Abstract
The majority of patients with advanced colorectal cancer die from hepatic metastases caused by disease progression; therefore, several novel technologies are in clinical development to potentially improve local control of liver disease. Radioembolization is a technique for administering radiotherapy internally to unresectable primary or secondary hepatic malignancies in a single procedure. This technique involves the injection of resin or glass microspheres that contain 90Y into the arterial supply of the liver. Clinical trials of radioembolization used with concomitant radiosensitizing chemotherapy have shown promising results in patients with metastatic colorectal cancer. Several reports suggest that radioembolization is associated with significant downsizing of liver metastases to permit subsequent surgical resection. In this article, the rationale for combining radioembolization with the cytotoxic and molecularly targeted agents licensed for the systemic treatment of colorectal cancer is outlined. Clinical data from trials of radioembolization with concomitant systemic treatment are reviewed, with an emphasis on the appropriateness of primary end points in large-scale trials and the practical aspects of surgical resection in patients whose tumors are successfully downsized by this chemoradiation approach.
Key Points
-
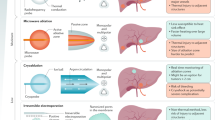
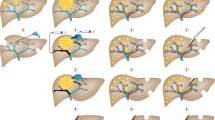
Radioembolization is a means of administering internal radiotherapy to unresectable hepatic malignancies by the injection of radioactive resin or glass microspheres into the arterial supply of the liver
-
Unlike local ablation techniques such as surgical resection or external-beam radiotherapy, radioembolization is not limited by the number and distribution of liver metastases
-
The safe administration and appropriate use of radioembolization depends on the experience of a multidisciplinary team, including an interventional vascular radiologist, radiation oncologist and nuclear medicine physician
-
Potential radiosensitizing effects of systemic agents used to treat metastatic colorectal cancer include cell-cycle arrest, apoptosis and direct and indirect effects on DNA bases, repair proteins and tumor vasculature
-
More than 10 prospective clinical trials of radioembolization are underway; these trials will determine whether it should be included in first-line treatment or subsequent lines of treatment
-
Hepatic artery embolization, fibrotic changes in the future liver remnant and portal hypertension raise questions as to the safety and radicality of resection following radioembolization
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
National Cancer Institute SEER Cancer Statistics Review, 1975–2005 [online], (2008).
Goyle, S. & Maraveyas, A. Chemotherapy for colorectal cancer. Dig. Surg. 22, 401–414 (2005).
Giantonio, B. J. et al. Bevacizumab in combination with oxaliplatin, fluorouracil, and leucovorin (FOLFOX4) for previously treated metastatic colorectal cancer: results from the Eastern Cooperative Oncology Group Study E3200. J. Clin. Oncol. 25, 1539–1544 (2007).
Nagorney, D. M. & Gigot, J. F. Primary epithelial hepatic malignancies: etiology, epidemiology, and outcome after subtotal and total hepatic resection. Surg. Oncol. Clin. N. Am. 5, 283–300 (1996).
Nordlinger, B. et al. Does chemotherapy prior to liver resection increase the potential for cure in patients with metastatic colorectal cancer? A report from the European Colorectal Metastases Treatment Group. Eur. J. Cancer 43, 2037–2045 (2007).
Hong, K., Georgiades, C. S. & Geschwind, J. F. Technology insight: image-guided therapies for hepatocellular carcinoma—intra-arterial and ablative techniques. Nat. Clin. Pract. Oncol. 3, 315–324 (2006).
Kennedy, A. et al. Recommendations for radioembolization of hepatic malignancies using 90Y microsphere brachytherapy: a consensus panel report from the radioembolization brachytherapy oncology consortium. Int. J. Radiat. Oncol. Biol. Phys. 68, 13–23 (2007).
Hamoudeh, M., Kamleh, M. A., Diab, R. & Fessi, H. Radionuclides delivery systems for nuclear imaging and radiotherapy of cancer. Adv. Drug Deliv. Rev. 60, 1329–1346 (2008).
Breedis, C. & Young, G. The blood supply of neoplasms in the liver. Am. J. Pathol. 30, 969–977 (1954).
Kennedy, A. S., Nutting, C., Coldwell, D., Gaiser, J. & Drachenberg, C. Pathologic response and microdosimetry of 90Y microspheres in man: review of four explanted whole livers. Int. J. Radiat. Oncol. Biol. Phys. 60, 1552–1563 (2004).
Campbell, A. M., Bailey, I. H. & Burton, M. A. Tumour dosimetry in human liver following hepatic 90Y microsphere therapy. Phys. Med. Biol. 46, 487–498 (2001).
Sirtex Medical Annual Report [online], (2009).
Gray, B. N., Burton, M. A., Kelleher, D. K., Anderson, J. & Klemp, P. Selective internal radiation (SIR) therapy for treatment of liver metastases: measurement of response rate. J. Surg. Oncol. 42, 192–196 (1989).
Gray, B. N. et al. Regression of liver metastases following treatment with 90Y microspheres. Aust. NZ J. Surg. 62, 105–110 (1992).
Stubbs, R. S., Cannan, R. J. & Mitchell, A. W. Selective internal radiation therapy (SIRT) with 90Yttrium microspheres for extensive colorectal liver metastases. Hepatogastroenterology 48, 333–337 (2001).
Gray, B. et al. Randomised trial of SIR-spheres plus chemotherapy vs chemotherapy alone for treating patients with liver metastases from primary large bowel cancer. Ann. Oncol. 12, 1711–1720 (2001).
Van Hazel, G. et al. Randomised phase 2 trial of SIR-spheres plus fluorouracil/leucovorin chemotherapy versus fluorouracil/leucovorin chemotherapy alone in advanced colorectal cancer. J. Surg. Oncol. 88, 78–85 (2004).
Therasse, P. et al. New guidelines to evaluate the response to treatment in solid tumors. European Organization for Research and Treatment of Cancer, National Cancer Institute of the United States, National Cancer Institute of Canada. J. Natl Cancer Inst. 92, 205–216 (2000).
Sharma, R. A. et al. Radioembolization of liver metastases from colorectal cancer using 90Yttrium microspheres with concomitant systemic oxaliplatin, fluorouracil, and leucovorin chemotherapy. J. Clin. Oncol. 25, 1099–1106 (2007).
Goldberg, R. M. et al. A randomized controlled trial of fluorouracil plus leucovorin, irinotecan, and oxaliplatin combinations in patients with previously untreated metastatic colorectal cancer. J. Clin. Oncol. 22, 23–30 (2004).
Van Hazel, G. et al. Treatment of fluourouracil-refractory patients with liver metastases from colorectal cancer by using yttrium-90 resin microspheres plus concomitant systemic irinotecan chemotherapy. J. Clin. Oncol. 27, 4089–4095 (2009).
Gulec, S. A. et al. A phase II clinical trial of yttrium-90 microspheres selective internal radiation treatment with concomitant chemotherapy as a front-line treatment in patients with colorectal cancer liver metastases: evaluation of objective tumor response by quantitative FDG-PET/CT imaging [abstract]. Ann. Surg. Oncol. 15 (Suppl. 2), a201 (2008).
Cosimelli, M. et al. Safety and efficacy of 90yttrium resin microspheres alone in unresectable, chemotherapy refractory colorectal liver metastases: results of a phase II trial [abstract]. J. Clin. Oncol. 26 (Suppl.), a4078 (2008).
Kennedy, A. S. et al. Resin 90Y-microsphere brachytherapy for unresectable colorectal liver metastases: modern USA experience. Int. J. Radiat. Oncol. Biol. Phys. 65, 412–425 (2006).
Jakobs, T. F. et al. Hepatic yttrium-90 radioembolization of chemotherapy-refractory colorectal cancer liver metastases. J. Vasc. Interv. Radiol. 19, 1187–1195 (2008).
Vente, M. A. et al. Yttrium-90 microsphere radioembolization for the treatment of liver malignancies: a structured meta-analysis. Eur. Radiol. 19, 951–959 (2008).
Steel, G. G. Terminology in the description of drug-radiation interactions. Int. J. Radiat. Oncol. Biol. Phys. 5, 1145–1150 (1979).
de Gramont, A. et al. Leucovorin and fluorouracil with or without oxaliplatin as first-line treatment in advanced colorectal cancer. J. Clin. Oncol. 18, 2938–2947 (2000).
Longley, D. B., Harkin, D. P. & Johnston, P. G. 5-fluorouracil: mechanisms of action and clinical strategies. Nat. Rev. Cancer 3, 330–338 (2003).
Spiegelman, S. et al. Improving the anti-tumor activity of 5-fluorouracil by increasing its incorporation into RNA via metabolic modulation. Proc. Natl Acad. Sci. USA 77, 4966–4970 (1980).
Bruso, C. E., Shewach, D. S. & Lawrence, T. S. Fluorodeoxyuridine-induced radiosensitization and inhibition of DNA double strand break repair in human colon cancer cells. Int. J. Radiat. Oncol. Biol. Phys. 19, 1411–1417 (1990).
Yoshioka, A. et al. Deoxyribonucleoside triphosphate imbalance. 5-fluorodeoxyuridine-induced DNA double strand breaks in mouse FM3A cells and the mechanism of cell death. J. Biol. Chem. 262, 8235–8241 (1987).
Lawrence, T. S., Davis, M. A. & Maybaum, J. Dependence of 5-fluorouracil-mediated radiosensitization on DNA-directed effects. Int. J. Radiat. Oncol. Biol. Phys. 29, 519–523 (1994).
Schlemmer, H. P. et al. Alterations of intratumoral pharmacokinetics of 5-fluorouracil in head and neck carcinoma during simultaneous radiochemotherapy. Cancer Res. 59, 2363–2369 (1999).
Miwa, M. et al. Design of a novel oral fluoropyrimidine carbamate, capecitabine, which generates 5-fluorouracil selectively in tumours by enzymes concentrated in human liver and cancer tissue. Eur. J. Cancer 34, 1274–1281 (1998).
Sawada, N., Ishikawa, T., Sekiguchi, F., Tanaka, Y. & Ishitsuka, H. X-ray irradiation induces thymidine phosphorylase and enhances the efficacy of capecitabine (Xeloda) in human cancer xenografts. Clin. Cancer Res. 5, 2948–2953 (1999).
Douillard, J. Y. et al. Irinotecan combined with fluorouracil compared with fluorouracil alone as first-line treatment for metastatic colorectal cancer: a multicentre randomised trial. Lancet 355, 1041–1047 (2000).
Hsiang, Y. H., Hertzberg, R., Hecht, S. & Liu, L. F. Camptothecin induces protein-linked DNA breaks via mammalian DNA topoisomerase I. J. Biol. Chem. 260, 14873–14878 (1985).
Giovanella, B. C. et al. DNA topoisomerase I—targeted chemotherapy of human colon cancer in xenografts. Science 246, 1046–1048 (1989).
Pizzolato, J. F. & Saltz, L. B. The camptothecins. Lancet 361, 2235–2242 (2003).
Chen, A. Y., Chou, R., Shih, S. J., Lau, D. & Gandara, D. Enhancement of radiotherapy with DNA topoisomerase I-targeted drugs. Crit. Rev. Oncol. Hematol. 50, 111–119 (2004).
Li, T. K. & Liu, L. F. Tumor cell death induced by topoisomerase-targeting drugs. Annu. Rev. Pharmacol. Toxicol. 41, 53–77 (2001).
Chen, A. Y. et al. Silatecan DB-67 is a novel DNA topoisomerase I-targeted radiation sensitizer. Mol. Cancer Ther. 4, 317–324 (2005).
Chen, A. Y., Okunieff, P., Pommier, Y. & Mitchell, J. B. Mammalian DNA topoisomerase I mediates the enhancement of radiation cytotoxicity by camptothecin derivatives. Cancer Res. 57, 1529–1536 (1997).
Cividalli, A. et al. Radiosensitization by oxaliplatin in a mouse adenocarcinoma: influence of treatment schedule. Int. J. Radiat. Oncol. Biol. Phys. 52, 1092–1098 (2002).
Kelland, L. The resurgence of platinum-based cancer chemotherapy. Nat. Rev. Cancer 7, 573–584 (2007).
Wu, B., Dröge, P. & Davey, C. A. Site selectivity of platinum anticancer therapeutics. Nat. Chem. Biol. 4, 110–112 (2008).
Espinosa, M. et al. Oxaliplatin activity in head and neck cancer cell lines. Cancer Chemother. Pharmacol. 55, 301–305 (2005).
Faivre, S., Chan, D., Salinas, R., Woynarowska, B. & Woynarowski, J. M. DNA strand breaks and apoptosis induced by oxaliplatin in cancer cells. Biochem. Pharmacol. 66, 225–237 (2003).
Koivusalo, R., Krausz, E., Ruotsalainen, P., Helenius, H. & Hietanen, S. Chemoradiation of cervical cancer cells: targeting human papillomavirus E6 and p53 leads to either augmented or attenuated apoptosis depending on the platinum carrier ligand. Cancer Res. 62, 7364–7371 (2002).
Prewett, M. C. et al. Enhanced antitumor activity of anti-epidermal growth factor receptor monoclonal antibody IMC-C225 in combination with irinotecan (CPT-11) against human colorectal tumor xenografts. Clin. Cancer Res. 8, 994–1003 (2002).
Dutta, P. R. & Maity, A. Cellular responses to EGFR inhibitors and their relevance to cancer therapy. Cancer Lett. 254, 165–177 (2007).
Yoshida, T., Okamoto, I., Iwasa, T., Fukuoka, M. & Nakagawa, K. The anti-EGFR monoclonal antibody blocks cisplatin-induced activation of EGFR signaling mediated by HB-EGF. FEBS Lett. 582, 4125–4130 (2008).
Bonner, J. A. et al. Radiotherapy plus cetuximab for squamous-cell carcinoma of the head and neck. N. Engl. J. Med. 354, 567–578 (2006).
Huang, S. M., Bock, J. M. & Harari, P. M. Epidermal growth factor receptor blockade with C225 modulates proliferation, apoptosis, and radiosensitivity in squamous cell carcinomas of the head and neck. Cancer Res. 59, 1935–1940 (1999).
Huang, S. M. & Harari, P. M. Modulation of radiation response after epidermal growth factor receptor blockade in squamous cell carcinomas: inhibition of damage repair, cell cycle kinetics, and tumor angiogenesis. Clin. Cancer Res. 6, 2166–2174 (2000).
Dittmann, K., Mayer, C. & Rodemann, H. P. Inhibition of radiation-induced EGFR nuclear import by C225 (cetuximab) suppresses DNA-PK activity. Radiother. Oncol. 76, 157–161 (2005).
Luwor, R. B. et al. The antiepidermal growth factor receptor monoclonal antibody cetuximab/C225 reduces hypoxia-inducible factor-1 alpha, leading to transcriptional inhibition of vascular endothelial growth factor expression. Oncogene 24, 4433–4441 (2005).
Fukusato, T., Mori, S., Kawamoto, T., Taniguchi, S. & Machinami, R. Immunohistochemical and ultrastructural localization of epidermal growth factor receptor in human liver and hepatocellular carcinoma tissues. Acta Pathol. Jpn. 40, 22–29 (1990).
Baselga, J. et al. Phase I studies of anti-epidermal growth factor receptor chimeric antibody C225 alone and in combination with cisplatin. J. Clin. Oncol. 18, 904–914 (2000).
Ma, J. et al. Pharmacodynamic-mediated reduction of temozolomide tumor concentrations by the angiogenesis inhibitor TNP-470. Cancer Res. 61, 5491–5498 (2001).
Jain, R. K. Normalization of tumor vasculature: an emerging concept in antiangiogenic therapy. Science 307, 58–62 (2005).
Gorski, D. H. et al. Blockage of the vascular endothelial growth factor stress response increases the antitumor effects of ionizing radiation. Cancer Res. 59, 3374–3378 (1999).
Lee, C. G. et al. Anti-vascular endothelial growth factor treatment augments tumor radiation response under normoxic or hypoxic conditions. Cancer Res. 60, 5565–5570 (2000).
Duda, D. G., Jain, R. K. & Willett, C. G. Antiangiogenics: the potential role of integrating this novel treatment modality with chemoradiation for solid cancers. J. Clin. Oncol. 25, 4033–4042 (2007).
Murthy, R. et al. Hepatic yttrium-90 radioembolotherapy in metastatic colorectal cancer treated with cetuximab or bevacizumab. J. Vasc. Interv. Radiol. 18, 1588–1591 (2007).
Riaz, A. et al. Complications following radioembolization with yttrium-90 microspheres: a comprehensive literature review. J. Vasc. Interv. Radiol. 20, 1121–1130 (2009).
Atassi, B. et al. Biliary sequelae following radioembolization with yttrium-90 microspheres. J. Vasc. Interv. Radiol. 19, 691–697 (2008).
Ng, S. S., Yu, S. C., Lai, P. B. & Lau, W. Y. Biliary complications associated with selective internal radiation (SIR) therapy for unresectable liver malignancies. Dig. Dis. Sci. 53, 2813–2817 (2008).
Van den Eynde, M. et al. Inducing resectability of chemotherapy refractory colorectal liver metastasis by radioembolization with yttrium-90 microspheres. Clin. Nucl. Med. 33, 697–699 (2008).
Jakobs, T. F. et al. Fibrosis, portal hypertension, and hepatic volume changes induced by intra-arterial radiotherapy with 90yttrium microspheres. Dig. Dis. Sci. 53, 2556–2563 (2008).
Vauthey, J. N. et al. Chemotherapy regimen predicts steatohepatitis and an increase in 90-day mortality after surgery for hepatic colorectal metastases. J. Clin. Oncol. 24, 2065–2072 (2006).
Lau, W. Y. et al. Salvage surgery following downstaging of unresectable hepatocellular carcinoma. Ann. Surg. 240, 299–305 (2004).
Stubbs, R. S. Portal hypertension and liver surgery following selective internal radiation therapy with 90Yttrium microspheres. J. Clin. Oncol. 24, e15 (2006).
Ayav, A., Habib, N. & Jiao, L. R. Portal hypertension secondary to 90Yttrium microspheres: an unknown complication. J. Clin. Oncol. 23, 8275–8276 (2005).
Gillams, A. R. & Lees, W. R. Radiofrequency ablation of colorectal liver metastases. Abdom. Imaging 30, 419–426 (2005).
Hoffmann, R. T. et al. Radiofrequency ablation after selective internal radiation therapy with 90Yttrium microspheres in metastatic liver disease-Is it feasible? Eur. J. Radiol. doi:10.1016/j.ejrad.2009.02.001.
Stubbs, R. & Wickremesekera, S. Selective internal radiation therapy (SIRT): a new modality for treating patients with colorectal liver metastases. HPB (Oxford) 6, 133–139 (2004).
Tan, T. H., Kosmider, S., Yip, D. & Gibbs, P. Clinical experience of selective internal radiation therapy in combination with systemic chemotherapy as first-line therapy in patients with unresectable hepatic metastases from colorectal cancer [abstract]. J. Clin. Oncol. 26 (Suppl.), a15080 (2008).
Ruers, T. et al. Radiofrequency ablation (RFA) combined with chemotherapy for unresectable colorectal liver metastases (CRC LM): interim results of a randomised phase II study of the EORTC-NCRI CCSG-ALM Intergroup 40004 (CLOCC) [abstract]. J. Clin. Oncol. 26 (Suppl.), a4012 (2008).
Adam, R. et al. Patients with initially unresectable colorectal liver metastases: is there a possibility of cure? J. Clin. Oncol. 27, 1829–1835 (2009).
Dzik-Jurasz, A. et al. Diffusion MRI for prediction of response of rectal cancer to chemoradiation. Lancet 360, 307–308 (2002).
Kalff, V., Duong, C., Drummond, E. G., Matthews, J. P. & Hicks, R. J. Findings on 18F-FDG PET scans after neoadjuvant chemoradiation provides prognostic stratification in patients with locally advanced rectal carcinoma subsequently treated by radical surgery. J. Nucl. Med. 47, 14–22 (2006).
Acknowledgements
This project is supported by the National Institute for Health Research Biomedical Research Centre Oxford, the Bobby Moore Fund of Cancer Research UK and the UK Medical Research Council. We thank Drs K. Vallis, D. Turner, M. Tapner and R. Adams for useful discussions and detailed comments on the manuscript.
Désirée Lie, University of California, Irvine, CA is the author of and is solely responsible for the content of the learning objectives, questions and answers of the MedscapeCME-accredited continuing medical education activity associated with this article.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
R. Sharma declares he is on the speakers' bureau and receives grant/research support from Sirtex Medical. The other authors, the Journal Editor Lisa Hutchinson and the CME questions author D. Lie declare no competing interests.
Rights and permissions
About this article
Cite this article
Nicolay, N., Berry, D. & Sharma, R. Liver metastases from colorectal cancer: radioembolization with systemic therapy. Nat Rev Clin Oncol 6, 687–697 (2009). https://doi.org/10.1038/nrclinonc.2009.165
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrclinonc.2009.165