Key Points
-
Adaptor proteins are as crucial as receptors and effectors as regulators of lymphocyte activation.
-
Adaptors serve as positive or negative regulators by nucleating intermolecular complexes and modulating effector protein activity.
-
Techniques to study the biology of adaptors involve genetic manipulation of cell lines and animals, biochemical assessment of intermolecular interactions, and imaging of the temporal and spatial organization of signalling complexes.
-
Adaptor domains within effector molecules may have vital roles for function. For example, SRC family kinases are regulated both by post-translational modifications (e.g. phosphorylation) and by intramolecular interactions mediated by SH2 and SH3 domains.
-
Adaptors may function cooperatively in signalling pathways (e.g. SLP76 and LAT).
-
Subcellular localization of adaptors is key to their function.
Abstract
Adaptor proteins, molecules that mediate intermolecular interactions, are now known to be as crucial for lymphocyte activation as are receptors and effectors. Extensive work from numerous laboratories has identified and characterized many of these adaptors, demonstrating their roles as both positive and negative regulators. Studies into the molecular basis for the actions of these molecules shows that they function in various ways, including: recruitment of positive or negative regulators into signalling networks, modulation of effector function by allosteric regulation of enzymatic activity, and by targeting other proteins for degradation. This review will focus on a number of adaptors that are important for lymphocyte function and emphasize the various ways in which these proteins carry out their essential roles.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Straus, D. B. & Weiss, A. Genetic evidence for the involvement of the lck tyrosine kinase in signal transduction through the T cell antigen receptor. Cell 70, 585–593 (1992).
June, C. H., Fletcher, M. C., Ledbetter, J. A. & Samelson, L. E. Increases in tyrosine phosphorylation are detectable before phospholipase C activation after T cell receptor stimulation. J. Immunol. 144, 1591–1599 (1990).
Reth, M. Antigen receptor tail clue. Nature 338, 383–384 (1989).
Chan, A. C., Irving, B. A., Fraser, J. D. & Weiss, A. The ζ chain is associated with a tyrosine kinase and upon T-cell antigen receptor stimulation associates with ZAP-70, a 70-kDa tyrosine phosphoprotein. Proc. Natl Acad. Sci. USA 88, 9166–9170 (1991).
Chan, A. C., Iwashima, M., Turck, C. W. & Weiss, A. ZAP-70: a 70 kd protein-tyrosine kinase that associates with the TCR ζ chain. Cell 71, 649–662 (1992).
Iwashima, M., Irving, B. A., van Oers, N. S., Chan, A. C. & Weiss, A. Sequential interactions of the TCR with two distinct cytoplasmic tyrosine kinases. Science 263, 1136–1139 (1994).
Jackman, J. K. et al. Molecular cloning of SLP-76, a 76-kDa tyrosine phosphoprotein associated with Grb2 in T cells. J. Biol. Chem. 270, 7029–7032 (1995).
Motto, D. G., Ross, S. E., Wu, J., Hendricks-Taylor, L. R. & Koretzky, G. A. Implication of the GRB2-associated phosphoprotein SLP-76 in T cell receptor-mediated interleukin 2 production. J. Exp. Med. 183, 1937–1943 (1996).
Yablonski, D., Kuhne, M. R., Kadlecek, T. & Weiss, A. Uncoupling of nonreceptor tyrosine kinases from PLC-γ1 in an SLP-76-deficient T cell. Science 281, 413–416 (1998).
Pivniouk, V. et al. Impaired viability and profound block in thymocyte development in mice lacking the adaptor protein SLP-76. Cell 94, 229–238 (1998).
Clements, J. L. et al. Requirement for the leukocyte-specific adapter protein SLP-76 for normal T cell development. Science 281, 416–419 (1998).References 10 and 11 describe mice with targeted disruption of the Slp76 gene. These mice have a complete block in thymocyte development, presumably due to impaired signalling through the pre-TCR, emphasizing the vital role played by SLP76 as a positive regulator of T-cell function.
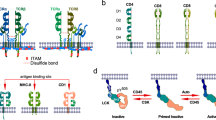
Zhang, W., Sloan-Lancaster, J., Kitchen, J., Trible, R. P. & Samelson, L. E. LAT: the ZAP-70 tyrosine kinase substrate that links T cell receptor to cellular activation. Cell 92, 83–92 (1998).
Zhang, W., Irvin, B. J., Trible, R. P., Abraham, R. T. & Samelson, L. E. Functional analysis of LAT in TCR-mediated signaling pathways using a LAT-deficient Jurkat cell line. Int. Immunol. 11, 943–950 (1999).
Zhang, W., Trible, R. P. & Samelson, L. E. LAT palmitoylation: its essential role in membrane microdomain targeting and tyrosine phosphorylation during T cell activation. Immunity 9, 239–246 (1998).
Lin, J., Weiss, A. & Finco, T. S. Localization of LAT in glycolipid-enriched microdomains is required for T cell activation. J. Biol. Chem. 274, 28861–28864 (1999).
Liu, S. K., Fang, N., Koretzky, G. A. & McGlade, C. J. The hematopoietic-specific adaptor protein Gads functions in T-cell signaling via interactions with the SLP-76 and LAT adaptors. Curr. Biol. 9, 67–75 (1999).
Asada, H. et al. Grf40, a novel Grb2 family member, is involved in T cell signaling through interaction with SLP-76 and LAT. J. Exp. Med. 189, 1383–1390 (1999).
Zhang, W. et al. Association of Grb2, Gads, and phospholipase C-γ 1 with phosphorylated LAT tyrosine residues. Effect of LAT tyrosine mutations on T cell angigen receptor-mediated signaling. J. Biol. Chem. 275, 23355–23361 (2000).
Law, C. L. et al. GrpL, a Grb2-related adaptor protein, interacts with SLP-76 to regulate nuclear factor of activated T cell activation. J. Exp. Med. 189, 1243–1253 (1999).
Zhang, W. et al. Essential role of LAT in T cell development. Immunity 10, 323–332 (1999).Similar to Slp76 -deficient animals, Lat−/− mice exhibit a complete arrest at the pro-T3 stage of thymocyte development. This result and studies in mutant variants of the Jurkat T-cell line indicate that Slp76 and LAT might function together as regulators of TCR signalling events.
Cheng, A. M. & Chan, A. C. Protein tyrosine kinases in thymocyte development. Curr. Opin. Immunol. 9, 528–533 (1997).
van Oers, N. S. T cell receptor-mediated signs and signals governing T cell development. Semin. Immunol. 11, 227–237 (1999).
Kruisbeek, A. M. et al. Branching out to gain control: how the pre-TCR is linked to multiple functions. Immunol. Today 21, 637–644 (2000).
Finco, T. S., Kadlecek, T., Zhang, W., Samelson, L. E. & Weiss, A. LAT is required for TCR-mediated activation of PLCγ1 and the Ras pathway. Immunity 9, 617–626 (1998).
Boerth, N. J. et al. Recruitment of SLP-76 to the membrane and glycolipid-enriched membrane microdomains replaces the requirement for linker for activation of T cells in T cell receptor signaling. J. Exp. Med. 192, 1047–1058 (2000).
Williams, B. L. et al. Phosphorylation of Tyr319 in ZAP-70 is required for T-cell antigen receptor-dependent phospholipase C-γ1 and Ras activation. EMBO J. 18, 1832–1844 (1999).
Lin, J. & Weiss, A. Identification of the minimal tyrosine residues required for LAT function. J. Biol. Chem. 276, 29588–29595 (2001).
Yablonski, D., Kadlecek, T. & Weiss, A. Identification of a phospholipase c-γ1 (Plc-γ1) SH3 domain-binding site in SLP-76 required for T-cell receptor-mediated activation of PLC-γ1 and NFAT. Mol. Cell. Biol. 21, 4208–4218 (2001).
Yoder, J. et al. Requirement for the SLP-76 adaptor GADS in T cell development. Science 291, 1987–1991 (2001).Describes mice made deficient in Gads. Unlike Slp76−/− or Lat−/− mice, thymocyte development is normal in Gads-deficient animals and mature T cells populate the periphery, but severe defects in thymocyte selection and function of the mature T cells which develop.
Kikuchi, K. et al. Suppression of thymic development by the dominant-negative form of Gads. Int. Immunol. 13, 777–783 (2001).
Gong, Q. et al. Disruption of T cell signaling networks and development by Grb2 haploid insufficiency. Nat. Immunol. 2, 29–36 (2001).
Krawczyk, C. et al. Cbl-b is a negative regulator of receptor clustering and raft aggregation in T cells. Immunity 13, 463–473 (2000).
Shan, X. et al. Deficiency of PTEN in Jurkat T cells causes constitutive localization of Itk to the plasma membrane and hyperresponsiveness to CD3 stimulation. Mol. Cell. Biol. 20, 6945–6957 (2000).
Fang, N. & Koretzky, G. A. SLP-76 and Vav function in separate, but overlapping pathways to augment interleukin-2 promoter activity. J. Biol. Chem. 274, 16206–16212 (1999).
Raab, M., da Silva, A. J., Findell, P. R. & Rudd, C. E. Regulation of Vav–SLP-76 binding by ZAP-70 and its relevance to TCR ζ/CD3 induction of interleukin-2. Immunity 6, 155–164 (1997).
Bubeck Wardenburg, J. et al. Regulation of PAK activation and the T cell cytoskeleton by the linker protein SLP-76. Immunity 9, 607–616 (1998).
Bokoch, G. M. et al. Interaction of the Nck adapter protein with p21-activated kinase (PAK1). J. Biol. Chem. 271, 25746–25749 (1996).
Galisteo, M. L., Chernoff, J., Su, Y. C., Skolnik, E. Y. & Schlessinger, J. The adaptor protein Nck links receptor tyrosine kinases with the serine-threonine kinase Pak1. J. Biol. Chem. 271, 20997–21000 (1996).
Lu, W., Katz, S., Gupta, R. & Mayer, B. J. Activation of Pak by membrane localization mediated by an SH3 domain from the adaptor protein Nck. Curr. Biol. 7, 85–94 (1997).
Miki, H., Yamaguchi, H., Suetsugu, S. & Takenawa, T. IRSp53 is an essential intermediate between Rac and WAVE in the regulation of membrane ruffling. Nature 408, 732–735 (2000).
Ku, G. M., Yablonski, D., Manser, E., Lim, L. & Weiss, A. A PAK1–PIX–PKL complex is activated by the T-cell receptor independent of Nck, Slp-76 and LAT. EMBO J. 20, 457–465 (2001).
Bunnell, S. C., Kapoor, V., Trible, R. P., Zhang, W. & Samelson, L. E. Dynamic actin polymerization drives T cell receptor-induced spreading: a role for the signal transduction adaptor LAT. Immunity 14, 315–329 (2001).Using real-time imaging, this paper shows that, unlike wild-type Jurkat cells, LAT -deficient Jurkat cells fail to spread on anti-TCR-coated coverslips. Reconstitution of LAT expression rescues this signalling defect; however, the structural features of LAT required for this function and the precise signalling pathways required remain to be determined.
Borroto, A. et al. Rho regulates T cell receptor ITAM-induced lymphocyte spreading in an integrin-independent manner. Eur. J. Immunol. 30, 3403–3410 (2000).
Kraynov, V. S. et al. Localized Rac activation dynamics visualized in living cells. Science 290, 333–337 (2000).Highlights the use of a method based on FRET modified to examine the spatio-temporal pattern of RAC1 activation in living cells.
Mochizuki, N. et al. Spatio-temporal images of growth-factor-induced activation of Ras and Rap1. Nature 411, 1065–1068 (2001).
Kang, P. J., Sanson, A., Lee, B. & Park, H. O. A GDP/GTP exchange factor involved in linking a spatial landmark to cell polarity. Science 292, 1376–1378 (2001).
Marston, A. L., Chen, T., Yang, M. C., Belhumeur, P. & Chant, J. A localized GTPase exchange factor, Bud5, determines the orientation of division axes in yeast. Curr. Biol. 11, 803–807 (2001).
Shamah, S. M. et al. EphA receptors regulate growth cone dynamics through the novel guanine nucleotide exchange factor ephexin. Cell 105, 233–244 (2001).
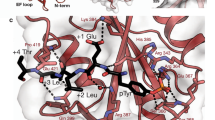
Young, M. A., Gonfloni, S., Superti-Furga, G., Roux, B. & Kuriyan, J. Dynamic coupling between the SH2 and SH3 domains of c-Src and Hck underlies their inactivation by C-terminal tyrosine phosphorylation. Cell 105, 115–126 (2001).Uses a combination of genetic and simulated crystallographic studies to show a unique role for the region linking the SH3 and SH2 domains of c-SRC and HCK.
Schmedt, C. et al. Csk controls antigen receptor-mediated development and selection of T-lineage cells. Nature 394, 901–904 (1998).
Chow, L. M., Fournel, M., Davidson, D. & Veillette, A. Negative regulation of T-cell receptor signalling by tyrosine protein kinase p50csk. Nature 365, 156–160 (1993).
Koretzky, G. A., Picus, J., Thomas, M. L. & Weiss, A. Tyrosine phosphatase CD45 is essential for coupling T-cell antigen receptor to the phosphatidyl inositol pathway. Nature 346, 66–68 (1990).
Kishihara, K. et al. Normal B lymphocyte development but impaired T cell maturation in CD45-exon6 protein tyrosine phosphatase-deficient mice. Cell 74, 143–156 (1993).
Brdicka, T. et al. Phosphoprotein associated with glycosphingolipid-enriched microdomains (PAG), a novel ubiquitously expressed transmembrane adaptor protein, binds the protein tyrosine kinase csk and is involved in regulation of T cell activation. J. Exp. Med. 191, 1591–1604 (2000).References 53 and 54 describe the initial characterization of PAG/CBP, indicating its localization in lipid rafts and its association with CSK. These papers provide evidence for how an adaptor protein might regulate an enzyme by directing its subcellular localization.
Kawabuchi, M. et al. Transmembrane phosphoprotein Cbp regulates the activities of Src-family tyrosine kinases. Nature 404, 999–1003 (2000).
Takeuchi, S., Takayama, Y., Ogawa, A., Tamura, K. & Okada, M. Transmembrane phosphoprotein Cbp positively regulates the activity of the carboxyl-terminal Src kinase, Csk. J. Biol. Chem. 275, 29183–29186 (2000).
Meng, W., Sawasdikosol, S., Burakoff, S. J. & Eck, M. J. Structure of the amino-terminal domain of Cbl complexed to its binding site on ZAP70 kinase. Nature 398, 84–90 (1999).
Joazeiro, C. A. et al. The tyrosine kinase negative regulator c-Cbl as a RING-type, E2-dependent ubiquitin-protein ligase. Science 286, 309–312 (1999)This study shows a function for c-Cbl as an E3 ubiquitin-protein ligase with direct in vitro evidence of a role for the TKB and RING domains.
Yokouchi, M. et al. Ligand-induced ubiquitination of the epidermal growth factor receptor involves the interaction of the c-Cbl RING finger and UbcH7. J. Biol. Chem. 274, 31707–31712 (1999).
Lee, P. S. et al. The Cbl protooncoprotein stimulates CSF-1 receptor multiubiquitination and endocytosis, and attenuates macrophage proliferation. EMBO J. 18, 3616–3628 (1999).
Miyake, S., Lupher, M. L. Jr, Druker, B. & Band, H. The tyrosine kinase regulator Cbl enhances the ubiquitination and degradation of the platelet-derived growth factor receptor alpha. Proc. Natl Acad. Sci. USA 95, 7927–7932 (1998).
Waterman, H., Levkowitz, G., Alroy, I. & Yarden, Y. The RING finger of c-Cbl mediates desensitization of the epidermal growth factor receptor. J. Biol. Chem. 274, 22151–22154 (1999).
Lupher, M. L. Jr et al. Cbl-mediated negative regulation of the Syk tyrosine kinase. A critical role for Cbl phosphotyrosine-binding domain binding to Syk phosphotyrosine 323. J. Biol. Chem. 273, 35273–35281 (1998).
Lupher, M. L. Jr, Reedquist, K. A., Miyake, S., Langdon, W. Y. & Band, H. A novel phosphotyrosine-binding domain in the N-terminal transforming region of Cbl interacts directly and selectively with ZAP-70 in T cells. J. Biol. Chem. 271, 24063–24068 (1996).
Lupher, M. L. Jr, Songyang, Z., Shoelson, S. E., Cantley, L. C. & Band, H. The Cbl phosphotyrosine-binding domain selects a D(N/D)XpY motif and binds to the Tyr292 negative regulatory phosphorylation site of ZAP-70. J. Biol. Chem. 272, 33140–33144 (1997).
Murphy, M. A. et al. Tissue hyperplasia and enhanced T-cell signalling via ZAP70 in c-Cbl-deficient mice. Mol. Cell. Biol. 18, 4872–4882 (1998).
Thien, C. B., Bowtell, D. D. & Langdon, W. Y. Perturbed regulation of ZAP-70 and sustained tyrosine phosphorylation of LAT and SLP76 in c-Cbl-deficient thymocytes. J. Immunol. 162, 7133–7139 (1999).
Zheng, N., Wang, P., Jeffrey, P. D. & Pavletich, N. P. Structure of a c-Cbl–UbcH7 complex: RING domain function in ubiquitin-protein ligases. Cell 102, 533–539 (2000).
Bachmaier, K. et al. Negative regulation of lymphocyte activation and autoimmunity by the molecular adaptor Cbl-b. Nature 403, 211–216 (2000).References 68 and 69 describe that Cbl-b−/− mice develop spontaneous autoimmunity after 6 months of age characterized by autoantibody production and generalized lymphocytic infiltration into multiple tissues.
Chiang, Y. J. et al. Cbl-b regulates the CD28 dependence of T-cell activation. Nature 403, 216–220 (2000).
Fang, D. et al. Cbl-b, a RING-type E3 ubiquitin ligase, targets phosphatidylinositol 3-kinase for ubiquitination in T cells. J. Biol. Chem. 276, 4872–4878 (2001).
Machesky, L. M. Putting on the brakes: a negative regulatory function for Ena/VASP proteins in cell migration. Cell 101, 685–688 (2000).
Gertler, F. B., Doctor, J. S. & Hoffmann, F. M. Genetic suppression of mutations in the Drosophila abl proto-oncogene homolog. Science 248, 857–860 (1990).
Halbrugge, M. & Walter, U. Analysis, purification and properties of a 50,000-dalton membrane-associated phosphoprotein from human platelets. J. Chromatogr. 521, 335–343 (1990).
Gertler, F. B., Niebuhr, K., Reinhard, M., Wehland, J. & Soriano, P. Mena, a relative of VASP and Drosophila Enabled, is implicated in the control of microfilament dynamics. Cell 87, 227–239 (1996).
Niebuhr, K. et al. A novel proline-rich motif present in ActA of Listeria monocytogenes and cytoskeletal proteins is the ligand for the EVH1 domain, a protein module present in the ENA/VASP family. EMBO J. 16, 5433–5444 (1997).
Carl, U. D. et al. Aromatic and basic residues within the EVH1 domain of VASP specify its interaction with proline-rich ligands. Curr. Biol. 9, 715–718 (1999).
Huttelmaier, S. et al. Characterization of the actin binding properties of the vasodilator-stimulated phosphoprotein VASP. FEBS Lett. 451, 68–74 (1999).
Bachmann, C., Fischer, L., Walter, U. & Reinhard, M. The EVH2 domain of the vasodilator-stimulated phosphoprotein mediates tetramerization, F-actin binding, and actin bundle formation. J. Biol. Chem. 274, 23549–23557 (1999).
Chakraborty, T. et al. A focal adhesion factor directly linking intracellularly motile Listeria monocytogenes and Listeria ivanovii to the actin-based cytoskeleton of mammalian cells. EMBO J. 14, 1314–1321 (1995).
Laurent, V. et al. Role of proteins of the Ena/VASP family in actin-based motility of Listeria monocytogenes. J. Cell Biol. 144, 1245–1258 (1999).
Loisel, T. P., Boujemaa, R., Pantaloni, D. & Carlier, M. F. Reconstitution of actin-based motility of Listeria and Shigella using pure proteins. Nature 401, 613–616 (1999).
Rottner, K., Behrendt, B., Small, J. V. & Wehland, J. VASP dynamics during lamellipodia protrusion. Nature Cell Biol. 1, 321–322 (1999).
Bashaw, G. J., Kidd, T., Murray, D., Pawson, T. & Goodman, C. S. Repulsive axon guidance: Abelson and Enabled play opposing roles downstream of the roundabout receptor. Cell 101, 703–715 (2000).
Bear, J. E. et al. Negative regulation of fibroblast motility by ENA/VASP proteins. Cell 101, 717–728 (2000).
Wu, J. Y. et al. The neuronal repellent Slit inhibits leukocyte chemotaxis induced by chemotactic factors. Nature 410, 948–952 (2001).
Musci, M. A. et al. Molecular cloning of SLAP130, an SLP76-associated substrate of the T cell antigen receptor-stimulated protein tyrosine kinases. J. Biol. Chem. 272, 11674–11677 (1997).
da Silva, A. J. et al. Cloning of a novel T-cell protein FYB that binds FYN and SH2-domain-containing leukocyte protein 76 and modulates interleukin 2 production. Proc. Natl Acad. Sci. USA 94, 7493–7498 (1997).
Krause, M. et al. Fyn-binding protein (Fyb)/SLP-76-associated protein (SLAP), ENA/vasodilator-stimulated phosphoprotein (VASP) proteins and the ARP2/3 complex link T cell receptor (TCR) signaling to the actin cytoskeleton. J. Cell Biol. 149, 181–194 (2000).
Obergfell, A. et al. The molecular adapter SLP-76 relays signals from platelet integrin αIIbβ3 to the actin cytoskeleton. J. Biol. Chem. 276, 5916–5923 (2001).
Fedorov, A. A., Fedorov, E., Gertler, F. & Almo, S. C. Structure of EVH1, a novel proline-rich ligand-binding module involved in cytoskeletal dynamics and neural function. Nature Struct. Biol. 6, 661–665 (1999).
Prehoda, K. E., Lee, D. J. & Lim, W. A. Structure of the enabled/VASP homology 1 domain-peptide complex: a key component in the spatial control of actin assembly. Cell 97, 471–480 (1999).
Pivniouk, V. I. et al. SLP-76 deficiency impairs signaling via the high-affinity IgE receptor in mast cells. J. Clin. Invest. 103, 1737–1743 (1999).
Clements, J. L. et al. Fetal hemorrhage and platelet dysfunction in Slp-76-deficient mice. J. Clin. Invest. 103, 19–25 (1999).
Saitoh, S. et al. LAT is essential for FcɛRI-mediated mast cell activation. Immunity 12, 525–535 (2000).
Xu, S. et al. B cell development and activation defects resulting in xid-like immunodeficiency in Blink/Slp-65-deficient mice. Int. Immunol. 12, 397–404 (2000).
Pappu, R. et al. Requirement for B cell linker protein (BLNK) in B cell development. Science 286, 1949–1954 (1999).
Jumaa, H. et al. Abnormal development and function of B lymphocytes in mice deficient for the signaling adaptor protein SLP-65. Immunity 11, 547–554 (1999).
Peterson E. J. et al. Coupling of the TCR to integrin activation by Slap-130/Fyb. Science 293, 2263–2265 (2001).
Griffiths, E. K. et al. Regulation of T cell activation and intregrin adhesion adapter Fyb/Slap. Science 293, 2260–2263 (2001).
Author information
Authors and Affiliations
Corresponding author
Glossary
- ADAPTOR MOLECULES
-
Molecules that lack any known intrinsic enzymatic, DNA binding or receptor functions, but mediate protein–protein or protein–lipid interactions. Most function as flexible molecular scaffolds by regulating the spatio-temporal dynamics of specific effector molecules.
- PROTEIN TYROSINE KINASES
-
(PTKs). Enzymes that catalyse the phosphorylation of proteins on tyrosine residues within the context of specific peptide motifs. PTKs can be generally categorized as either receptor PTKs or as cytosolic PTKs (e.g. ZAP70).
- IMMUNORECEPTOR TYROSINE-BASED ACTIVATION MOTIFS
-
(ITAMs). Regions found within the CD3 chains of the TCR and other immunoreceptors characterized by tyrosine and leucine or isoleucine residues with discrete spacing. Following receptor engagement, the tyrosines are inducibly phosphorylated and become docking sites for SH2 domain containing proteins including SYK-family protein tyrosine kinases.
- JURKAT T CELL
-
Human leukaemic T-cell line used to study several aspects of T-cell biology and signalling — in particular, signal-transduction events initiated by the TCR.
- LIPID RAFTS
-
Micro-aggregates of cholesterol and sphingomyelin thought to occur in the plasma membrane. Also described as glycolipid-enriched membrane microdomains (GEMs) or detergent-insoluble glycosphingolipid-enriched membrane microdomains (DIGs).
- GUANINE NUCLEOTIDE-EXCHANGE FACTOR
-
(GEF). Proteins that activate low-molecular-mass GTPases, such as RHO-family GTPases and RAS by stimulating the dissociation of GDP, and therefore promoting formation of the active GTP-bound state of these GTPases.
- LAMELLIPODIA
-
Thin sheet-like processes, which extend at the leading edge of moving cells or neuronal growth cones in an actin-dependent fashion; promoted by the RHO-family GTPase, RAC1.
Rights and permissions
About this article
Cite this article
Koretzky, G., Myung, P. Positive and negative regulation of t-cell activation by adaptor proteins. Nat Rev Immunol 1, 95–107 (2001). https://doi.org/10.1038/35100523
Issue Date:
DOI: https://doi.org/10.1038/35100523
This article is cited by
-
Spatial organization and signal transduction at intercellular junctions
Nature Reviews Molecular Cell Biology (2010)
-
Annexin‐A1: a pivotal regulator of the innate and adaptive immune systems
British Journal of Pharmacology (2008)
-
Efficient T-cell receptor signaling requires a high-affinity interaction between the Gads C-SH3 domain and the SLP-76 RxxK motif
The EMBO Journal (2007)
-
Csk binding protein
AfCS-Nature Molecule Pages (2006)
-
Identification and characterization of two related murine genes, Eat2a and Eat2b, encoding single SH2-domain adapters
Immunogenetics (2006)