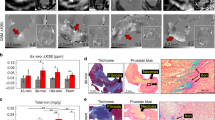
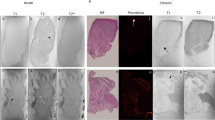
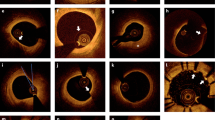
Abstract
Imaging myocardial angiogenesis presents a major technical challenge because the ideal spatial resolution required is substantially higher than that available with standard X-ray angiography and nuclear medicine imaging. Moreover, these clinical imaging methods are currently inadequate (because of insufficient resolution) for clinical trials of angiogenic agents for the treatment of ischemic heart disease. Specialized techniques in MRI, ultrasonography, echocardiography and CT that are under development might provide improved means of imaging myocardial angiogenesis. Molecular imaging technologies are also being developed to improve resolution and to provide a better mechanistic insight into angiogenic therapies for ischemic heart diseases. This Review examines advanced methods for imaging angiogenesis. These technologies might soon permit data to be obtained directly from scientific studies and clinical trials.
Key Points
-
Progress has been made in imaging technologies to assess myocardial angiogenesis
-
The development of molecular probes specific for different stages of angiogenesis might further improve the resolution of clinical imaging
-
Two-photon emission microscopy is an excellent example of combining activated probes to examine subcellular processes in the myocardium in vivo
-
Generation of molecular beacons for receptors, proteins and markers of angiogenesis conjugated to iron and gadolinium allows for even more accurate magnetic resonance imaging
-
Objective imaging end points will limit placebo effects seen in patients with chronic ischemia when subjective clinical outcome measures such as angina and quality of life questionnaires are used
-
Large-scale clinical studies should be undertaken using new imaging technologies with higher sensitivity and specificity
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
AHA. AHA statistics. American Heart Association [online], (2004).
Jolicoeur, E. M. et al. Clinical and research issues regarding chronic advanced coronary artery disease: Report from a think tank. Part 1: Contemporary and emerging therapies. Am. Heart J. 155, 418–434 (2008).
Folkman, J. Angiogenic therapy of the human heart. Circulation 97, 628–629 (1998).
Henry, T. D. Can we really grow new blood vessels? Lancet 351, 1826–1827 (1998).
Simons, M. Angiogenesis: Where do we stand now? Circulation 111, 1556–1566 (2005).
Carmeliet, P. Mechanisms of angiogenesis and arteriogenesis. Nat. Med. 6, 389–395 (2000).
Carmeliet, P. Basic concepts of (myocardial) angiogenesis: role of vascular endothelial growth factor and angiopoietin. Curr. Interv. Cardiol. Rep. 1, 322–335 (1999).
Losordo, D. W. & Dimmeler, S. Therapeutic angiogenesis and vasculogenesis for ischemic disease: Part I: Angiogenic cytokines. Circulation 109, 2487–2491 (2004).
Losordo, D. W. & Dimmeler, S. Therapeutic angiogenesis and vasculogenesis for ischemic disease: Part II: Cell-based therapies. Circulation 109, 2692–2697 (2004).
Eliceiri, B. P. & Cheresh, D. A. The role of αV integrins during angiogenesis: insights into potential mechanisms of action and clinical development. J. Clin. Invest. 103, 1227–1230 (1999).
Eliceiri, B. P. & Cheresh, D. A. The role of αV integrins during angiogenesis. Mol. Med. 4, 741–750 (1998).
Eliceiri, B. P. et al. Selective requirement for Src kinases during VEGF-induced angiogenesis and vascular permeability. Mol. Cell 4, 915–924 (1999).
Varner, J. A., Brooks, P. C. & Cheresh, D. A. Review: the integrin αVβ3: angiogenesis and apoptosis. Cell Adhes. Commun. 3, 367–374 (1995).
Lindahl, P. et al. Role of platelet-derived growth factors in angiogenesis and alveogenesis. Curr. Top. Pathol. 93, 27–33 (1999).
Gohongi, T. et al. Tumor-host interactions in the gallbladder suppress distal angiogenesis and tumor growth: involvement of transforming growth factor β1. Nat. Med. 5, 1203–1208 (1999).
Schaper, W. & Ito, W. D. Therapeutic targets in cardiovascular disorders. Curr. Opin. Biotechnol. 7, 635–640 (1996).
Schaper, W. & Ito, W. D. Molecular mechanisms of coronary collateral vessel growth. Circ. Res. 79, 911–919 (1996).
Semenza, G. L. Hypoxia-inducible factor 1: master regulator of O2 homeostasis. Curr. Opin. Genet. Dev. 8, 588–594 (1998).
Li, J. et al. PR39, a peptide regulator of angiogenesis. Nat. Med. 6, 49–55 (2000).
Kodama, K. et al. Collateral channels that develop after an acute myocardial infarction prevent subsequent left ventricular dilation. J. Am. Coll. Cardiol. 27, 1133–1139 (1996).
Schumacher, B., Pecher, P., von Specht, B. U. & Stegmann, T. Induction of neoangiogenesis in ischemic myocardium by human growth factors: first clinical results of a new treatment of coronary heart disease. Circulation 97, 645–650 (1998).
Laham, R. J. et al. Local perivascular delivery of basic fibroblast growth factor in patients undergoing coronary bypass surgery: results of a phase I randomized, double-blind, placebo-controlled trial. Circulation 100, 1865–1871 (1999).
Simons, M. et al. Pharmacological treatment of coronary artery disease with recombinant fibroblast growth factor-2: double-blind, randomized, controlled clinical trial. Circulation 105, 788–793 (2002).
Henry, T. D. et al. The VIVA trial: Vascular endothelial growth factor in Ischemia for Vascular Angiogenesis. Circulation 107, 1359–1365 (2003).
Henry, T. D. et al. Effects of Ad5FGF-4 in patients with angina. An analysis of pooled data from the AGENT-3 and AGENT-4 trials. J. Am. Coll. Cardiol. 50, 1038–1046 (2007).
Kastrup, J. et al. Direct intramyocardial plasmid vascular endothelial growth factor-A165 gene therapy in patients with stable severe angina pectoris: a randomized double-blind placebo-controlled study: the Euroinject One Trial. J. Am. Coll. Cardiol. 45, 982–988 (2005).
Losordo, D. W. et al. Intramyocardial transplantation of autologous CD34+ stem cells for intractable angina: a phase I/IIa double-blind randomized controlled trial. Circulation 115, 3165–3172 (2007).
Miettinen, M., Lindenmayer, A. E. & Chaubal, A. Endothelial cell markers CD31, CD34, and BNH9 antibody to H- and Y-antigens--evaluation of their specificity and sensitivity in the diagnosis of vascular tumors and comparison with von Willebrand factor. Mod. Pathol. 7, 82–90 (1994).
Montanez, E., Casaroli-Marano, R. P., Vilaro, S. & Pagan, R. Comparative study of tube assembly in three-dimensional collagen matrix and on Matrigel coats. Angiogenesis 5, 167–172 (2002).
Lamping, K. G., Christensen, L. P. & Tomanek, R. J. Estrogen therapy induces collateral and microvascular remodeling. Am. J. Physiol. Heart Circ. Physiol. 285, H2039–H2044 (2003).
Wang, X., Zheng, W., Christensen, L. P. & Tomanek, R. J. DITPA stimulates bFGF, VEGF, angiopoietin, and Tie-2 and facilitates coronary arteriolar growth. Am. J. Physiol. Heart Circ. Physiol. 284, H613–H618 (2003).
Laham, R. J. et al. Intrapericardial delivery of fibroblast growth factor-2 induces neovascularization in a porcine model of chronic myocardial ischemia. J. Pharmacol. Exp. Ther. 292, 795–802 (2000).
Reinhardt, C. P., Dalhberg, S., Tries, M. A., Marcel, R. & Leppo, J. A. Stable labeled microspheres to measure perfusion: validation of a neutron activation assay technique. Am. J. Physiol. Heart Circ. Physiol. 280, H108–H116 (2001).
Ruel, M. et al. Inhibition of the cardiac angiogenic response to surgical FGF-2 therapy in a swine endothelial dysfunction model. Circulation 108, II335–II340 (2003).
Pearlman, J. D., Laham, R. J. & Simons, M. Coronary angiogenesis: detection in vivo with MR imaging sensitive to collateral neocirculation—preliminary study in pigs. Radiology 214, 801–807 (2000).
Yang, M. et al. Dual-color fluorescence imaging distinguishes tumor cells from induced host angiogenic vessels and stromal cells. Proc. Natl Acad. Sci. USA 100, 14259–14262 (2003).
Rubart, M. Two-photon microscopy of cells and tissue. Circ. Res. 95, 1154–1166 (2004).
Rubart, M. et al. Physiological coupling of donor and host cardiomyocytes after cellular transplantation. Circ. Res. 92, 1217–1224 (2003).
Rubart, M., Soonpaa, M. H., Nakajima, H. & Field, L. J. Spontaneous and evoked intracellular calcium transients in donor-derived myocytes following intracardiac myoblast transplantation. J. Clin. Invest. 114, 775–783 (2004).
Levene, M. J., Dombeck, D. A., Kasischke, K. A., Molloy, R. P. & Webb, W. W. In vivo multiphoton microscopy of deep brain tissue. J. Neurophysiol. 91, 1908–1912 (2004).
Peters, N. S., Coromilas, J., Severs, N. J. & Wit, A. L. Disturbed connexin43 gap junction distribution correlates with the location of reentrant circuits in the epicardial border zone of healing canine infarcts that cause ventricular tachycardia. Circulation 95, 988–996 (1997).
Kleinfeld, D., Mitra, P. P., Helmchen, F. & Denk, W. Fluctuations and stimulus-induced changes in blood flow observed in individual capillaries in layers 2 through 4 of rat neocortex. Proc. Natl Acad. Sci. USA 95, 15741–15746 (1998).
Freedman, S. B., Dunn, R. F., Bernstein, L., Morris, J. & Kelly, D. T. Influence of coronary collateral blood flow on the development of exertional ischemia and Q wave infarction in patients with severe single-vessel disease. Circulation 71, 681–686 (1985).
Pearlman, J. D., Laham, R. J., Post, M., Leiner, T. & Simons, M. Medical imaging techniques in the evaluation of strategies for therapeutic angiogenesis. Curr. Pharm. Des. 8, 1467–1496 (2002).
Simons, M. et al. Clinical trials in coronary angiogenesis: issues, problems, consensus: An expert panel summary. Circulation 102, E73–E86 (2000).
Muzik, O. et al. Assessment of diagnostic performance of quantitative flow measurements in normal subjects and patients with angiographically documented coronary artery disease by means of nitrogen-13 ammonia and positron emission tomography. J. Am. Coll. Cardiol. 31, 534–540 (1998).
Anagnostopoulos, C. et al. Quantitative relationship between coronary vasodilator reserve assessed by 82Rb PET imaging and coronary artery stenosis severity. Eur. J. Nucl. Med. Mol. Imaging 35, 1593–1601 (2008).
Meoli, D. F. et al. Noninvasive imaging of myocardial angiogenesis following experimental myocardial infarction. J. Clin. Invest. 113, 1684–1691 (2004).
Johnson, L. L. et al. Radiolabeled arginine-glycine-aspartic acid peptides to image angiogenesis in swine model of hibernating myocardium. JACC Cardiovasc. Imaging 1, 511–514 (2008).
Wei, L. et al. 64Cu-labeled CB-TE2A and diamsar-conjugated RGD peptide analogs for targeting angiogenesis: comparison of their biological activity. Nucl. Med. Biol. 36, 277–285 (2009).
Wei, K. et al. Noninvasive quantification of coronary blood flow reserve in humans using myocardial contrast echocardiography. Circulation 103, 2560–2565 (2001).
Jayaweera, A. R. et al. Role of capillaries in determining CBF reserve: new insights using myocardial contrast echocardiography. Am. J. Physiol. 277, H2363–H2372 (1999).
Villanueva, F. S. et al. Myocardial contrast echocardiography can be used to assess the microvascular response to vascular endothelial growth factor-121. Circulation 105, 759–765 (2002).
Kaufmann, B. A. & Lindner, J. R. Molecular imaging with targeted contrast ultrasound. Curr. Opin. Biotechnol. 18, 11–16 (2007).
Gibson, C. M. et al. Angiographic methods to assess human coronary angiogenesis. Am. Heart J. 137, 169–179 (1999).
Asahara, T. et al. Synergistic effect of vascular endothelial growth factor and basic fibroblast growth factor on angiogenesis in vivo. Circulation 92, II365–II371 (1995).
Yanagisawa-Miwa, A. et al. Salvage of infarcted myocardium by angiogenic action of basic fibroblast growth factor. Science 257, 1401–1403 (1992).
Rentrop, K. P., Cohen, M., Blanke, H. & Phillips, R. A. Changes in collateral channel filling immediately after controlled coronary artery occlusion by an angioplasty balloon in human subjects. J. Am. Coll. Cardiol. 5, 587–592 (1985).
Chittenden, T. W. et al. Transcriptional profiling in coronary artery disease: indications for novel markers of coronary collateralization. Circulation 114, 1811–1820 (2006).
Kyriakides, Z. S., Antoniadis, A., Kolettis, T. M. & Kremastinos, D. T. Coronary flow reserve in the contralateral artery increases after successful coronary angioplasty in patients with spontaneously visible collateral vessels. Heart 80, 493–498 (1998).
Werner, G. S. & Figulla, H. R. Direct assessment of coronary steal and associated changes of collateral hemodynamics in chronic total coronary occlusions. Circulation 106, 435–440 (2002).
De Bruyne, B. et al. Fractional flow reserve in patients with prior myocardial infarction. Circulation 104, 157–162 (2001).
Montisci, R. et al. Non-invasive coronary flow reserve is correlated with microvascular integrity and myocardial viability after primary angioplasty in acute myocardial infarction. Heart 92, 1113–1118 (2006).
Wikstrom, J., Gronros, J., Bergstrom, G. & Gan, L. M. Functional and morphologic imaging of coronary atherosclerosis in living mice using high-resolution color Doppler echocardiography and ultrasound biomicroscopy. J. Am. Coll. Cardiol. 46, 720–727 (2005).
Lee, T. Y., Purdie, T. G. & Stewart, E. CT imaging of angiogenesis. Q. J. Nucl. Med. 47, 171–187 (2003).
Miles, K. A. et al. Application of CT in the investigation of angiogenesis in oncology. Acad. Radiol. 7, 840–850 (2000).
Rybicki, F. J. et al. Initial evaluation of coronary images from 320-detector row computed tomography. Int. J. Cardiovasc. Imaging 24, 535–546 (2008).
Choi, S, I, et al. Recent developments in wide-detector cardiac computed tomography. Int. J. Cardiovasc. Imaging 25, 23–29 (2009).
George, R. T. et al. Quantification of myocardial perfusion using dynamic 64-detector computed tomography. Invest. Radiol. 42, 815–822 (2007).
Kitagawa, T. et al. Characterization of noncalcified coronary plaques and identification of culprit lesions in patients with acute coronary syndrome by 64-slice computed tomography. J. Am. Coll. Cardiol. 2, 153–160 (2009).
Pearlman, J. D., Laham, R. J., Simons, M., Gladstone, S. & Raptopoulos, V. Extent of myocardial collateralization: determination with three-dimensional elastic-subtraction spiral CT. Acad. Radiol. 4, 680–686 (1997).
Bremer, C. et al. Steady-state blood volume measurements in experimental tumors with different angiogenic burdens a study in mice. Radiology 226, 214–220 (2003).
Turetschek, K. et al. MRI monitoring of tumor response following angiogenesis inhibition in an experimental human breast cancer model. Eur. J. Nucl. Med. Mol. Imaging 30, 448–455 (2003).
Bhujwalla, Z. M., Artemov, D., Natarajan, K., Ackerstaff, E. & Solaiyappan, M. Vascular differences detected by MRI for metastatic versus nonmetastatic breast and prostate cancer xenografts. Neoplasia 3, 143–153 (2001).
Pearlman, J. D., Gertz, Z. M., Wu, Y., Simons, M. & Post, M. J. Serial motion assessment by reference tracking (SMART): application to detection of local functional impact of chronic myocardial ischemia. J. Comput. Assist. Tomogr. 25, 558–562 (2001).
Pearlman, J. D. et al. Magnetic resonance mapping demonstrates benefits of VEGF-induced myocardial angiogenesis. Nat. Med. 1, 1085–1089 (1995).
Vallee, J. P. et al. Quantification of myocardial perfusion with FAST sequence and Gd bolus in patients with normal cardiac function. J. Magn. Reson. Imaging 9, 197–203 (1999).
Bassingthwaighte, J. B. Physiology and theory of tracer washout techniques for the estimation of myocardial blood flow: flow estimation from tracer washout. Prog. Cardiovasc. Dis. 20, 165–189 (1977).
Beard, D. A. & Bassingthwaighte, J. B. The fractal nature of myocardial blood flow emerges from a whole-organ model of arterial network. J. Vasc. Res. 37, 282–296 (2000).
Laham, R. J. et al. Intracoronary basic fibroblast growth factor (FGF-2) in patients with severe ischemic heart disease: results of a phase I open-label dose escalation study. J. Am. Coll. Cardiol. 36, 2132–2139 (2000).
Laham, R. J., Simons, M., Pearlman, J. D., Ho, K. K. & Baim, D. S. Magnetic resonance imaging demonstrates improved regional systolic wall motion and thickening and myocardial perfusion of myocardial territories treated by laser myocardial revascularization. J. Am. Coll. Cardiol. 39, 1–8 (2002).
Schachinger, V. et al. Transplantation of progenitor cells and regeneration enhancement in acute myocardial infarction: final one-year results of the TOPCARE-AMI Trial. J. Am. Coll. Cardiol. 44, 1690–1699 (2004).
Wollert, K. C. & Drexler, H. Clinical applications of stem cells for the heart. Circ. Res. 96, 151–163 (2005).
Lin, P. C. Optical imaging and tumor angiogenesis. J. Cell Biochem. 90, 484–491 (2003).
Neeman, M. & Dafni, H. Structural, functional, and molecular MR imaging of the microvasculature. Annu. Rev. Biomed. Eng. 5, 29–56 (2003).
Jayson, G. C. et al. Molecular imaging and biological evaluation of HuMV833 anti-VEGF antibody: implications for trial design of antiangiogenic antibodies. J. Natl Cancer Inst. 94, 1484–1493 (2002).
Weissleder, R., Bogdanov, A. Jr, Tung, C. H. & Weinmann, H. J. Size optimization of synthetic graft copolymers for in vivo angiogenesis imaging. Bioconjugate Chem. 12, 213–219 (2001).
Kang, H. W., Josephson, L., Petrovsky, A., Weissleder, R. & Bogdanov, A. Jr. Magnetic resonance imaging of inducible E-selectin expression in human endothelial cell culture. Bioconjugate Chem. 13, 122–127 (2002).
Winter, P. M. et al. Molecular imaging of angiogenesis in nascent Vx-2 rabbit tumors using a novel α(nu)β3-targeted nanoparticle and 1.5 tesla magnetic resonance imaging. Cancer Res. 63, 5838–5843 (2003).
Lazarous, D. F. et al. Effects of chronic systemic administration of basic fibroblast growth factor on collateral development in the canine heart. Circulation 91, 145–153 (1995).
Giordano, F. J. et al. Intracoronary gene transfer of fibroblast growth factor-5 increases blood flow and contractile function in an ischemic region of the heart. Nat. Med. 2, 534–539 (1996).
Unger, E. F. et al. Basic fibroblast growth factor enhances myocardial collateral flow in a canine model. Am. J. Physiol. 266, H1588–H1595 (1994).
Hendel, R. C. et al. Effect of intracoronary recombinant human vascular endothelial growth factor on myocardial perfusion: evidence for a dose-dependent effect. Circulation 101, 118–121 (2000).
Henry, T. D. & Satran, D. in Coronary Artery Disease: New Approaches without Traditional Revascularization (eds Barsness, G. W. & Holmes, D. R.) (Springer, New York City, 2007).
Udelson, J. E. et al. Therapeutic angiogenesis with recombinant fibroblast growth factor-2 improves stress and rest myocardial perfusion abnormalities in patients with severe symptomatic chronic coronary artery disease. Circulation 102, 1605–1610 (2000).
Grines, C. L. et al. A randomized, double-blind, placebo-controlled trial of Ad5FGF-4 gene therapy and its effect on myocardial perfusion in patients with stable angina. J. Am. Coll. Cardiol. 42, 1339–1347 (2003).
Pijls, N. H., Bos, H. S., Uijen, G. J. & Van der Werf, T. Is nonionic isotonic iohexol the contrast agent of choice for quantitative myocardial videodensitometry? Int. J. Card. Imaging 3, 117–126 (1988).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Table 1
Spatial resolution of different imaging modalities (DOC 40 kb)
Supplementary Figure 1
Diagram of the multiple factors leading to production of new vessels (DOC 174 kb)
Rights and permissions
About this article
Cite this article
Wykrzykowska, J., Henry, T., Lesser, J. et al. Imaging myocardial angiogenesis. Nat Rev Cardiol 6, 648–658 (2009). https://doi.org/10.1038/nrcardio.2009.157
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrcardio.2009.157
This article is cited by
-
Effect of Korean red ginseng extract on flap survival and angiogenesis in rat model
European Journal of Plastic Surgery (2015)
-
Angiogenic therapy for cardiac repair based on protein delivery systems
Heart Failure Reviews (2012)