ras pathway in enhancing androgenic signaling in prostate cancer. Mol Endocrinol 2007; 21: 2056–2070.
Thiagalingam A, De Bustros A, Borges M, Jasti R, Compton D, Diamond L et al. RREB-1, a novel zinc finger protein, is involved in the differentiation response to Ras in human medullary thyroid carcinomas. Mol Cell Biol 1996; 16: 5335–5345.
Thiagalingam A, Lengauer C, Baylin SB, Nelkin BD . RREB1, a ras responsive element binding protein, maps to human chromosome 6p25. Genomics 1997; 45: 630–632.
Uren AG, Kool J, Matentzoglu K, de Ridder J, Mattison J, van Uitert M et al. Large-scale mutagenesis in p19(ARF)- and p53-deficient mice identifies cancer genes and their collaborative networks. Cell 2008; 133: 727–741.
Zhang S, Qian X, Redman C, Bliskovski V, Ramsay ES, Lowy DR et al. p16 INK4a gene promoter variation and differential binding of a repressor, the ras-responsive zinc-finger transcription factor, RREB. Oncogene 2003; 22: 2285–2295.
Zhang L, Zhao J, Edenberg HJ . A human Raf-responsive zinc-finger protein that binds to divergent sequences. Nucleic Acids Res 1999; 27: 2947–2956.
Chen X, Guo X, Zhang H, Xiang Y, Chen J, Yin Y et al. Role of miR-143 targeting KRAS in colorectal tumorigenesis. Oncogene 2009; 28: 1385–1392.
Michael MZ, O'Connor SM, van Holst PNG, Young GP, James RJ . Reduced accumulation of specific microRNAs in colorectal neoplasia. Mol Cancer Res 2003; 1: 882–891.
Akao Y, Nakagawa Y, Naoe T . MicroRNAs 143 and 145 are possible common onco-microRNAs in human cancers. Oncol Rep 2006; 16: 845–850.
Kressner U, Glimelius B, Bergström R, Påhlman L, Larsson A, Lindmark G . Increased serum p53 antibody levels indicate poor prognosis in patients with colorectal cancer. Br J Cancer 1998; 11: 1848–1851.
Brink M, de Goeij AF, Weijenberg MP, Roemen GM, Lentjes MH, Pachen MM et al. K-ras oncogene mutations in sporadic colorectal cancer in The Netherlands Cohort Study. Carcinogenesis 2003; 24: 703–710.
Calcagno SR, Li S, Colon M, Kreinest PA, Thompson EA, Fields AP et al. Oncogenic K-ras promotes early carcinogenesis in the mouse proximal colon. Int J Cancer 2008; 122: 2462–2470.
Slaby O, Svoboda M, Fabian P, Smerdova T, Knoflickova D, Bednarikova M et al. Altered expression of miR-21, miR-31, miR-143 and miR-145 is related to clinicopathologic features of colorectal cancer. Oncology 2007; 72: 397–402.
Mosakhani N, Sarhadi VK, Borze I, Karjalainen-Lindsberg ML, Sundström J, Ristamäki R et al. MicroRNA profiling differentiates colorectal cancer according to KRAS status. Genes Chromosomes Cancer 2012; 51: 1–9.
Rhodes DR, Yu J, Shanker K, Deshpande N, Varambally R, Ghosh D et al. ONCOMINE: a cancer microarray database and integrated data-mining platform. Neoplasia 2004; 6: 1–6.
Kaiser S, Park YK, Franklin JL, Halberg RB, Yu M, Jessen WJ et al. Transcriptional recapitulation and subversion of embryonic colon development by mouse colon tumor models and human colon cancer. Genome Biol 2007; 8: R131.
Gaspar C, Cardoso J, Franken P, Molenaar L, Morreau H, Möslein G et al. Cross-species comparison of human and mouse intestinal polyps reveals conserved mechanisms in adenomatous polyposis coli (APC)-driven tumorigenesis. Am J Pathol 2008; 172: 1363–1380.
Sabates-Bellver J, Van der Flier LG, de Palo M, Cattaneo E, Maake C, Rehrauer H et al. Transcriptome profile of human colorectal adenomas. Mol Cancer Res 2007; 5: 1263–1275.
Zhu H, Dougherty U, Robinson V, Mustafi R, Pekow J, Kupfer S et al. EGFR signals downregulate tumor suppressors miR-143 and miR-145 in Western diet-promoted murine colon cancer: role of G1 regulators. Mol Cancer Res 2011; 9: 960–975.
Cordes KR, Sheehy NT, White MP, Berry EC, Morton SU, Muth AN et al. miR-145 and miR-143 regulate smooth muscle cell fate and plasticity. Nature 2009; 460: 705–710.
Nitz MD, Harding MA, Smith SC, Thomas S, Theodorescu D . RREB1 transcription factor splice variants in urologic cancer. Am J Pathol 2011; 179: 477–486.
Shirasawa S, Furuse M, Yokoyama N, Sasazuki T . Altered growth of human colon cancer cell lines disrupted at activated Ki-ras. Science 1993; 260: 85–88.
Keller JW, Haigis KM, Franklin JL, Whitehead RH, Jacks T, Coffey RJ . Oncogenic K-RAS subverts the antiapoptotic role of N-RAS and alters modulation of the N-RAS: gelsolin complex. Oncogene 2007; 26: 3051–3059.
Papadopoulos GL, Alexiou P, Maragkakis M, Reczko M, Hatzigeorgiou AG . DIANA-mirPath: integrating human and mouse microRNAs in pathways. Bioinformatics 2009; 25: 1991–1993.
Starr TK, Allaei R, Silverstein KA, Staggs RA, Sarver AL, Bergemann TL et al. A transposon-based genetic screen in mice identifies genes altered in colorectal cancer. Science 2009; 323: 1747–1750.
Vogelstein B, Fearon ER, Hamilton SR, Kern SE, Preisinger AC, Leppert M et al. Genetic alterations during colorectal-tumor development. N Engl J Med 1988; 319: 525–532.
Bandres E, Cubedo E, Agirre X, Malumbres R, Zarate R, Ramirez N et al. Identification by Real-time PCR of 13 mature microRNAs differentially expressed in colorectal cancer and non-tumoral tissues. Mol Cancer 2006; 5: 29.
Barker N, Ridgway RA, van Es JH, van de Wetering H, Begthel H, van den Born M et al. Crypt stem cells as the cells-of-origin of intestinal cancer. Nature 2009; 457: 608–611.
Ng EK, Tsang WP, Ng SS, Jin HC, Yu J, Li JJ et al. MicroRNA-143 targets DNA methyltransferases 3A in colorectal cancer. Br J Cancer 2009; 101: 699–706.
Borralho PM, Simões AE, Gomes SE, Lima RT, Carvalho T, Ferreira DM et al. miR-143 overexpression impairs growth of human colon carcinoma xenografts in mice with induction of apoptosis and inhibition of proliferation. PLoS One 2011; 6: e23787.
Sachdeva M, Zhu S, Wu F, Wu H, Walia V, Kumar S et al. p53 represses c-Myc through induction of the tumor suppressor miR-145. Proc Natl Acad Sci USA 2009; 106: 3207–3212.
Bissonnette M, Khare S, von Lintig FC, Wali RK, Nguyen L, Zhang Y et al. Mutational and nonmutational activation of p21ras in rat colonic azoxymethane-induced tumors: effects on mitogen-activated protein kinase, cyclooxygenase-2, and cyclin D1. Cancer Res 2000; 60: 4602–4609.
Buzzi N, Colicheo A, Boland R, de Boland AR . MAP kinases in proliferating human colon cancer Caco-2 cells. Mol Cell Biochem 2009; 328: 201–208.
Esau C, Kang X, Peralta E, Hanson E, Marcusson EG, Ravichandran LV et al. MicroRNA-143 regulates adipocyte differentiation. J Biol Chem 2004; 279: 52361–52365.
Clape C, Fritz V, Henriquet C, Apparailly F, Fernandez PL, Iborra F et al. miR-143 interferes with ERK5 signaling, and abrogates prostate cancer progression in mice. PLoS One 2009; 4: e7542.
Borralho PM, Kren BT, Castro RE, da Silva IB, Steer CJ, Rodrigues CM . MicroRNA-143 reduces viability and increases sensitivity to 5-fluorouracil in HCT116 human colorectal cancer cells. FEBS J 2009; 276: 6689–6700.
Han SX, Zhu Q, Ma JL, Zhao J, Huang C, Jia X et al. Lowered HGK expression inhibits cell invasion and adhesion in hepatocellular carcinoma cell line HepG2. World J Gastroenterol 2010; 16: 4541–4548.
Liu AW, Cai J, Zhao XL, Jiang TH, He TF, Fu HQ et al. ShRNA-targeted MAP4K4 inhibits hepatocellular carcinoma growth. Clin Cancer Res 2011; 17: 710–720.
Hao JM, Chen JZ, Sui HM, Si-Ma XQ, Li GQ, Liu C et al. A five-gene signature as a potential predictor of metastasis and survival in colorectal cancer. J Pathol 2010; 220: 475–489.
Dérijard B, Hibi M, Wu IH, Barrett T, Su B, Deng T et al. JNK1: a protein kinase stimulated by UV light and Ha-Ras that binds and phosphorylates the c-Jun activation domain. Cell 1994; 76: 1025–1037.
Cellurale C, Sabio G, Kennedy NJ, Das M, Barlow M, Sandy P et al. Requirement of c-Jun NH(2)-terminal kinase for Ras-initiated tumor formation. Mol Cell Biol 2011; 31: 1565–1576.
Sancho R, Nateri AS, de Vinuesa AG, Aguilera C, Nye E, Spencer-Dene B et al. JNK signalling modulates intestinal homeostasis and tumourigenesis in mice. EMBO J 2009; 28: 1843–1854.
Adler V, Qu Y, Smith SJ, Izotova L, Pestka S, Kung HF et al. Functional interactions of Raf and MEK with Jun-N-terminal kinase (JNK) result in a positive feedback loop on the oncogenic Ras signaling pathway. Biochemistry 2005; 44: 10784–10795.
Feldmann G, Habbe N, Dhara S, Bisht S, Alvarez H, Fendrich V et al. Hedgehog inhibition prolongs survival in a genetically engineered mouse model of pancreatic cancer. Gut 2008; 57: 1420–1430.
Campbell PM, Groehler AL, Lee KM, Ouellette MM, Khazak V, Der CJ . K-Ras promotes growth transformation and invasion of immortalized human pancreatic cells by Raf and phosphatidylinositol 3-kinase signaling. Cancer Res 2007; 67: 2098–2106.
Kent OA, Mullendore M, Wentzel EA, Lopez-Romero P, Tan AC, Alvarez H et al. A resource for analysis of microRNA expression and function in pancreatic ductal adenocarcinoma cells. Cancer Biol Ther 2009; 8: 2013–2024.
Acknowledgements
We would like to thank Dr Joshua Mendell and Dr Anirban Maitra for helpful discussions and feedback during preparation of the manuscript. We thank Dr James Eshleman for kindly providing cell lines and Norman Barker for photography assistance. We would like to thank Dr Dan Durocher and members of the Durocher laboratory at the Samuel Lunenfeld Research Institute at Mount Sinai Hospital Toronto for kindly providing laboratory space and reagents and helpful discussions during the review process of the manuscript. This work was supported in part through the NIH (R01CA120185) and by a Clinician Scientist Award to MKH from the Doris Duke Foundation Grant #: 2009040. OAK is a Life Sciences Research Foundation Fellow.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Supplementary Information accompanies the paper on the Oncogene website
Supplementary information
Rights and permissions
About this article
Cite this article
Kent, O., Fox-Talbot, K. & Halushka, M. RREB1 repressed miR-143/145 modulates KRAS signaling through downregulation of multiple targets. Oncogene 32, 2576–2585 (2013). https://doi.org/10.1038/onc.2012.266
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/onc.2012.266
Keywords
This article is cited by
-
Novel insights on perils and promises of miRNA in understanding colon cancer metastasis and progression
Medical Oncology (2023)
-
Promotion or remission: a role of noncoding RNAs in colorectal cancer resistance to anti-EGFR therapy
Cell Communication and Signaling (2022)
-
Transcriptome analysis reveals hub genes in the hepatopancreas of Exopalaemon carinicauda in response to hypoxia and reoxygenation
Aquaculture International (2021)
-
Disease-specific alteration of karyopherin-α subtype establishes feed-forward oncogenic signaling in head and neck squamous cell carcinoma
Oncogene (2020)
-
TGF-β orchestrates fibrogenic and developmental EMTs via the RAS effector RREB1
Nature (2020)