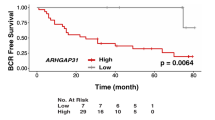
Abstract
Cystic fibrosis (CF) transmembrane conductance regulator (CFTR) is expressed in the epithelial cells of a wide range of organs/tissues from which most cancers are derived. Although accumulating reports have indicated the association of cancer incidence with genetic variations in CFTR gene, the exact role of CFTR in cancer development and the possible underlying mechanism have not been elucidated. Here, we report that CFTR expression is significantly decreased in both prostate cancer cell lines and human prostate cancer tissue samples. Overexpression of CFTR in prostate cancer cell lines suppresses tumor progression (cell growth, adhesion and migration), whereas knockdown of CFTR leads to enhanced malignancies both in vitro and in vivo. In addition, we demonstrate that CFTR knockdown-enhanced cell proliferation, cell invasion and migration are significantly reversed by antibodies against either urokinase plasminogen activator (uPA) or uPA receptor (uPAR), which are known to be involved in various malignant traits of cancer development. More interestingly, overexpression of CFTR suppresses uPA by upregulating the recently described tumor suppressor microRNA-193b (miR-193b), and overexpression of pre-miR-193b significantly reverses CFTR knockdown-enhanced malignant phenotype and abrogates elevated uPA activity in prostate cancer cell line. Finally, we show that CFTR gene transfer results in significant tumor repression in prostate cancer xenografts in vivo. Taken together, the present study has demonstrated a previously undefined tumor-suppressing role of CFTR and its involvement in regulation of miR-193b in prostate cancer development.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Collins FS. Cystic fibrosis: molecular biology and therapeutic implications. Science 1992; 256: 774–779.
Riordan JR, Rommens JM, Kerem B, Alon N, Rozmahel R, Grzelczak Z et al. Identification of the cystic fibrosis gene: cloning and characterization of complementary DNA. Science 1989; 245: 1066–1073.
Cuthbert AW. New horizons in the treatment of cystic fibrosis. Br J Pharmacol 2011; 163: 173–183.
Wiehe M, Arndt K. Cystic fibrosis: a systems review. AANA J 2010; 78: 246–251.
McWilliams RR, Rabe KG, Olswold C, De AM, Petersen GM. Risk of malignancy in first-degree relatives of patients with pancreatic carcinoma. Cancer 2005; 104: 388–394.
Neglia JP, FitzSimmons SC, Maisonneuve P, Schoni MH, Schoni-Affolter F, Corey M et al. The risk of cancer among patients with cystic fibrosis. Cystic Fibrosis and Cancer Study Group. N Engl J Med 1995; 332: 494–499.
McWilliams RR, Petersen GM, Rabe KG, Holtegaard LM, Lynch PJ, Bishop MD et al. Cystic fibrosis transmembrane conductance regulator (CFTR) gene mutations and risk for pancreatic adenocarcinoma. Cancer 2010; 116: 203–209.
Warren N, Holmes JA, al-Jader L, West RR, Lewis DC, Padua RA. Frequency of carriers of cystic fibrosis gene among patients with myeloid malignancy and melanoma. BMJ 1991; 302: 760–761.
Abraham EH, Vos P, Kahn J, Grubman SA, Jefferson DM, Ding I et al. Cystic fibrosis hetero- and homozygosity is associated with inhibition of breast cancer growth. Nat Med 1996; 2: 593–596.
Padua RA, Warren N, Grimshaw D, Smith M, Lewis C, Whittaker J et al. The cystic fibrosis delta F508 gene mutation and cancer. Hum Mutat 1997; 10: 45–48.
Qiao D, Yi L, Hua L, Xu Z, Ding Y, Shi D et al. Cystic fibrosis transmembrane conductance regulator (CFTR) gene 5T allele may protect against prostate cancer: a case-control study in Chinese Han population. J Cyst Fibros 2008; 7: 210–214.
Li Y, Sun Z, Wu Y, Babovic-Vuksanovic D, Li Y, Cunningham JM et al. Cystic fibrosis transmembrane conductance regulator gene mutation and lung cancer risk. Lung Cancer 2010; 70: 14–21.
Ding S, Gong BD, Yu J, Gu J, Zhang HY, Shang ZB et al. Methylation profile of the promoter CpG islands of 14 ‘drug-resistance’ genes in hepatocellular carcinoma. World J Gastroenterol 2004; 10: 3433–3440.
Moribe T, Iizuka N, Miura T, Kimura N, Tamatsukuri S, Ishitsuka H et al. Methylation of multiple genes as molecular markers for diagnosis of a small, well-differentiated hepatocellular carcinoma. Int J Cancer 2009; 125: 388–397.
Mishra DK, Chen Z, Wu Y, Sarkissyan M, Koeffler HP, Vadgama JV. Global methylation pattern of genes in androgen-sensitive and androgen-independent prostate cancer cells. Mol Cancer Ther 2010; 9: 33–45.
Son JW, Kim YJ, Cho HM, Lee SY, Lee SM, Kang JK et al. Promoter hypermethylation of the CFTR gene and clinical/pathological features associated with non-small cell lung cancer. Respirology 2011; 16: 1203–1209.
McMahon B, Kwaan HC. The plasminogen activator system and cancer. Pathophysiol Haemost Thromb 2008; 36: 184–194.
Malinowsky K, Bollner C, Hipp S, Berg D, Schmitt M, Becker KF. UPA and PAI-1 analysis from fixed tissues—new perspectives for a known set of predictive markers. Curr Med Chem 2010; 17: 4370–4377.
Gregory PA, Bert AG, Paterson EL, Barry SC, Tsykin A, Farshid G et al. The miR-200 family and miR-205 regulate epithelial to mesenchymal transition by targeting ZEB1 and SIP1. Nat Cell Biol 2008; 10: 593–601.
Korpal M, Lee ES, Hu G, Kang Y. The miR-200 family inhibits epithelial-mesenchymal transition and cancer cell migration by direct targeting of E-cadherin transcriptional repressors ZEB1 and ZEB2. J Biol Chem 2008; 283: 14910–14914.
Rauhala HE, Jalava SE, Isotalo J, Bracken H, Lehmusvaara S, Tammela TL et al. miR-193b is an epigenetically regulated putative tumor suppressor in prostate cancer. Int J Cancer 2010; 127: 1363–1372.
Xu C, Liu S, Fu H, Li S, Tie Y, Zhu J et al. MicroRNA-193b regulates proliferation, migration and invasion in human hepatocellular carcinoma cells. Eur J Cancer 2010; 46: 2828–2836.
Chen J, Zhang X, Lentz C, bi-Daoud M, Pare GC, Yang X et al. miR-193b Regulates Mcl-1 in Melanoma. Am J Pathol 2011; 179: 2162–2168.
Li XF, Yan PJ, Shao ZM. Downregulation of miR-193b contributes to enhance urokinase-type plasminogen activator (uPA) expression and tumor progression and invasion in human breast cancer. Oncogene 2009; 28: 3937–3948.
Hou CC, Wang W, Huang XR, Fu P, Chen TH, Sheikh-Hamad D et al. Ultrasound-microbubble-mediated gene transfer of inducible Smad7 blocks transforming growth factor-beta signaling and fibrosis in rat remnant kidney. Am J Pathol 2005; 166: 761–771.
Singh AP, Chauhan SC, Andrianifahanana M, Moniaux N, Meza JL, Copin MC et al. MUC4 expression is regulated by cystic fibrosis transmembrane conductance regulator in pancreatic adenocarcinoma cells via transcriptional and post-translational mechanisms. Oncogene 2007; 26: 30–41.
Singh AP, Moniaux N, Chauhan SC, Meza JL, Batra SK. Inhibition of MUC4 expression suppresses pancreatic tumor cell growth and metastasis. Cancer Res 2004; 64: 622–630.
Southey MC, Batten L, Andersen CR, McCredie MR, Giles GG, Dite G et al. CFTR deltaF508 carrier status, risk of breast cancer before the age of 40 and histological grading in a population-based case-control study. Int J Cancer 1998; 79: 487–489.
Smith HW, Marshall CJ. Regulation of cell signalling by uPAR. Nat Rev Mol Cell Biol 2010; 11: 23–36.
Cozzi PJ, Wang J, Delprado W, Madigan MC, Fairy S, Russell PJ et al. Evaluation of urokinase plasminogen activator and its receptor in different grades of human prostate cancer. Hum Pathol 2006; 37: 1442–1451.
Kumano M, Miyake H, Muramaki M, Furukawa J, Takenaka A, Fujisawa M. Expression of urokinase-type plasminogen activator system in prostate cancer: correlation with clinicopathological outcomes in patients undergoing radical prostatectomy. Urol Oncol 2009; 27: 180–186.
Sen R, Smale ST. Selectivity of the NF-{kappa}B response. Cold Spring Harb Perspect Biol 2010; 2: a000257.
Lawrence T. The nuclear factor NF-kappaB pathway in inflammation. Cold Spring Harb Perspect Biol 2009; 1: a001651.
Wu Y, Zhou BP. TNF-alpha/NF-kappaB/Snail pathway in cancer cell migration and invasion. Br J Cancer 2010; 102: 639–644.
Boncoeur E, Roque T, Bonvin E, Saint-Criq V, Bonora M, Clement A et al. Cystic fibrosis transmembrane conductance regulator controls lung proteasomal degradation and nuclear factor-kappaB activity in conditions of oxidative stress. Am J Pathol 2008; 172: 1184–1194.
DiMango E, Ratner AJ, Bryan R, Tabibi S, Prince A. Activation of NF-kappaB by adherent Pseudomonas aeruginosa in normal and cystic fibrosis respiratory epithelial cells. J Clin Invest 1998; 101: 2598–2605.
Vij N, Mazur S, Zeitlin PL. CFTR is a negative regulator of NFkappaB mediated innate immune response. PLoS One 2009; 4: e4664.
Hsing AW, Chokkalingam AP. Prostate cancer epidemiology. Front Biosci 2006; 11: 1388–1413.
Jemal A, Siegel R, Ward E, Murray T, Xu J, Thun MJ. Cancer statistics, 2007. Cancer J Clin 2007; 57: 43–66.
Niu Y, Altuwaijri S, Lai KP, Wu CT, Ricke WA, Messing EM et al. Androgen receptor is a tumor suppressor and proliferator in prostate cancer. Proc Natl Acad Sci USA 2008; 105: 12182–12187.
Zuker M. Mfold web server for nucleic acid folding and hybridization prediction. Nucleic Acids Res 2003; 31: 3406–3415.
Acknowledgements
The work was supported by the Focused Investment Scheme of the Chinese University of Hong Kong and National 973 project (2012CB944900), GRF-CUHK466111, and the Fundamental Research Funds for the Central Universities (JiNan University). We are grateful to Professor Tzyh-Chang Hwang at Department of Biological Engineering, University of Missouri-Columbia, USA for providing CFTR-peGFP-C3 and control plasmids. The authors thank Mrs Huang Xiao ru and Mr Qin Wei from Li Ka Shing Institute of Health Sciences, The Chinese University of Hong Kong, for their helpful technical assistance.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Supplementary Information accompanies the paper on the Oncogene website
Rights and permissions
About this article
Cite this article
Xie, C., Jiang, X., Zhang, J. et al. CFTR suppresses tumor progression through miR-193b targeting urokinase plasminogen activator (uPA) in prostate cancer. Oncogene 32, 2282–2291 (2013). https://doi.org/10.1038/onc.2012.251
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/onc.2012.251
Keywords
This article is cited by
-
Androgen-induced upregulation of CFTR in pancreatic β-cell contributes to hyperinsulinemia in PCOS model
Endocrine (2023)
-
A comprehensive review on rhubarb astringent/ laxative actions and the role of aquaporins as hub genes
Phytochemistry Reviews (2023)
-
Cystic fibrosis transmembrane conductance regulator (CFTR): beyond cystic fibrosis
Egyptian Journal of Medical Human Genetics (2022)
-
DNA Methylation-Mediated Low Expression of CFTR Stimulates the Progression of Lung Adenocarcinoma
Biochemical Genetics (2022)
-
MicroRNA-1246 regulates proliferation, invasion, and differentiation in human vascular smooth muscle cells by targeting cystic fibrosis transmembrane conductance regulator (CFTR)
Pflügers Archiv - European Journal of Physiology (2021)