Key Points
-
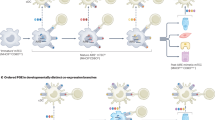
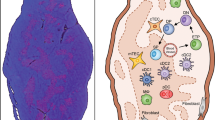
The thymus is the primary site of T-cell development. It consists of a series of stromal microenvironments that guide the differentiation of developing thymocytes into self-tolerant T cells.
-
T-cell selection events in the thymus involve the positive selection of thymocytes by cortical thymic epithelial cells (cTECs), and the negative selection of potentially autoreactive thymocytes by dendritic cells and medullary thymic epithelial cells TECs (mTECs).
-
Although cTECs and mTECs are phenotypically and functionally distinct lineages, recent studies have identified a common progenitor for both cell types in the embryonic and postnatal thymus.
-
A subset of mTECs expresses the transcriptional regulator autoimmune regulator (AIRE). This protein controls the expression of a wide array of so-called tissue-restricted antigens in the thymus so that the developing T-cell repertoire is purged of reactivity to peripheral tissues.
-
The development of AIRE-expressing mTECs is regulated by interactions between the cell-surface receptor RANK (receptor activator of nuclear factor κB) and RANK ligand, the latter being provided by a CD3−CD4+ lymphoid-tissue inducer (LTi) cell population that has been recently identified in the thymic medulla.
-
LTi cells were previously described for their key role in the development of secondary lymphoid tissues and in the propagation of memory T-cell responses. The identification of LTi cells in the thymic medulla is an example of emerging similarities in primary (thymus) and secondary (spleen) lymphoid tissues that support the development and function of self-tolerant and functional T cells.
Abstract
αβ T cells pass through a series of lymphoid tissue stromal microenvironments to acquire self tolerance and functional competence. In the thymus, positive selection of the developing T-cell receptor repertoire occurs in the cortex, whereas events in the medulla purge the system of self reactivity. T cells that survive are exported to secondary lymphoid organs where they direct first primary and then memory immune responses. This Review focuses on the microenvironments that nurture T-cell development rather than on T cells themselves. We summarize current knowledge on the formation of thymic epithelial-cell microenvironments, and highlight similarities between the environments that produce T cells and those that select and maintain them during immune responses.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Davis, M. M. et al. T-cells as a self-referential, sensory organ. Ann. Rev. Immunol. 25, 681–695 (2007).
Takahama, T. Journey through the thymus: stromal guides for T-cell development and selection. Nature Rev. Immunol. 6, 127–135 (2006).
Hayday, A. C. & Pennington, D. J. Key factors in the organised chaos of early T-cell development. Nature Immunol. 8, 137–144 (2007).
Lind, E. F., Prockop, S. E., Porritt, H. E. & Petrie, H. T. Mapping precursor movement through the postnatal thymus reveals specific microenvironments supporting defined stages of early lymphoid development. J. Exp. Med. 194, 127–134 (2001).
Kampinga, J. et al. Thymic epithelial antibodies: immunohistological analysis and introduction of nomenclature. Thymus 13, 165–173 (1989).
van de Wijngaert, F. P., Kendall, M. D., Schuurman, H. J., Rademakers, L. H. & Kater, L. Heterogeneity of epithelial cells in the human thymus. An ultrastructural study. Cell Tissue Res. 237, 227–237 (1984).
Boehm, T. & Bleul, C. C. Thymus-homing precursors and the thymic microenvironment. Trends Immunol. 27, 477–484 (2006).
Gordon, J. et al. Functional evidence for a single endodermal origin for the thymic epithelium. Nature Immunol. 5, 546–553 (2004).
Gray, D. H. D. et al. Developmental kinetics, turnover, and stimulatory capacity of thymic epithelial cells. Blood 108, 3777–3785 (2006).
Klug, D. B. et al. Interdependence of cortical thymic epithelial cell differentiation and T-lineage commitment. Proc. Natl Acad. Sci. USA 95, 11822–11827 (1998).
Klug, D. B., Carter, C., Giminez-Conti, I. B. & Richie, E. R. Thymocyte-independent and thymocyte dependent phases of epithelial patterning in the fetal thymus. J. Immunol. 169, 2842–2845 (2002).
Rossi, S. W., Jenkinson, W. E., Anderson, G. & Jenkinson, E. J. Clonal analysis reveals a common progenitor for thymic cortical and medullary epithelium. Nature 441, 988–991 (2006).
Bleul, C. C. et al. Formation of a functional thymus initiated by a postnatal epithelial progenitor cell. Nature 441, 992–996 (2006). Together with reference 12, this study provides the first evidence for the existence of bipotent TEC progenitors that give rise to both cTECs and mTECs.
Gill, J., Malin, M., Hollander, G. & Boyd, R. L. Generation of a complete thymic microenvironment by MTS24+ thymic epithelial cells. Nature Immunol. 3, 635–642 (2002).
Bennett, A. R. et al. Identification and characterization of thymic epithelial progenitor cells. Immunity 16, 803–814 (2002).
Nijhof, J. G. et al. The cell-surface marker MTS24 identifies a novel population of follicular keratinocytes with characteristics of progenitor cells. Development 133, 3027–3037 (2006).
Rossi, S. W. et al. Redefining epithelial progenitor potential in the developing thymus. Eur. J. Immunol. 37, 2411–2418 (2007).
Barthlott, T., Keller, M. P., Krenger, W. & Hollander, G. A. A short primer on early molecular and cellular events in thymus organogenesis and replacement. Swiss Med. Weekly 136, 365–369 (2006).
Redvers, R. P., Li, A. & Kaur, P. Side population in adult murine epidermis exhibits phenotypic and functional characteristics of keratinocyte stem cells. Proc. Natl Acad. Sci. USA 103, 13168–13173 (2006).
Rossi, S. W. et al. RANK signals from CD4+3− inducer cells regulate development of Aire-expressing epithelial cells in the thymic medulla. J. Exp. Med. 204, 267–1272 (2007). This study identifies an intrathymic CD3−CD4+ cell population as the provider of RANK signals that lead to the development of AIRE-expressing mTECs.
Jenkinson, W. E., Jenkinson, E. J. & Anderson, G. Differential requirement for mesenchyme in the proliferation and maturation of thymic epithelial progenitors. J. Exp. Med. 198, 325–332 (2003).
Jenkinson, W. E., Rossi, S. W., Parnell, S. M., Jenkinson, E. J. & Anderson, G. PDGFRα-expressing mesenchyme regulates thymus growth and the availability of intrathymic niches. Blood 109, 954–960 (2007).
Terszowski, G. et al. Evidence for a functional second thymus in mice. Science 312, 284–287 (2006).
Dooley, J., Erickson, M., Gillard, G. O. & Farr, A. G. Cervical thymus in the mouse. J. Immunol. 176, 6484–6489 (2006).
Jenkinson, W. E., Rossi, S. W., Jenkinson, E. J. & Anderson, G. Development of functional thymic epithelial cells occurs independently of lymphostromal interactions. Mech. Dev. 122, 1294–1299 (2005).
Hens, J. R. & Wysolmerski, J. J. Key stages of mammary gland development: molecular mechanisms involved in the formation of the embryonic mammary gland. Breast Cancer Res. 7, 220–224 (2005).
Blanpain, C., Horsley, V. & Fuchs, E. Epithelial stem cells: turning over new leaves. Cell 128, 445–458 (2007).
Bockman, D. E. & Kirby M. L. Dependence of thymus development on derivatives of the neural crest. Science 223, 498–500 (1984).
Yamazaki, H. et al. Presence and distribution of neural crest-derived cells in the murine developing thymus and their potential for differentiation. Int. Immunol. 17, 549–558 (2005).
Revest, J. M., Suniara, R. K., Kerr, K., Owen, J. J. & Dickson, C. Development of the thymus requires signaling through the fibroblast growth factor receptor R2-IIIb. J. Immunol. 167, 1954–1961 (2001).
Muller, S. M. et al. Gene targeting of VEGF-A in thymus epithelium disrupts thymus blood vessel architecture. Proc. Natl Acad. Sci. USA 102, 10587–10592 (2005). This study describes the use of a nude mouse blastocyst complementation strategy to investigate the role of candidate genes in TEC development.
Gray, D. H. et al. A unique thymic fibroblast population revealed by the monoclonal antibody MTS-15. J. Immunol. 178, 4956–4965 (2007).
Senoo, M., Pinto, F., Crum, C. P. & McKeon, F. p63 is essential for proliferative potential of stem cells in stratified epithelia. Cell 129, 523–536 (2007).
Prockop, S. E. & Petrie, H. T. Regulation of thymus size by competition for stromal niches among early T cell progenitors. J. Immunol. 173, 1604–1611 (2004).
Rossi, S. W. et al. Keratinocyte growth factor (KGF) enhances postnatal T-cell development via enhancements in proliferation and function of thymic epithelial cells. Blood 109, 3803–3811 (2007).
Penit, C., Lucas, B., Vasseur, F., Rieker, T. & Boyd, R. L. Thymic medulla epithelial cells acquire specific markers by post-mitotic maturation. Dev. Immunol. 5, 25–36 (1996).
Derbinksi, J., Schulte, A., Kyewski, B. & Klein, L. Promiscuous gene expression medullary thymic epithelial cells mirrors the peripheral self. Nature Immunol. 2, 1032–1039 (2001).
Osada, M. et al. The Wnt signaling antagonist Kremen1 is required for development of thymic architecture. Clin. Dev. Immunol. 13, 299–319 (2006).
Kuragichi, M. et al. Adenomatous polyposis coli (APC) is required for normal development of skin and thymus. PLoS Genetics 2, 1362–1374 (2006).
Pongracz, J., Hare, K., Harman, B., Anderson, G. & Jenkinson, E. J. Thymic epithelial cells provide Wnt signals to developing thymocytes. Eur. J. Immunol. 33, 1949–1956 (2003).
Goldman, K. P., Park, C. S., Kim, M., Matzinger, P. & Anderson, C. C. Thymic cortical epithelium induces self-tolerance. Eur. J. Immunol. 35, 709–717 (2005).
Anderson, G., Owen, J. J., Moore, N. C. & Jenkinson, E. J. Thymic epithelial cells provide unique signals for positive selection of CD4+8+ thymocytes in vitro. J. Exp. Med. 179, 2027–2031 (1994).
Fujimoto, Y. et al. CD83 expression influences CD4+ T-cell development in the thymus. Cell 108, 755–767 (2002).
Bowlus, C. L., Ahn, J., Chu, T. & Gruen, J. R. Cloning of a novel MHC-encoded serine peptidase highly expressed by cortical epithelial cells of the thymus. Cell. Immunol. 196, 80–86 (1999).
Murata, S. et al. Regulation of CD8+ T-cell development by thymus-specific proteases. Science 316, 1349–1353 (2007). This study identifies a new mechanism of MHC class I antigen processing and presentation in TECs that influences positive selection of CD8+ T cells.
Beers, C., et al. Cathepsin-S controls MHC class II-mediated presentation by epithelial cells in vivo. J. Immunol. 174, 1205–1212 (2005).
Nakagawa, T. et al. Cathepsin-L: critical role in Ii degradation and CD4 T-cell selection in the thymus. Science 280, 450–453 (1998).
Martinic, M. M. et al. Efficient T-cell repertoire selection in tetraparental chimaeric mice independent of thymic epithelial MHC. Proc. Natl Acad. Sci. USA 100, 1861–1866 (2003).
Choi, E. Y. et al. Thymocyte-thymocyte interaction for efficient positive selection and matureation of CD4+ T-cells. Immunity 23, 387–396 (2005).
Berg, L. J. Signalling through TEC kinases regulates conventional versus innate CD8 T-cell development. Nature Rev. Immunol. 7, 479–485 (2007).
Anderson, M., Anderson, S. K. & Farr, A. G. Thymic vasculature: organiser of the medullary epithelial compartment? Int. Immunol. 12, 1105–1110 (2000).
Rodewald, H. R., Paul, S., Haller, C., Bluethmann, H. & Blum, C. Thymus medulla consisting of epithelial islets each derived from a single progenitor. Nature 414, 763–768 (2001). This reference provides the first evidence for the existence of clonal TEC progenitors.
Naquet, P, Naspetti, M. & Boyd, R. L. Development, organisation and function of the thymic medulla in normal, immunodeficient or autoimmune mice. Semin. Immunol. 11, 47–55 (1999).
Fontenot, J. D. & Rudensky, A. A well adapted regulatory contrivance: regulatory T-cell development and the forkhead family transcription factor family Foxp3. Nature Immunol. 6, 331–337 (2005).
Aschenbrenner, K. et al. Selection of Foxp3+ regulatory T-cells specific for self antigen expressed and presented by Aire+ medullary thymic epithelial cells. Nature Immunol. 8, 351–359 (2007).
Tai, X., Cowan, M., Feigenbaum, L. & Singer, A. CD28 costimulation of developing thymocytes induces Foxp3 expression and regulatory T-cell differentiation independently of interleukin-2. Nature Immunol. 6, 152–162 (2005).
Bjorses, P., Aaltonen, J., Horelli-Kuitunen, N., Yaspo, M. L. & Peltonen, L. Gene defect behind APECED: a new clue to autoimmunity. Hum. Mol. Genet. 7, 1547–1553 (1998).
Bleschscmidt, K. et al. The mouse Aire gene: comparative genomic sequencing, gene organisation and expression. Genome Res. 9, 158–166 (1999).
Anderson, M. S. et al. Projection of an immunological self-shadow within the thymus by the aire protein. Science 298, 1395–1401 (2002). This study shows that AIRE expression by TECs is necessary for the establishment of tolerance to peripheral antigens.
Liston, A., Lesage, S., Wilson, J., Peltonen, L. & Goodnow, C. C. Aire regulates negative selection of organ-specific T-cells. Nature Immunol. 4, 350–354 (2003).
Derbinksi, J. et al. Promiscuous gene expression in thymic epithelial cells is regulated at multiple levels. J. Exp. Med. 202, 33–45 (2005).
Gillard, G. O., Dooley, J. Erikson, M. Peltonen, L. & Farr, A. G. Aire-dependent alterations in medullary thymic epithelium indicate a role for Aire in thymic epithelial differentiation. J. Immunol. 178, 3007–3015 (2007).
Gillard, G. O. & Farr, A. G. Features of medullary thymic epithelium implicate postnatal development in maintaining epithelial heterogeneity and tissue-restricted antigen expression. J. Immunol. 176, 5815–5824 (2006).
Gillard, G. O. & Farr, A. G. Contrasting models of promiscuous gene expression by thymic epithelium. J. Exp. Med. 202, 15–19 (2005).
Hamazaki, Y. et al. Medullary thymic epithelial cells expressing Aire represent a unique lineage derived from cells expressing claudin. Nature Immunol. 8, 304–311 (2007).
Kim, M. Y. et al. CD4+CD3− accessory cells costimulate primed CD4 T-cells through OX40 and CD30 at sites where T-cells collaborate with B-cells. Immunity 18, 643–654 (2003). This study identifies LTi cells in adult secondary lymphoid tissues and describes their role in T-cell responses.
Kim, M. Y. et al. Function of CD4+CD3− cells in relation to B- and T-zone stroma in spleen. Blood 109, 1602–1610 (2007).
Cupedo, T., Kraal, G. & Mebius R. E. The role of CD45+CD4+CD3− cells in lymphoid organ development. Immunol. Rev. 189, 41–50 (2002).
Derbinski, J. & Kyewski, B. Linking signalling pathways, thymic stroma integrity and autoimmunity. Trends Immunol. 26, 503–506 (2005).
Lomada, D., Liu, B., Coghlan, L., Hu, Y. & Richie, E. R. Thymus medulla formation and central tolerance are restored in IKKα−/− mice that express an IKKα transgene in keratin 5+ thymic epithelial cells. J. Immunol. 178, 829–837 (2007).
Boehm, T., Scheu, S., Pfeffer, K. & Bleul, C. C. Thymic medullary epithelial cell differentiation, thymocyte emigration, and the control of autoimmunity require lympho-epithelial cross talk via LTβR. J. Exp. Med. 198, 757–769 (2003). This study demonstrates the importance of LTβR signalling in the formation of the thymic medulla.
Chin, R. K. et al. Lymphotoxin pathway directs thymic Aire expression. Nat. Immunol. 11, 1121–1127 (2003).
Akiyama, T. et al. Dependence of self-tolerance on TRAF-6 directed development of thymic stroma. Science 308, 248–251 (2005). This paper describes the identification of a TRAF6 signalling pathway essential for the development of AIRE-expressing mTECs.
Nolte, M. A. et al. A conduit system distributes chemokines and small blood-borne molecules through the splenic white pulp. J. Exp. Med. 198, 505–512 (2003).
Drumea-Mirancea, M. et al. Characterisation of a conduit system containing laminin-5 in the thymus: a potential transport system for small molecules. J. Cell Sci. 119, 1396–1405 (2006).
Kim, M. Y. et al. OX40 ligand and CD30 ligand are expressed on adult but not neonatal CD4+3− inducer cells: evidence that IL-7 signals regulate CD30 ligand but not OX40 ligand expression. J. Immunol. 174, 6686–6691 (2005).
Kelly, K. A. & Scollay, R. Seeding of neonatal lymph nodes by T-cells and identification of a novel population of CD3−CD4+ cells. Eur. J. Immunol. 22, 329–334 (1992).
Mebius, R. E., Rennert, P. & Weissman, I. L. Developing lymph nodes collect CD4+CD3− LTβ+ cells that can differentiate into APC, NK cells, and follicular cells but not T or B cells. Immunity 7, 493–504 (1997).
Mebius, R. E., Streeter, P. R., Michie, S., Butcher, E. C. & Weissman, I. L. A developmental switch in lymphocyte homing receptor and endothelial vascular addressin expression regulates lymphocyte homing and permits CD4+CD3− cells to colonise lymph nodes. Proc. Natl Acad. Sci. USA 93, 11019–11024 (1996). References 77–79 provide the first characterization of CD3−CD4+ LTi cells in secondary lymphoid tissues.
Eberl, G. et al. An essential function for the nuclear receptor RORγt in the generation of fetal lymphoid tissue inducer cells. Nature Immunol. 5, 64–73 (2004). This study identifies RORγt as a key molecule during LTi development and the subsequent development of secondary lympoid tissues.
Boos, M. D., Yokota, Y., Eberl. G. & Kee B. L. Mature natural killer cell and lymphoid-tissue inducing cell development requires Id2-mediated suppression of E protein activity. J. Exp. Med. 204, 1119–1130 (2007).
Cupedo, T., Jansen, W., Kraal, G. & Mebius R. E. Induction of secondary and tertiary lymphoid structures in the skin. Immunity 21, 655–667 (2004).
Meier, D. et al. Ectopic lymphoid-organ development occurs through interleukin-7 mediated enhanced survival of lymphoid tissue inducer cells. Immunity 26, 643–654 (2007).
Nehls, M, Pfeifer, D., Schorpp, M., Hedrich, H. & Boehm, T. New member of the winged-helix protein family disrupted in mouse and rat nude mutations. Nature 372, 103–107 (1994). This is the first report identifying the genetic nature of the thymus defect in nude rodents.
Nehls, M. et al. Two genetically separable steps in the differentiation of thymic epithelium. Science 272, 886–889 (1996).
Blackburn, C. C. et al. The nu gene acts cell-autonomously and is required for differentiation of thymic epithelial progenitors. Proc. Natl Acad. Sci. USA 93, 5742–5746 (1996).
Dooley, J., Erickson, M. & Farr, A. G. An organised medullary epithelial structure in the normal thymus expresses molecules of respiratory epithelium and resembles the epithelial thymic rudiment of nude mice. J. Immunol. 175, 4331–4337 (2005).
Su, D. M., Navarre, S., Oh, W. J., Condie, B. G. & Manley N. R. A domain of Foxn1 required for crosstalk-dependent thymic epithelial cell differentiation. Nature Immunol. 4, 1128–1135 (2003).
Jerome, L. A. & Papaioannou, V. E. DiGeorge syndrome phenotype in mice mutant for the T-box gene, Tbx1. Nature Genet. 27, 286–291 (2001).
Ivins, S. et al. Microarray analysis detects differentially expressed genes in the pharyngeal region of mice lacking Tbx1. Dev. Biol. 285, 554–549 (2005).
Manley, N. R., Selleri, L., Brendolan, A., Gordon, J. & Cleary M. L. Abnormalities of caudal pharyngeal pouch development by Pbx1 knockout mice mimic loss of Hoxa3 paralogs. Dev. Biol. 276, 301–312 (2004).
Xu, P. X. et al. Eya1 is required for the morphogenesis of mammalian thymus, parathyroid and thyroid. Development 129, 3033–3044 (2002).
Zou, D. et al. Patterning of the third pharyngeal pouch into thymus/parathyroid by Six and Eya1. Dev. Biol. 293, 499–512 (2006).
Laclef, C., Souil, E., Demignon, J. & Maire, P. Thymus, kidney and craniofacial abnormalities in Six 1 deficient mice. Mech. Dev. 120, 669–679 (2003).
Wallin, J. et al. Pax1 is expressed during development of the thymus epithelium and is required for normal T-cell maturation. Development 122, 23–30 (1996).
Su, D. M., Ellis, S., Napier, A., Lee, K. & Manley, N. R. Hoxa3 and pax1 regulate epithelial cell death and proliferation during thymus and parathyroid organogenesis. Dev. Biol. 236, 316–329 (2001).
Su, D. M. & Manley, N. R. Hoxa3 and pax1 transcription factors regulate the ability of fetal thymic epithelial cells to promote thymocyte development. J. Immunol. 164, 5753–5760 (2000).
Peters, H., Neubuser, A., Kratochwil, K. & Balling, R. Pax9-deficient mice lack pharyngeal pouch derivatives and teeth and exhibit craniofacial and limb abnormalities. Genes Dev. 12, 2735–2747 (1998).
Hetzer-Egger, C. et al. Thymopoiesis requires Pax9 function in thymic epithelial cells. Eur. J. Immunol. 32, 1175–1181 (2002).
Manley, N. R. & Capecchi, M. R. The role of Hoxa-3 in mouse thymus and parathyroid development. Development 121, 1989–2003 (1995).
Acknowledgements
We would like to thank all current and previous laboratory members for their work leading up to this Review, and D. Withers and F. Gaspal for the confocal pictures of the thymus and spleen. We also thank many colleagues including J. Penninger, Y. Takahama, J. Caamano, R. Boyd, H.R. Rodewald and T. Boehm for useful discussions. This work was supported by the EU-funded Thymaide Integrated Project, The Wellcome Trust and The Medical Research Council.
Author information
Authors and Affiliations
Corresponding author
Related links
Glossary
- Pre-T-cell receptor
-
(pre-TCR). A cell-surface receptor complex consisting of TCRβ, pre-TCRα and CD3 that is expressed by immature CD4−CD8− thymocytes. Signalling through this complex is essential for maturation to the CD4+CD8+ stage.
- Nude mouse strain
-
A strain of mice with a naturally occurring mutation in the Foxn1 gene, that results in an early block in thymic epithelial-cell development.
- Cre-LoxP recombination
-
A tool for tissue-specific manipulation of gene expression.
- Hoechst 33342
-
An ultra-violet-excitable nucleic-acid stain that is taken up by all cells.
- Side population
-
A term used to describe stem-cell populations identified by flow cytometry on the basis of Hoechst 33342 dye efflux.
- Positive selection
-
The process by which immature CD4+CD8+ thymocytes expressing T-cell receptors with low affinity and/or avidity for self-peptide–MHC complexes are induced to differentiate into mature CD4+ and CD8+ thymocytes.
- Negative selection
-
The process by which CD4+CD8+ thymocytes expressing potentially autoreactive T-cell receptors are induced to undergo apoptosis in the thymus.
- Invariant chain
-
A protein that associates with MHC class II molecules to prevent MHC class II loading by intracellular peptides.
- Regulatory T cells
-
(TReg cells). Naturally occurring TReg cells expressing forkead box P3 (FOXP3) and CD25 that are selected in the thymus and control the activity of self-reactive T cells in the periphery.
- APECED
-
(Autoimmune polyendocrinopathy-candidiasis-ectodermal dystrophy). A polyendocrine autoimmune disease caused by loss of autoimmune regulator (AIRE) function.
Rights and permissions
About this article
Cite this article
Anderson, G., Lane, P. & Jenkinson, E. Generating intrathymic microenvironments to establish T-cell tolerance. Nat Rev Immunol 7, 954–963 (2007). https://doi.org/10.1038/nri2187
Issue Date:
DOI: https://doi.org/10.1038/nri2187
This article is cited by
-
miR-155 exerts posttranscriptional control of autoimmune regulator (Aire) and tissue-restricted antigen genes in medullary thymic epithelial cells
BMC Genomics (2022)
-
Thymic stromal cell subsets for T cell development
Cellular and Molecular Life Sciences (2016)
-
FSP1+ fibroblast subpopulation is essential for the maintenance and regeneration of medullary thymic epithelial cells
Scientific Reports (2015)
-
Analyses of the TCR repertoire of MHC class II-restricted innate CD4+ T cells
Experimental & Molecular Medicine (2015)
-
Efficient in vitro generation of functional thymic epithelial progenitors from human embryonic stem cells
Scientific Reports (2015)