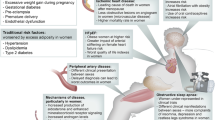
Abstract
A wealth of investigations, ranging from clinical and animal model studies to in vitro analyses, have generated great interest in the cardiovascular effects of leptin. Accordingly, many studies have examined the contribution of leptin to cardiac remodeling in heart failure and whether the effects of leptin on metabolism, apoptosis, extracellular matrix remodeling, and hypertrophy could explain the so-called obesity paradox. Furthermore, obesity and hyperleptinemia have often been associated with hypertension, and regulation of sympathetic tone or direct effects of leptin on contributors such as atherosclerosis, endothelial dysfunction, and thrombosis have been documented. Unfortunately, translating basic research studies in vitro, or in animal models, to human physiology has proven difficult. The degree of leptin resistance in obesity is one intriguing issue that must be resolved. Furthermore, the importance of autocrine and paracrine effects of leptin derived from the heart and perivascular adipose tissue must be further studied. Carefully planned and executed research to conclusively establish distinct effects of leptin on the cardiovascular system in normal and diseased states will be essential to harness any therapeutic potential associated with leptin's effects.
Key Points
-
Leptin regulates various cardiovascular effects, yet many paradoxical observations have been reported and several controversies remain
-
Reasons for these discrepancies may include the temporal nature of cardiovascular disease, actual leptin concentration examined, and the degree of crosstalk with other cardioregulatory factors
-
Leptin resistance is thought to develop in obese individuals, yet may be selective to only a subset of the physiological effects of leptin
-
Excess or inadequate leptin signaling are likely to result in unfavorable outcomes, and the maintenance of homeostatic leptin effects may be a beneficial treatment strategy
-
The myocardium and perivascular adipose tissue are known sources of leptin, which might exert important autocrine and paracrine effects
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Abel, E. D., Litwin, S. E. & Sweeney, G. Cardiac remodeling in obesity. Physiol. Rev. 88, 389–419 (2008).
Sweeney, G. Unraveling the mechanisms linking obesity and heart failure: the role of adipokines. Expert Rev. Endocrinol. Metab. 4, 95–97 (2009).
Rajapurohitam, V., Javadov, S., Purdham, D. M., Kirshenbaum, L. A. & Karmazyn, M. An autocrine role for leptin in mediating the cardiomyocyte hypertrophic effects of angiotensin II and endothelin-1. J. Mol. Cell. Cardiol. 41, 265–274 (2006).
Purdham, D. M., Zou, M. X., Rajapurohitam, V. & Karmazyn, M. Rat heart is a site of leptin production and action. Am. J. Physiol. Heart Circ. Physiol. 287, H2877–H2884 (2004).
Cheng, K. H. et al. Adipocytokines and proinflammatory mediators from abdominal and epicardial adipose tissue in patients with coronary artery disease. Int. J. Obes. (Lond.) 32, 268–274 (2008).
Rabkin, S. W. Epicardial fat: properties, function and relationship to obesity. Obes. Rev. 8, 253–261 (2007).
Silaghi, A. et al. Epicardial adipose tissue extent: relationship with age, body fat distribution, and coronaropathy. Obesity (Silver Spring) 16, 2424–2430 (2008).
Sarin, S. et al. Clinical significance of epicardial fat measured using cardiac multislice computed tomography. Am. J. Cardiol. 102, 767–771 (2008).
Iacobellis, G. et al. Influence of excess fat on cardiac morphology and function: study in uncomplicated obesity. Obes. Res. 10, 767–773 (2002).
Iacobellis, G. & Leonetti, F. Epicardial adipose tissue and insulin resistance in obese subjects. J. Clin. Endocrinol. Metab. 90, 6300–6302 (2005).
Kim, M. K. et al. Aerobic exercise training reduces epicardial fat in obese men. J. Appl. Physiol. 106, 5–11 (2009).
Willens, H. J. et al. Effects of weight loss after bariatric surgery on epicardial fat measured using echocardiography. Am. J. Cardiol. 99, 1242–1245 (2007).
Iacobellis, G., Singh, N., Wharton, S. & Sharma, A. M. Substantial changes in epicardial fat thickness after weight loss in severely obese subjects. Obesity (Silver Spring) 16, 1693–1697 (2008).
Iacobellis, G., Gao, Y. J. & Sharma, A. M. Do cardiac and perivascular adipose tissue play a role in atherosclerosis? Curr. Diab. Rep. 8, 20–24 (2008).
Stern, N. & Marcus, Y. Perivascular fat: innocent bystander or active player in vascular disease? J. Cardiometab. Syndr. 1, 115–120 (2006).
Knudson, J. D., Payne, G. A., Borbouse, L. & Tune, J. D. Leptin and mechanisms of endothelial dysfunction and cardiovascular disease. Curr. Hypertens. Rep. 10, 434–439 (2008).
Barandier, C., Montani, J. P. & Yang, Z. Mature adipocytes and perivascular adipose tissue stimulate vascular smooth muscle cell proliferation: effects of aging and obesity. Am. J. Physiol. Heart Circ. Physiol. 289, H1807–H1813 (2005).
Ahima, R. S. & Flier, J. S. Leptin. Annu. Rev. Physiol. 62, 413–437 (2000).
Martin, S. S., Qasim, A. & Reilly, M. P. Leptin resistance: a possible interface of inflammation and metabolism in obesity-related cardiovascular disease. J. Am. Coll. Cardiol. 52, 1201–1210 (2008).
Enriori, P., Evans, A. E., Sinnayah, P. & Cowley, M. A. Leptin resistance and obesity. Obesity (Silver Spring) 14, 254S–258S (2006).
Koh, K. K., Park, S. M. & Quon, M. J. Leptin and cardiovascular disease: response to therapeutic interventions. Circulation 117, 3238–3249 (2008).
Buchanan, J. et al. Reduced cardiac efficiency and altered substrate metabolism precedes the onset of hyperglycemia and contractile dysfunction in two mouse models of insulin resistance and obesity. Endocrinology 146, 5341–5349 (2005).
Christoffersen, C. et al. Cardiac lipid accumulation associated with diastolic dysfunction in obese mice. Endocrinology 144, 3483–3490 (2003).
Semeniuk, L. M., Kryski, A. J. & Severson, D. L. Echocardiographic assessment of cardiac function in diabetic db/db and transgenic db/db-hGLUT4 mice. Am. J. Physiol. Heart Circ. Physiol. 283, H976–H982 (2002).
Young, M. E. et al. Impaired long-chain fatty acid oxidation and contractile dysfunction in the obese Zucker rat heart. Diabetes 51, 2587–2595 (2002).
Konstantinides, S., Schafer, K., Koschnick, S. & Loskutoff, D. J. Leptin-dependent platelet aggregation and arterial thrombosis suggests a mechanism for atherothrombotic disease in obesity. J. Clin. Invest. 108, 1533–1540 (2001).
Bodary, P. F., Westrick, R. J., Wickenheiser, K. J., Shen, Y. & Eitzman, D. T. Effect of leptin on arterial thrombosis following vascular injury in mice. JAMA 287, 1706–1709 (2002).
Beltowski, J. Leptin and atherosclerosis. Atherosclerosis 189, 47–60 (2006).
Bell-Anderson, K. S. & Bryson, J. M. Leptin as a potential treatment for obesity: progress to date. Treat. Endocrinol. 3, 11–18 (2004).
Correia, M. L. & Rahmouni, K. Role of leptin in the cardiovascular and endocrine complications of metabolic syndrome. Diabetes Obes. Metab. 8, 603–610 (2006).
Hintz, K. K., Aberle, N. S. & Ren, J. Insulin resistance induces hyperleptinemia, cardiac contractile dysfunction but not cardiac leptin resistance in ventricular myocytes. Int. J. Obes. Relat. Metab. Disord. 27, 1196–1203 (2003).
Lopaschuk, G. D., Folmes, C. D. & Stanley, W. C. Cardiac energy metabolism in obesity. Circ. Res. 101, 335–347 (2007).
Atkinson, L. L., Fischer, M. A. & Lopaschuk, G. D. Leptin activates cardiac fatty acid oxidation independent of changes in the AMP-activated protein kinase-acetyl-CoA carboxylase-malonyl-CoA axis. J. Biol. Chem. 277, 29424–29430 (2002).
Sharma, V. et al. Stimulation of cardiac fatty acid oxidation by leptin is mediated by a nitric oxide-p38 MAPK-dependent mechanism. Eur. J. Pharmacol. 617, 113–117 (2009).
Palanivel, R., Eguchi, M., Shuralyova, I., Coe, I. & Sweeney, G. Distinct effects of short- and long-term leptin treatment on glucose and fatty acid uptake and metabolism in HL-1 cardiomyocytes. Metabolism 55, 1067–1075 (2006).
Berk, P. D. et al. Uptake of long chain free fatty acids is selectively up-regulated in adipocytes of Zucker rats with genetic obesity and non-insulin-dependent diabetes mellitus. J. Biol. Chem. 272, 8830–8835 (1997).
Luiken, J. J. et al. Increased rates of fatty acid uptake and plasmalemmal fatty acid transporters in obese Zucker rats. J. Biol. Chem. 276, 40567–40573 (2001).
Sharma, S. et al. Intramyocardial lipid accumulation in the failing human heart resembles the lipotoxic rat heart. FASEB J. 18, 1692–1700 (2004).
McGavock, J. M., Victor, R. G., Unger, R. H. & Szczepaniak, L. S. Adiposity of the heart, revisited. Ann. Intern. Med. 144, 517–524 (2006).
Szczepaniak, L. S. et al. Myocardial triglycerides and systolic function in humans: in vivo evaluation by localized proton spectroscopy and cardiac imaging. Magn. Reson. Med. 49, 417–423 (2003).
Mazumder, P. K. et al. Impaired cardiac efficiency and increased fatty acid oxidation in insulin-resistant ob/ob mouse hearts. Diabetes 53, 2366–2374 (2004).
Kamohara, S., Burcelin, R., Halaas, J. L., Friedman, J. M. & Charron, M. J. Acute stimulation of glucose metabolism in mice by leptin treatment. Nature 389, 374–377 (1997).
Haap, M. et al. Insulin-like effect of low-dose leptin on glucose transport in Langendorff rat hearts. Exp. Clin. Endocrinol. Diabetes 111, 139–145 (2003).
Wang, P., Lloyd, S. G., Zeng, H., Bonen, A. & Chatham, J. C. Impact of altered substrate utilization on cardiac function in isolated hearts from Zucker diabetic fatty rats. Am. J. Physiol. Heart Circ. Physiol. 288, H2102–H2110 (2005).
Golfman, L. S. et al. Activation of PPARgamma enhances myocardial glucose oxidation and improves contractile function in isolated working hearts of ZDF rats. Am. J. Physiol. Endocrinol. Metab. 289, E328–E336 (2005).
Lee, Y. & Gustafsson, A. B. Role of apoptosis in cardiovascular disease. Apoptosis 14, 536–548 (2009).
Narula, J. et al. Apoptosis in myocytes in end-stage heart failure. N. Engl. J. Med. 335, 1182–1189 (1996).
Olivetti, G. et al. Apoptosis in the failing human heart. N. Engl. J. Med. 336, 1131–1141 (1997).
Todor, A. et al. Hypoxia-induced cleavage of caspase-3 and DFF45/ICAD in human failed cardiomyocytes. Am. J. Physiol. Heart Circ. Physiol. 283, H990–H995 (2002).
Sung, M. M. et al. Matrix metalloproteinase-2 degrades the cytoskeletal protein alpha-actinin in peroxynitrite mediated myocardial injury. J. Mol. Cell. Cardiol. 43, 429–436 (2007).
Communal, C. et al. Functional consequences of caspase activation in cardiac myocytes. Proc. Natl Acad. Sci. USA 99, 6252–6256 (2002).
Shin, E. J., Schram, K., Zheng, X. L. & Sweeney, G. Leptin attenuates hypoxia/reoxygenation-induced apoptosis in rat H9c2 cardiomyocytes. J. Cell. Physiol. (in press).
Eguchi, M., Liu, Y., Shin, E. J. & Sweeney, G. Leptin protects H9c2 rat cardiomyocytes from H2O2-induced apoptosis. FEBS J. 275, 3136–3144 (2008).
McGaffin, K. R., Zou, B., McTiernan, C. F. & O'Donnell, C. P. Leptin attenuates cardiac apoptosis after chronic ischaemic injury. Cardiovasc. Res. 83, 313–324 (2009).
Barouch, L. et al. Cardiac myocytes apopotosis is associated with increased DNA damage and decreased survival in murine models of obesity. Circ. Res. 98, 119–124 (2006).
Trivedi, P., Yang, R. & Barouch, L. A. Decreased p110alpha catalytic activity accompanies increased myocyte apoptosis and cardiac hypertrophy in leptin deficient ob/ob mice. Cell Cycle 7, 560–565 (2008).
Smith, C. C. et al. Leptin, the obesity-associated hormone, exhibits direct cardioprotective effects. Br. J. Pharmacol. 149, 5–13 (2006).
Zhou, Y. T. et al. Lipotoxic heart disease in obese rats: implications for human obesity. Proc. Natl Acad. Sci. USA 97, 1784–1789 (2000).
Dixon, R. A., Davidson, S. M., Wynne, A. M., Yellon, D. M. & Smith, C. C. The cardioprotective actions of leptin are lost in the Zucker obese (fa/fa) rat. J. Cardiovasc. Pharmacol. 53, 311–317 (2009).
Florea, V. G. et al. Left ventricular remodelling: common process in patients with different primary myocardial disorders. Int. J. Cardiol. 68, 281–287 (1999).
Cohn, J. N. Structural basis for heart failure. Ventricular remodeling and its pharmacological inhibition. Circulation 91, 2504–2507 (1995).
Frey, N. & Olson, E. N. Cardiac hypertrophy: the good, the bad, and the ugly. Annu. Rev. Physiol. 65, 45–79 (2003).
Frey, N., Katus, H. A., Olson, E. N. & Hill, J. A. Hypertrophy of the heart: a new therapeutic target? Circulation 109, 1580–1589 (2004).
Madani, S., De Girolamo, S., Munoz, D. M., Li, R. K. & Sweeney, G. Direct effects of leptin on size and extracellular matrix components of human pediatric ventricular myocytes. Cardiovasc. Res. 69, 716–725 (2006).
Rajapurohitam, V., Gan, X. T., Kirshenbaum, L. A. & Karmazyn, M. The obesity-associated peptide leptin induces hypertrophy in neonatal rat ventricular myocytes. Circ. Res. 93, 277–279 (2003).
Xu, F. P. et al. Leptin induces hypertrophy via endothelin-1-reactive oxygen species pathway in cultured neonatal rat cardiomyocytes. Circulation 110, 1269–1275 (2004).
Majumdar, P. et al. Leptin and endothelin-1 mediated increased extracellular matrix protein production and cardiomyocyte hypertrophy in diabetic heart disease. Diabetes Metab. Res. Rev. 25, 452–463 (2009).
Zeidan, A., Javadov, S. & Karmazyn, M. Essential role of Rho/ROCK-dependent processes and actin dynamics in mediating leptin-induced hypertrophy in rat neonatal ventricular myocytes. Cardiovasc. Res. 72, 101–111 (2006).
Zeidan, A., Javadov, S., Chakrabarti, S. & Karmazyn, M. Leptin-induced cardiomyocyte hypertrophy involves selective caveolae and RhoA/ROCK-dependent p38 MAPK translocation to nuclei. Cardiovasc. Res. 77, 64–72 (2008).
Abe, Y. et al. Leptin induces elongation of cardiac myocytes and causes eccentric left ventricular dilatation with compensation. Am. J. Physiol. Heart Circ. Physiol. 292, H2387–H2396 (2007).
Pinieiro, R. et al. Leptin does not induce hypertrophy, cell cycle alterations, or production of MCP-1 in cultured rat and mouse cardiomyocytes. Endocr. Res. 31, 375–386 (2005).
Park, S. Y. et al. Unraveling the temporal pattern of diet-induced insulin resistance in individual organs and cardiac dysfunction in C57BL/6 mice. Diabetes 54, 3530–3540 (2005).
Purdham, D. M. et al. A neutralizing leptin receptor antibody mitigates hypertrophy and hemodynamic dysfunction in the postinfarcted rat heart. Am. J. Physiol. Heart Circ. Physiol. 295, H441–H446 (2008).
Madani, S., De Girolamo, S., Muñoz, D. M., Li, R. K. & Sweeney, G. Direct effects of leptin on size and extracellular matrix components of human pediatric ventricular myocytes. Cardiovasc. Res. 69, 716–725 (2005).
Wakatsuki, T., Schlessinger, J. & Elson, E. L. The biochemical response of the heart to hypertension and exercise. Trends Biochem. Sci. 29, 609–617 (2004).
Dorn, G. W. 2nd & Force, T. Protein kinase cascades in the regulation of cardiac hypertrophy. J. Clin. Invest. 115, 527–537 (2005).
McGaffin, K. R. et al. Leptin signalling reduces the severity of cardiac dysfunction and remodelling after chronic ischaemic injury. Cardiovasc. Res. 77, 54–63 (2008).
Barouch, L. A., Berkowitz, D. E., Harrison, R. W., O'Donnell, C. P. & Hare, J. M. Disruption of leptin signaling contributes to cardiac hypertrophy independently of body weight in mice. Circulation 108, 754–759 (2003).
Fedak, P., Verma, S., Weisel, R. D. & Li, R. K. Cardiac remodeling and failure: From molecules to man (Part II). Cardiovasc. Pathol. 14, 49–60 (2005).
Fedak, P., Verma, S., Weisel, R. D. & Li, R. K. Cardiac remodeling and failure: from molecules to man (Part I). Cardiovasc. Pathol. 14, 1–11 (2005).
Graham, H. K., Horn, M. & Trafford, A. W. Extracellular matrix profiles in the progression to heart failure. European Young Physiologists Symposium Keynote Lecture-Bratislava 2007. Acta Physiol. (Oxf.) 194, 3–21 (2008).
Schram, K. & Sweeney, G. Implications of myocardial matrix remodeling by adipokines in obesity-related heart failure. Trends Cardiovasc. Med. 18, 199–205 (2008).
Schram, K. et al. Increased expression and cell surface localization of MT1-MMP plays a role in stimulation of MMP-2 activity by leptin in neonatal rat cardiac myofibroblasts. J. Mol. Cell. Cardiol. 44, 874–881 (2008).
Zaman, A. K. et al. Angiotensin-converting enzyme inhibition attenuates hypofibrinolysis and reduces cardiac perivascular fibrosis in genetically obese diabetic mice. Circulation 103, 3123–3128 (2001).
Zaman, A. K. et al. Salutary effects of attenuation of angiotensin II on coronary perivascular fibrosis associated with insulin resistance and obesity. J. Mol. Cell. Cardiol. 37, 525–535 (2004).
Toblli, J. E., Cao, G., DeRosa, G. & Forcada, P. Reduced cardiac expression of plasminogen activator inhibitor 1 and transforming growth factor beta1 in obese Zucker rats by perindopril. Heart 91, 80–86 (2005).
Xia, Q. et al. Improvement of chronic heart failure by dexamehtason is not associated with downregulation of leptin in rats. Acta Pharmacologica Sinica 28, 202–210 (2007).
Parish, R. C. & Evans, J. D. Inflammation in chronic heart failure. Ann. Pharmacother. 42, 1002–1016 (2008).
Yndestad, A. et al. Role of inflammation in the progression of heart failure. Curr. Cardiol. Rep. 9, 236–241 (2007).
Yndestad, A. et al. Systemic inflammation in heart failure--the whys and wherefores. Heart Fail. Rev. 11, 83–92 (2006).
Chao, W. Toll-like receptor signaling: a critical modulator of cell survival and ischemic injury in the heart. Am. J. Physiol. Heart Circ. Physiol. 296, H1–H12 (2009).
Satoh, M., Ishikawa, Y., Minami, Y., Takahashi, Y. & Nakamura, M. Role of Toll like receptor signaling pathway in ischemic coronary artery disease. Front. Biosci. 13, 6708–6715 (2008).
Batra, A. et al. Leptin-dependent toll-like receptor expression and responsiveness in preadipocytes and adipocytes. Am. J. Pathol. 170, 1931–1941 (2007).
Heymans, S. et al. Inflammation as a therapeutic target in heart failure? A scientific statement from the Translational Research Committee of the Heart Failure Association of the European Society of Cardiology. Eur. J. Heart Fail. 11, 119–129 (2009).
Haynes, W. G. Role of leptin in obesity-related hypertension. Exp. Physiol. 90, 683–688 (2005).
Rahmouni, K., Morgan, D. A., Morgan, G. M., Mark, A. L. & Haynes, W. G. Role of selective leptin resistance in diet-induced obesity hypertension. Diabetes 54, 2012–2018 (2005).
Tumer, N., Erdos, B., Matheny, M., Cudykier, I. & Scarpace, P. J. Leptin antagonist reverses hypertension caused by leptin overexpression, but fails to normalize obesity-related hypertension. J. Hypertens. 25, 2471–2478 (2007).
Su, W. et al. Hypertension and disrupted blood pressure circadian rhythm in type 2 diabetic db/db mice. Am. J. Physiol. Heart Circ. Physiol. 295, H1634–H1641 (2008).
Osmond, J. M., Mintz, J. D., Dalton, B. & Stepp, D. W. Obesity increases blood pressure, cerebral vascular remodeling, and severity of stroke in the Zucker rat. Hypertension 53, 381–386 (2009).
Konstantinidis, D., Paletas, K., Koliakos, G. & Kaloyianni, M. Signaling components involved in leptin-induced amplification of the atherosclerosis-related properties of human monocytes. J. Vasc. Res. 46, 199–208 (2009).
Korda, M., Kubant, R., Patton, S. & Malinski, T. Leptin-induced endothelial dysfunction in obesity. Am. J. Physiol. Heart Circ. Physiol. 295, H1514–H1521 (2008).
Knudson, J. D. et al. Leptin receptors are expressed in coronary arteries, and hyperleptinemia causes significant coronary endothelial dysfunction. Am. J. Physiol. Heart Circ. Physiol. 289, H48–H56 (2005).
Procopio, C. et al. Leptin-stimulated endothelial nitric-oxide synthase via an adenosine 5′-monophosphate-activated protein kinase/Akt signaling pathway is attenuated by interaction with C-reactive protein. Endocrinology 150, 3584–3593 (2009).
Knudson, J. D. et al. Leptin resistance extends to the coronary vasculature in prediabetic dogs and provides a protective adaptation against endothelial dysfunction. Am. J. Physiol. Heart Circ. Physiol. 289, H1038–H1046 (2005).
Bodary, P. F. Links between adipose tissue and thrombosis in the mouse. Arterioscler. Thromb. Vasc. Biol. 27, 2284–2291 (2007).
Nakata, M., Yada, T., Soejima, N. & Maruyama, I. Leptin promotes aggregation of human platelets via the long form of its receptor. Diabetes 48, 426–429 (1999).
Dellas, C. et al. Absence of leptin resistance in platelets from morbidly obese individuals may contribute to the increased thrombosis risk in obesity. Thromb. Haemost. 100, 1123–1129 (2008).
Elbatarny, H. S. & Maurice, D. H. Leptin-mediated activation of human platelets: involvement of a leptin receptor and phosphodiesterase 3A-containing cellular signaling complex. Am. J. Physiol. Endocrinol. Metab. 289, E695–E702 (2005).
Acknowledgements
The author's work is funded by the Canadian Institutes of Health Research, Heart & Stroke Foundation of Canada, Canadian Diabetes Association and Korean Ministry of Education Science and Technology. Figures are intended to convey general conclusions from published literature and exceptions may exist.
Author information
Authors and Affiliations
Ethics declarations
Competing interests
The author declares no competing financial interests.
Rights and permissions
About this article
Cite this article
Sweeney, G. Cardiovascular effects of leptin. Nat Rev Cardiol 7, 22–29 (2010). https://doi.org/10.1038/nrcardio.2009.224
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrcardio.2009.224
This article is cited by
-
Atherosclerotic plaque development in mice is enhanced by myeloid ZEB1 downregulation
Nature Communications (2023)
-
A murine model of ischemia–reperfusion: the perfusion with leptin promotes the apoptosis-related relocation of mitochondrial proteins Bax and cytochrome c
Bulletin of the National Research Centre (2022)
-
Highlights of mechanisms and treatment of obesity-related hypertension
Journal of Human Hypertension (2022)