Abstract
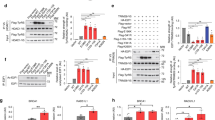
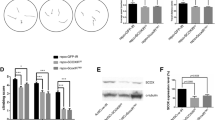
We identified a mutation in Aats-gly (also known as gars or glycyl-tRNA synthetase), the Drosophila melanogaster ortholog of the human GARS gene that is associated with Charcot-Marie-Tooth neuropathy type 2D (CMT2D), from a mosaic genetic screen. Loss of gars in Drosophila neurons preferentially affects the elaboration and stability of terminal arborization of axons and dendrites. The human and Drosophila genes each encode both a cytoplasmic and a mitochondrial isoform. Using additional mutants that selectively disrupt cytoplasmic or mitochondrial protein translation, we found that cytoplasmic protein translation is required for terminal arborization of both dendrites and axons during development. In contrast, disruption of mitochondrial protein translation preferentially affects the maintenance of dendritic arborization in adults. We also provide evidence that human GARS shows equivalent functions in Drosophila, and that CMT2D causal mutations show loss-of-function properties. Our study highlights different demands of protein translation for the development and maintenance of axons and dendrites.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout








Similar content being viewed by others
References
Steward, O. & Schuman, E.M. Compartmentalized synthesis and degradation of proteins in neurons. Neuron 40, 347–359 (2003).
Martin, K.C. Local protein synthesis during axon guidance and synaptic plasticity. Curr. Opin. Neurobiol. 14, 305–310 (2004).
Piper, M. & Holt, C. RNA translation in axons. Annu. Rev. Cell Dev. Biol. 20, 505–523 (2004).
Bailey, C.H., Bartsch, D. & Kandel, E.R. Toward a molecular definition of long-term memory storage. Proc. Natl. Acad. Sci. USA 93, 13445–13452 (1996).
Horton, A.C. et al. Polarized secretory trafficking directs cargo for asymmetric dendrite growth and morphogenesis. Neuron 48, 757–771 (2005).
Grossman, A.W., Aldridge, G.M., Weiler, I.J. & Greenough, W.T. Local protein synthesis and spine morphogenesis: Fragile X syndrome and beyond. J. Neurosci. 26, 7151–7155 (2006).
Lee, J.W. et al. Editing-defective tRNA synthetase causes protein misfolding and neurodegeneration. Nature 443, 50–55 (2006).
Li, Z., Okamoto, K., Hayashi, Y. & Sheng, M. The importance of dendritic mitochondria in the morphogenesis and plasticity of spines and synapses. Cell 119, 873–887 (2004).
Stowers, R.S., Megeath, L.J., Gorska-Andrzejak, J., Meinertzhagen, I.A. & Schwarz, T.L. Axonal transport of mitochondria to synapses depends on milton, a novel Drosophila protein. Neuron 36, 1063–1077 (2002).
Verstreken, P. et al. Synaptic mitochondria are critical for mobilization of reserve pool vesicles at Drosophila neuromuscular junctions. Neuron 47, 365–378 (2005).
Guo, X. et al. The GTPase dMiro is required for axonal transport of mitochondria to Drosophila synapses. Neuron 47, 379–393 (2005).
Chan, D.C. Mitochondria: dynamic organelles in disease, aging and development. Cell 125, 1241–1252 (2006).
Kujoth, G.C. et al. Mitochondrial DNA mutations, oxidative stress and apoptosis in mammalian aging. Science 309, 481–484 (2005).
Antonellis, A. et al. Glycyl tRNA synthetase mutations in Charcot-Marie-Tooth disease type 2D and distal spinal muscular atrophy type V. Am. J. Hum. Genet. 72, 1293–1299 (2003).
Shy, M.E. Charcot-Marie-Tooth disease: an update. Curr. Opin. Neurol. 17, 579–585 (2004).
Jefferis, G.S., Marin, E.C., Stocker, R.F. & Luo, L. Target neuron prespecification in the olfactory map of Drosophila. Nature 414, 204–208 (2001).
Marin, E.C., Jefferis, G.S., Komiyama, T., Zhu, H. & Luo, L. Representation of the glomerular olfactory map in the Drosophila brain. Cell 109, 243–255 (2002).
Wong, A.M., Wang, J.W. & Axel, R. Spatial representation of the glomerular map in the Drosophila protocerebrum. Cell 109, 229–241 (2002).
Jefferis, G.S. et al. Comprehensive maps of Drosophila higher olfactory centers: spatially segregated fruit and pheromone representation. Cell 128, 1187–1203 (2007).
Lee, T. & Luo, L. Mosaic analysis with a repressible cell marker for studies of gene function in neuronal morphogenesis. Neuron 22, 451–461 (1999).
Robinson, I.M., Ranjan, R. & Schwarz, T.L. Synaptotagmins I and IV promote transmitter release independently of Ca2+ binding in the C(2)A domain. Nature 418, 336–340 (2002).
Ng, M. et al. Transmission of olfactory information between three populations of neurons in the antennal lobe of the fly. Neuron 36, 463–474 (2002).
Berger, J. et al. Genetic mapping with SNP markers in Drosophila. Nat. Genet. 29, 475–481 (2001).
Zhai, R.G. et al. Mapping Drosophila mutations with molecularly defined P element insertions. Proc. Natl. Acad. Sci. USA 100, 10860–10865 (2003).
Shiba, K., Schimmel, P., Motegi, H. & Noda, T. Human glycyl-tRNA synthetase. Wide divergence of primary structure from bacterial counterpart and species-specific aminoacylation. J. Biol. Chem. 269, 30049–30055 (1994).
Ibba, M. & Soll, D. Aminoacyl-tRNA synthesis. Annu. Rev. Biochem. 69, 617–650 (2000).
Jefferis, G.S. et al. Developmental origin of wiring specificity in the olfactory system of Drosophila. Development 131, 117–130 (2004).
Turner, R.J., Lovato, M. & Schimmel, P. One of two genes encoding glycyl-tRNA synthetase in Saccharomyces cerevisiae provides mitochondrial and cytoplasmic functions. J. Biol. Chem. 275, 27681–27688 (2000).
Chang, K.J. & Wang, C.C. Translation initiation from a naturally occurring non-AUG codon in Saccharomyces cerevisiae. J. Biol. Chem. 279, 13778–13785 (2004).
Seshaiah, P. & Andrew, D.J. WRS-85D: a tryptophanyl-tRNA synthetase expressed to high levels in the developing Drosophila salivary gland. Mol. Biol. Cell 10, 1595–1608 (1999).
Royden, C.S., Pirrotta, V. & Jan, L.Y. The tko locus, site of a behavioral mutation in D. melanogaster, codes for a protein homologous to prokaryotic ribosomal protein S12. Cell 51, 165–173 (1987).
Toivonen, J.M. et al. Technical knockout, a Drosophila model of mitochondrial deafness. Genetics 159, 241–254 (2001).
Heisenberg, M. Mushroom body memoir: from maps to models. Nat. Rev. Neurosci. 4, 266–275 (2003).
Lee, T., Lee, A. & Luo, L. Development of the Drosophila mushroom bodies: sequential generation of three distinct types of neurons from a neuroblast. Development 126, 4065–4076 (1999).
Watts, R.J., Hoopfer, E.D. & Luo, L. Axon pruning during Drosophila metamorphosis: evidence for local degeneration and requirement of the ubiquitin-proteasome system. Neuron 38, 871–885 (2003).
Sivakumar, K. et al. Phenotypic spectrum of disorders associated with glycyl-tRNA synthetase mutations. Brain 128, 2304–2314 (2005).
Dubourg, O. et al. The G526R glycyl-tRNA synthetase gene mutation in distal hereditary motor neuropathy type V. Neurology 66, 1721–1726 (2006).
Del Bo, R. et al. Coexistence of CMT-2D and distal SMA-V phenotypes in an Italian family with a GARS gene mutation. Neurology 66, 752–754 (2006).
Bonnefond, L. et al. Toward the full set of human mitochondrial aminoacyl-tRNA synthetases: characterization of AspRS and TyrRS. Biochemistry 44, 4805–4816 (2005).
Mudge, S.J. et al. Complex organisation of the 5′ end of the human glycine tRNA synthetase gene. Gene 209, 45–50 (1998).
Lee, S.W., Cho, B.H., Park, S.G. & Kim, S. Aminoacyl-tRNA synthetase complexes: beyond translation. J. Cell Sci. 117, 3725–3734 (2004).
Goldberg, G.S., Lampe, P.D. & Nicholson, B.J. Selective transfer of endogenous metabolites through gap junctions composed of different connexins. Nat. Cell Biol. 1, 457–459 (1999).
Gorska-Andrzejak, J. et al. Mitochondria are redistributed in Drosophila photoreceptors lacking milton, a kinesin-associated protein. J. Comp. Neurol. 463, 372–388 (2003).
Overly, C.C., Rieff, H.I. & Hollenbeck, P.J. Organelle motility and metabolism in axons versus dendrites of cultured hippocampal neurons. J. Cell Sci. 109, 971–980 (1996).
Seburn, K.L., Nangle, L.A., Cox, G.A., Schimmel, P. & Burgess, R.W. An active dominant mutation of glycyl-tRNA synthetase causes neuropathy in a Charcot-Marie-Tooth 2D mouse model. Neuron 51, 715–726 (2006).
Antonellis, A. et al. Functional analyses of glycyl-tRNA synthetase mutations suggest a key role for tRNA-charging enzymes in peripheral axons. J. Neurosci. 26, 10397–10406 (2006).
Jordanova, A. et al. Disrupted function and axonal distribution of mutant tyrosyl-tRNA synthetase in dominant intermediate Charcot-Marie-Tooth neuropathy. Nat. Genet. 38, 197–202 (2006).
Bilen, J. & Bonini, N.M. Drosophila as a model for human neurodegenerative disease. Annu. Rev. Genet. 39, 153–171 (2005).
Acknowledgements
We thank R.J. Watts, E.D. Hoopfer and O. Schuldiner for contributions to mosaic genetic screening; H.T. Jocobs, D.J. Andrew, the Bloomington Drosophila Stock Center and the Kyoto Drosophila Genetic Resource Center for fly stocks; D. Berdnik, T. Komiyama, O. Schuldiner, B. Tasic and H. Zhu for comments on the manuscripts, and M. Miura for supporting T.C. to complete this work. T.C. was a recipient of a Overseas Research Fellowship from Japan Science and Technology Agency and a Postdoctoral Fellowship for Research Abroad from Japan Society for the Promotion of Science. This work was supported by US National Institutes of Health grant R01-DC005982 to L.L. and by the Sumitomo Foundation and a grant from Japan Society for the Promotion of Science to T.C. L.L. is an investigator of the Howard Hughes Medical Institute.
Author information
Authors and Affiliations
Contributions
T.C. designed the study with the help of L.L. T.C. conducted the experimental work and analyzed the data, D.L. assisted with the forward genetic screen, and T.C. and L.L. wrote the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Fig. 1
Lack of axon terminal arborization in team−/− projection neurons. (PDF 794 kb)
Supplementary Fig. 2
MARCM rescue experiments of garsEX34/EX34 projection neuron clones with either cytoplasmic or mitochondrial GARS. (PDF 3795 kb)
Supplementary Fig. 3
Generation of Df(3L)mito. (PDF 120 kb)
Rights and permissions
About this article
Cite this article
Chihara, T., Luginbuhl, D. & Luo, L. Cytoplasmic and mitochondrial protein translation in axonal and dendritic terminal arborization. Nat Neurosci 10, 828–837 (2007). https://doi.org/10.1038/nn1910
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nn1910
This article is cited by
-
The psychiatric risk gene BRD1 modulates mitochondrial bioenergetics by transcriptional regulation
Translational Psychiatry (2022)
-
Glutamate Receptors Mediate Changes to Dendritic Mitochondria in Neurons Grown on Stiff Substrates
Annals of Biomedical Engineering (2022)
-
The role of suboptimal mitochondrial function in vulnerability to post‐traumatic stress disorder
Journal of Inherited Metabolic Disease (2018)
-
Pathogenic variants in glutamyl-tRNAGln amidotransferase subunits cause a lethal mitochondrial cardiomyopathy disorder
Nature Communications (2018)
-
Chronic Cerebral Hypoperfusion Induced Synaptic Proteome Changes in the rat Cerebral Cortex
Molecular Neurobiology (2017)