Abstract
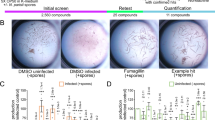
Traditional antimicrobial screens focus on compounds that block the growth of microbial organisms. A new Caenorhabditis elegans-based bioassay can be used for the identification of antifungal compounds that are effective against Candida albicans. According to the protocol, adult nematodes are infected with C. albicans and moved to 96-well plates containing the tested compounds. In the presence of compounds with no antifungal activity, the fungus kills the worms and develops filaments that penetrate through the cuticle. In contrast to traditional screening methods and mammalian models, this facile, time-efficient and less costly assay allows the study of Candida cells in nonplanktonic form and may allow the concurrent evaluation of toxicity and antifungal activity and identify compounds that target virulence factors or modify host immune response. The screening assay takes about 5–6 d depending on the experimental design.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Spanakis, E.K., Aperis, G. & Mylonakis, E. New agents for the treatment of fungal infections: clinical efficacy and gaps in coverage. Clin. Infect. Dis. 43, 1060–1068 (2006).
Slavin, M. et al. Burden of hospitalization of patients with Candida and Aspergillus infections in Australia. Int. J. Infect. Dis. 8, 111–120 (2004).
Berman, J. & Sudbery, P.E. Candida albicans: a molecular revolution built on lessons from budding yeast. Nat. Rev. Genet. 3, 918–930 (2002).
Rentz, A.M., Halpern, M.T. & Bowden, R. The impact of candidemia on length of hospital stay, outcome, and overall cost of illness. Clin. Infect. Dis. 27, 781–788 (1998).
Akins, R.A. An update on antifungal targets and mechanisms of resistance in Candida albicans . Med. Mycol. 43, 285–318 (2005).
Anderson, J.B. Evolution of antifungal-drug resistance: mechanisms and pathogen fitness. Nat. Rev. Microbiol. 3, 547–556 (2005).
Alegado, R.A., Campbell, M.C., Chen, W.C., Slutz, S.S. & Tan, M.W. Characterization of mediators of microbial virulence and innate immunity using the Caenorhabditis elegans host–pathogen model. Cell Microbiol. 5, 435–444 (2003).
Mylonakis, E., Casadevall, A. & Ausubel, F.M. Exploiting amoeboid and non-vertebrate animal model systems to study the virulence of human pathogenic fungi. PLoS Pathog. 3, e101 (2007).
Sifri, C.D., Begun, J. & Ausubel, F.M. The worm has turned—microbial virulence modeled in Caenorhabditis elegans . Trends Microbiol. 13, 119–127 (2005).
Breger, J. et al. Antifungal chemical compounds identified using a C. elegans pathogenicity assay. PLoS Pathog. 3, e18 (2007).
Kumamoto, C.A. & Vinces, M.D. Contributions of hyphae and hypha-co-regulated genes to Candida albicans virulence. Cell Microbiol. 7, 1546–1554 (2005).
Lo, H.J. et al. Nonfilamentous C. albicans mutants are avirulent. Cell 90, 939–949 (1997).
Douglas, L.J. Candida biofilms and their role in infection. Trends Microbiol. 11, 30–36 (2003).
Aperis, G. et al. Galleria mellonella as a model host to study infection by the Francisella tularensis live vaccine strain. Microbes Infect 9, 729–734 (2007).
Scully, L.R. & Bidochka, M.J. Developing insect models for the study of current and emerging human pathogens. FEMS Microbiol. Lett. 263, 1–9 (2006).
Moy, T.I. et al. Identification of novel antimicrobials using a live-animal infection model. Proc. Natl. Acad. Sci. USA 103, 10414–10419 (2006).
Kaletta, T. & Hengartner, M.O. Finding function in novel targets: C. elegans as a model organism. Nat. Rev. Drug Discov. 5, 387–398 (2006).
Artal-Sanz, M., de Jong, L. & Tavernarakis, N. Caenorhabditis elegans: a versatile platform for drug discovery. Biotechnol. J. 1, 1405–1418 (2006).
Bhavsar, A.P. & Brown, E.D. The worm turns for antimicrobial discovery. Nat. Biotechnol. 24, 1098–1100 (2006).
Kurz, C.L. & Ewbank, J.J. Infection in a dish: high-throughput analyses of bacterial pathogenesis. Curr. Opin. Microbiol. 10, 10–16 (2007).
Darby, C. Interactions with microbial pathogens. WormBook (http://www.wormbook.org), Sept. 6, 1–15 (2005).
Mylonakis, E. & Aballay, A. Worms and flies as genetically tractable animal models to study host-pathogen interactions. Infect Immun. 73, 3833–3841 (2005).
Sochova, I., Hofman, J. & Holoubek, I. Using nematodes in soil ecotoxicology. Environ. Int. 32, 374–383 (2006).
Beanan, M.J. & Strome, S. Characterization of a germ-line proliferation mutation in C. elegans . Development 116, 755–766 (1992).
Kim, D.H. et al. A conserved p38 MAP kinase pathway in Caenorhabditis elegans innate immunity. Science 297, 623–626 (2002).
Richard, M.L., Nobile, C.J., Bruno, V.M. & Mitchell, A.P. Candida albicans biofilm-defective mutants. Eukaryot Cell 4, 1493–1502 (2005).
Carpenter, A.E. et al. CellProfiler: image analysis software for identifying and quantifying cell phenotypes. Genome Biol. 7, R100 (2006).
Acknowledgements
Support was provided by NIH R01 award AI075286 (E.M.). We are grateful to Melanie de Silva of Broad Institute of Massachusetts Institute of Technology, USA, for technical assistance.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Tampakakis, E., Okoli, I. & Mylonakis, E. A C. elegans-based, whole animal, in vivo screen for the identification of antifungal compounds. Nat Protoc 3, 1925–1931 (2008). https://doi.org/10.1038/nprot.2008.193
Published:
Issue Date:
DOI: https://doi.org/10.1038/nprot.2008.193
This article is cited by
-
Eicosapentaenoic acid influences the pathogenesis of Candida albicans in Caenorhabditis elegans via inhibition of hyphal formation and stimulation of the host immune response
Medical Microbiology and Immunology (2023)
-
Polymeric micelles with anti-virulence activity against Candida albicans in a single- and dual-species biofilm
Drug Delivery and Translational Research (2021)
-
Genetically Compromising Phospholipid Metabolism Limits Candida albicans’ Virulence
Mycopathologia (2019)
-
A short D-enantiomeric antimicrobial peptide with potent immunomodulatory and antibiofilm activity against multidrug-resistant Pseudomonas aeruginosa and Acinetobacter baumannii
Scientific Reports (2017)
-
A high-throughput assay of yeast cell lysis for drug discovery and genetic analysis
Nature Protocols (2010)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.