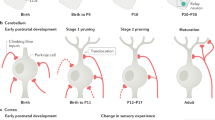
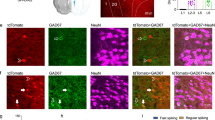
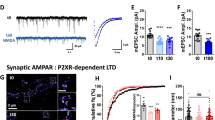
Abstract
In long-term depression (LTD) at synapses in the adult brain, synaptic strength is reduced in an experience-dependent manner. LTD thus provides a cellular mechanism for information storage in some forms of learning. A similar activity-dependent reduction in synaptic strength also occurs in the developing brain and there provides an essential step in synaptic pruning and the postnatal development of neural circuits. Here we review evidence suggesting that LTD and synaptic pruning share components of their underlying molecular machinery and may thus represent two developmental stages of the same type of synaptic modulation that serve different, but related, functions in neural circuit plasticity. We also assess the relationship between LTD and synaptic pruning in the context of recent findings of LTD dysregulation in several mouse models of autism spectrum disorder (ASD) and discuss whether LTD deficits can indicate impaired pruning processes that are required for proper brain development.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Huttenlocher, P.R. Morphometric study of human cerebral cortex development. Neuropsychologia 28, 517–527 (1990).
LaMantia, A.S. & Rakic, P. Axon overproduction and elimination in the corpus callosum of the developing rhesus monkey. J. Neurosci. 10, 2156–2175 (1990).
Tapia, J.C. et al. Pervasive synaptic branch removal in the mammalian neuromuscular system at birth. Neuron 74, 816–829 (2012).
Redfern, P.A. Neuromuscular transmission in new-born rats. J. Physiol. (Lond.) 209, 701–709 (1970).
Colman, H., Nabekura, J. & Lichtman, J.W. Alterations in synaptic strength preceding axon withdrawal. Science 275, 356–361 (1997).
Turney, S.G. & Lichtman, J.W. Reversing the outcome of synapse elimination at developing neuromuscular junctions in vivo: evidence for synaptic competition and its mechanism. PLoS Biol. 10, e1001352 (2012).
Malenka, R.C. & Nicoll, R.A. NMDA-receptor-dependent synaptic plasticity: multiple forms and mechanisms. Trends Neurosci. 16, 521–527 (1993).
Singer, W. Development and plasticity of cortical processing architectures. Science 270, 758–764 (1995).
Cooper, L.N. & Bear, M.F. The BCM theory of synapse modification at 30: interaction of theory with experiment. Nat. Rev. Neurosci. 13, 798–810 (2012).
Hebb, D.O. The Organization of Behavior (Wiley, New York, 1949).
Stent, G.S. A physiological mechanism for Hebb's postulate of learning. Proc. Natl. Acad. Sci. USA 70, 997–1001 (1973).
Wiesel, T.N. & Hubel, D.H. Effects of visual deprivation on the morphology and physiology of cells in the cat's lateral geniculate body. J. Neurophysiol. 26, 978–993 (1963).
Bear, M.F. & Rittenhouse, C.D. Molecular basis for induction of ocular dominance plasticity. J. Neurobiol. 41, 83–91 (1999).
Grant, S.G. Synaptopathies: diseases of the synaptome. Curr. Opin. Neurobiol. 22, 522–529 (2012).
Zoghbi, H.Y. & Bear, M.F. Synaptic dysfunction in neurodevelopmental disorders associated with autism and intellectual disabilities. Cold Spring Harb. Perspect. Biol. 4, a009886 (2012).
Courchesne, E., Carper, R. & Akshoomoff, N. Evidence of brain overgrowth in the first year of life in autism. J. Am. Med. Assoc. 290, 337–344 (2003).
Supekar, K. et al. Brain hyperconnectivity in children with autism and its links to social deficits. Cell Rep. 5, 738–747 (2013).
Mostofsky, S.H. et al. Decreased connectivity and cerebellar activity in autism during motor task performance. Brain 132, 2413–2425 (2009).
Di Martino, A. et al. The autism brain imaging data exchange: towards a large-scale evaluation of the intrinsic brain architecture in autism. Mol. Psychiatry 19, 659–667 (2014).
Markram, H., Rinaldi, T. & Markram, K. The intense world syndrome–an alternative hypothesis for autism. Front. Neurosci. 1, 77–96 (2007).
Holtmaat, A. & Svoboda, K. Experience-dependent structural synaptic plasticity in the mammalian brain. Nat. Rev. Neurosci. 10, 647–658 (2009).
Fatemi, S.H. et al. Consensus paper: pathological role of the cerebellum in autism. Cerebellum 11, 777–807 (2012).
Wang, S.S.H., Kloth, A.D. & Badura, A. The cerebellum, sensitive periods, and autism. Neuron 83, 518–532 (2014). This perspective paper argues that autism-related deficits in cerebellar output may prevent the proper development of neocortical circuits, providing an example of developmental diaschisis.
Rittenhouse, C.D., Shouval, H.Z., Paradiso, M.A. & Bear, M.F. Monocular deprivation induces homosynaptic long-term depression in visual cortex. Nature 397, 347–350 (1999).
Hensch, T.K. Critical period plasticity in local cortical circuits. Nat. Rev. Neurosci. 6, 877–888 (2005).
Takesian, A.E. & Hensch, T.K. Balancing plasticity/stability across brain development. Prog. Brain Res. 207, 3–34 (2013).
Bienenstock, E.L., Cooper, L.N. & Munro, P.W. Theory for the development of neuron selectivity: orientation specificity and binocular interaction in visual cortex. J. Neurosci. 2, 32–48 (1982).
Heynen, A.J. et al. Molecular mechanism for loss of visual cortical responsiveness following brief monocular deprivation. Nat. Neurosci. 6, 854–862 (2003).This study demonstrates that the same AMPA receptor subunit phosphorylation and dephosphorylation steps occur in LTD and ocular dominance plasticity in the visual cortex, thus providing strong cortical evidence for an overlap in molecular pathways involved in LTD and synaptic pruning.
Artola, A. & Singer, W. Long-term potentiation and NMDA receptors in rat visual cortex. Nature 330, 649–652 (1987).
Kirkwood, A., Dudek, S.M., Gold, J.T., Aizenman, C.D. & Bear, M.F. Common forms of synaptic plasticity in the hippocampus and neocortex in vitro. Science 260, 1518–1521 (1993).
Kirkwood, A. & Bear, M.F. Homosynaptic long-term depression in the visual cortex. J. Neurosci. 14, 3404–3412 (1994).
Hansel, C., Artola, A. & Singer, W. Relation between dendritic Ca2+ levels and the polarity of synaptic long-term modifications in rat visual cortex neurons. Eur. J. Neurosci. 9, 2309–2322 (1997).
Kleinschmidt, A., Bear, M.F. & Singer, W. Blockade of “NMDA” receptors disrupts experience-dependent plasticity of kitten striate cortex. Science 238, 355–358 (1987).
Bear, M.F., Kleinschmidt, A., Gu, Q.A. & Singer, W. Disruption of experience-dependent synaptic modifications in striate cortex by infusion of an NMDA receptor antagonist. J. Neurosci. 10, 909–925 (1990).
Sidorov, M.S., Kaplan, E.S., Osterweil, E.K., Lindemann, L. & Bear, M.F. Metabotropic glutamate receptor signaling is required for NMDA receptor-dependent ocular dominance plasticity and LTD in visual cortex. Proc. Natl. Acad. Sci. USA 112, 12852–12857 (2015).
Trachtenberg, J.T. et al. Long-term in vivo imaging of experience-dependent synaptic plasticity in adult cortex. Nature 420, 788–794 (2002).
Alvarez, V.A. & Sabatini, B.L. Anatomical and physiological plasticity of dendritic spines. Annu. Rev. Neurosci. 30, 79–97 (2007).
Bhatt, D.H., Zhang, S. & Gan, W.B. Dendritic spine dynamics. Annu. Rev. Physiol. 71, 261–282 (2009).
Zhou, Q., Homma, K.J. & Poo, M.M. Shrinkage of dendritic spines associated with long-term depression of hippocampal synapses. Neuron 44, 749–757 (2004).
Zuo, Y., Yang, G., Kwon, E. & Gan, W.B. Long-term sensory deprivation prevents dendritic spine loss in primary somatosensory cortex. Nature 436, 261–265 (2005).
Ito, M., Yamaguchi, K., Nagao, S. & Yamazaki, T. Long-term depression as a model of cerebellar plasticity. Prog. Brain Res. 210, 1–30 (2014).
Jörntell, H. & Hansel, C. Synaptic memories upside down: bidirectional plasticity at cerebellar parallel fiber-Purkinje cell synapses. Neuron 52, 227–238 (2006).
Hansel, C., Linden, D.J. & D'Angelo, E. Beyond parallel fiber LTD: the diversity of synaptic and non-synaptic plasticity in the cerebellum. Nat. Neurosci. 4, 467–475 (2001).
Gao, Z., van Beugen, B.J. & De Zeeuw, C.I. Distributed synergistic plasticity and cerebellar learning. Nat. Rev. Neurosci. 13, 619–635 (2012).
Aiba, A. et al. Deficient cerebellar long-term depression and impaired motor learning in mGluR1 mutant mice. Cell 79, 377–388 (1994).
Conquet, F. et al. Motor deficit and impairment of synaptic plasticity in mice lacking mGluR1. Nature 372, 237–243 (1994).
Ichise, T. et al. mGluR1 in cerebellar Purkinje cells essential for long-term depression, synapse elimination, and motor coordination. Science 288, 1832–1835 (2000).
Kano, M., Hashimoto, K. & Tabata, T. Type-1 metabotropic glutamate receptor in cerebellar Purkinje cells: a key molecule responsible for long-term depression, endocannabinoid signalling and synapse elimination. Phil. Trans. R. Soc. Lond. B 363, 2173–2186 (2008).
Knöpfel, T. & Grandes, P. Metabotropic glutamate receptors in the cerebellum with a focus on their function in Purkinje cells. Cerebellum 1, 19–26 (2002).
Belmeguenai, A. et al. Alcohol impairs long-term depression at the cerebellar parallel fiber-Purkinje cell synapse. J. Neurophysiol. 100, 3167–3174 (2008).
Hartmann, J. et al. Distinct roles of Gαq and Gα11 for Purkinje cell signaling and motor behavior. J. Neurosci. 24, 5119–5130 (2004).
Hashimoto, K., Miyata, M., Watanabe, M. & Kano, M. Roles of phospholipase Cbeta4 in synapse elimination and plasticity in developing and mature cerebellum. Mol. Neurobiol. 23, 69–82 (2001).
Linden, D.J. & Connor, J.A. Participation of postsynaptic PKC in cerebellar long-term depression in culture. Science 254, 1656–1659 (1991).
Chen, C. et al. Impaired motor coordination correlates with persistent multiple climbing fiber innervation in PKC γ mutant mice. Cell 83, 1233–1242 (1995).
De Zeeuw, C.I. et al. Expression of a protein kinase C inhibitor in Purkinje cells blocks cerebellar LTD and adaptation of the vestibulo-ocular reflex. Neuron 20, 495–508 (1998).
Chung, H.J., Steinberg, J.P., Huganir, R.L. & Linden, D.J. Requirement of AMPA receptor GluR2 phosphorylation for cerebellar long-term depression. Science 300, 1751–1755 (2003).
Leitges, M., Kovac, J., Plomann, M. & Linden, D.J. A unique PDZ ligand in PKCalpha confers induction of cerebellar long-term synaptic depression. Neuron 44, 585–594 (2004).
Baude, A., Molnár, E., Latawiec, D., McIlhinney, R.A. & Somogyi, P. Synaptic and nonsynaptic localization of the GluR1 subunit of the AMPA-type excitatory amino acid receptor in the rat cerebellum. J. Neurosci. 14, 2830–2843 (1994).
Crepel, F., Mariani, J. & Delhaye-Bouchaud, N. Evidence for a multiple innervation of Purkinje cells by climbing fibers in the immature rat cerebellum. J. Neurobiol. 7, 567–578 (1976).
Hashimoto, K. & Kano, M. Synapse elimination in the developing cerebellum. Cell. Mol. Life Sci. 70, 4667–4680 (2013).
Hashimoto, K. & Kano, M. Functional differentiation of multiple climbing fiber inputs during synapse elimination in the developing cerebellum. Neuron 38, 785–796 (2003).
Hashimoto, K. et al. Postsynaptic P/Q-type Ca2+ channel in Purkinje cell mediates synaptic competition and elimination in developing cerebellum. Proc. Natl. Acad. Sci. USA 108, 9987–9992 (2011).
Kawamura, Y. et al. Spike timing-dependent selective strengthening of single climbing fibre inputs to Purkinje cells during cerebellar development. Nat. Commun. 4, 2732 (2013).
Hashimoto, K., Ichikawa, R., Kitamura, K., Watanabe, M. & Kano, M. Translocation of a “winner” climbing fiber to the Purkinje cell dendrite and subsequent elimination of “losers” from the soma in developing cerebellum. Neuron 63, 106–118 (2009). This paper characterizes how the synaptic competition of climbing fiber inputs takes place at the Purkinje cell soma and how the strongest CF input invades the growing dendrite, while weaker inputs are removed.
Carrillo, J., Nishiyama, N. & Nishiyama, H. Dendritic translocation establishes the winner in cerebellar climbing fiber synapse elimination. J. Neurosci. 33, 7641–7653 (2013).
Kakegawa, W. et al. Anterograde C1ql1 signaling is required in order to determine and maintain a single-winner climbing fiber in the mouse cerebellum. Neuron 85, 316–329 (2015).
Hashimoto, K. et al. Influence of parallel fiber-Purkinje cell synapse formation on postnatal development of climbing fiber-Purkinje cell synapses in the cerebellum. Neuroscience 162, 601–611 (2009).
Nakayama, H. et al. GABAergic inhibition regulates developmental synapse elimination in the cerebellum. Neuron 74, 384–396 (2012).
Paolicelli, R.C. et al. Synaptic pruning by microglia is necessary for normal brain development. Science 333, 1456–1458 (2011).
Hoshiko, M., Arnoux, I., Avignone, E., Yamamoto, N. & Audinat, E. Deficiency of the microglial receptor CX3CR1 impairs postnatal functional development of thalamocortical synapses in the barrel cortex. J. Neurosci. 32, 15106–15111 (2012).
Zhan, Y. et al. Deficient neuron-microglia signaling results in impaired functional brain connectivity and social behavior. Nat. Neurosci. 17, 400–406 (2014).
Chu, Y. et al. Enhanced synaptic connectivity and epilepsy in C1q knockout mice. Proc. Natl. Acad. Sci. USA 107, 7975–7980 (2010).
Kano, M. et al. Persistent multiple climbing fiber innervation of cerebellar Purkinje cells in mice lacking mGluR1. Neuron 18, 71–79 (1997).
Levenes, C., Daniel, H., Jaillard, D., Conquet, F. & Crépel, F. Incomplete regression of multiple climbing fibre innervation of cerebellar Purkinje cells in mGLuR1 mutant mice. Neuroreport 8, 571–574 (1997).
Offermanns, S. et al. Impaired motor coordination and persistent multiple climbing fiber innervation of cerebellar Purkinje cells in mice lacking Galphaq. Proc. Natl. Acad. Sci. USA 94, 14089–14094 (1997).
Kano, M. et al. Phospholipase cbeta4 is specifically involved in climbing fiber synapse elimination in the developing cerebellum. Proc. Natl. Acad. Sci. USA 95, 15724–15729 (1998).
Kano, M. et al. Impaired synapse elimination during cerebellar development in PKC γ mutant mice. Cell 83, 1223–1231 (1995).
Ichikawa, R. et al. Territories of heterologous inputs onto Purkinje cell dendrites are segregated by mGluR1-dependent parallel fiber synapse elimination. Proc. Natl. Acad. Sci. USA 113, 2282–2287 (2016). This paper is the first to demonstrate mGluR1-PKC dependent parallel fiber synapse elimination during cerebellar development.
Hansel, C. et al. alphaCaMKII Is essential for cerebellar LTD and motor learning. Neuron 51, 835–843 (2006).
Kawaguchi, S.Y. & Hirano, T. Gating of long-term depression by Ca2+/calmodulin-dependent protein kinase II through enhanced cGMP signalling in cerebellar Purkinje cells. J. Physiol. (Lond.) 591, 1707–1730 (2013).
Belmeguenai, A. & Hansel, C. A role for protein phosphatases 1, 2A, and 2B in cerebellar long-term potentiation. J. Neurosci. 25, 10768–10772 (2005).
Launey, T., Endo, S., Sakai, R., Harano, J. & Ito, M. Protein phosphatase 2A inhibition induces cerebellar long-term depression and declustering of synaptic AMPA receptor. Proc. Natl. Acad. Sci. USA 101, 676–681 (2004).
Shen, K. & Meyer, T. Dynamic control of CaMKII translocation and localization in hippocampal neurons by NMDA receptor stimulation. Science 284, 162–166 (1999).
Misra, C., Brickley, S.G., Wyllie, D.J. & Cull-Candy, S.G. Slow deactivation kinetics of NMDA receptors containing NR1 and NR2D subunits in rat cerebellar Purkinje cells. J. Physiol. (Lond.) 525, 299–305 (2000).
Piochon, C. et al. NMDA receptor contribution to the climbing fiber response in the adult mouse Purkinje cell. J. Neurosci. 27, 10797–10809 (2007).
Renzi, M., Farrant, M. & Cull-Candy, S.G. Climbing-fibre activation of NMDA receptors in Purkinje cells of adult mice. J. Physiol. (Lond.) 585, 91–101 (2007).
Piochon, C., Levenes, C., Ohtsuki, G. & Hansel, C. Purkinje cell NMDA receptors assume a key role in synaptic gain control in the mature cerebellum. J. Neurosci. 30, 15330–15335 (2010).
Rabacchi, S., Bailly, Y., Delhaye-Bouchaud, N. & Mariani, J. Involvement of the N-methyl D-aspartate (NMDA) receptor in synapse elimination during cerebellar development. Science 256, 1823–1825 (1992).
Kakizawa, S., Yamasaki, M., Watanabe, M. & Kano, M. Critical period for activity-dependent synapse elimination in developing cerebellum. J. Neurosci. 20, 4954–4961 (2000).
Shepherd, J.D. & Bear, M.F. New views of Arc, a master regulator of synaptic plasticity. Nat. Neurosci. 14, 279–284 (2011).
Smith-Hicks, C. et al. SRF binding to SRE 6.9 in the Arc promoter is essential for LTD in cultured Purkinje cells. Nat. Neurosci. 13, 1082–1089 (2010).
Mikuni, T. et al. Arc/Arg3.1 is a postsynaptic mediator of activity-dependent synapse elimination in the developing cerebellum. Neuron 78, 1024–1035 (2013).
Kawata, S. et al. Global scaling down of excitatory postsynaptic responses in cerebellar Purkinje cells impairs developmental synapse elimination. Cell Rep. 8, 1119–1129 (2014).
Hansel, C. & Linden, D.J. Long-term depression of the cerebellar climbing fiber–Purkinje neuron synapse. Neuron 26, 473–482 (2000).
Carta, M., Mameli, M. & Valenzuela, C.F. Alcohol potently modulates climbing fiber–>Purkinje neuron synapses: role of metabotropic glutamate receptors. J. Neurosci. 26, 1906–1912 (2006).
Shen, Y., Hansel, C. & Linden, D.J. Glutamate release during LTD at cerebellar climbing fiber-Purkinje cell synapses. Nat. Neurosci. 5, 725–726 (2002).
Miyata, M., Okada, D., Hashimoto, K., Kano, M. & Ito, M. Corticotropin-releasing factor plays a permissive role in cerebellar long-term depression. Neuron 22, 763–775 (1999).
Schmolesky, M.T., De Ruiter, M.M., De Zeeuw, C.I. & Hansel, C. The neuropeptide corticotropin-releasing factor regulates excitatory transmission and plasticity at the climbing fibre-Purkinje cell synapse. Eur. J. Neurosci. 25, 1460–1466 (2007).
Bosman, L.W., Takechi, H., Hartmann, J., Eilers, J. & Konnerth, A. Homosynaptic long-term synaptic potentiation of the “winner” climbing fiber synapse in developing Purkinje cells. J. Neurosci. 28, 798–807 (2008).
Ohtsuki, G. & Hirano, T. Bidirectional plasticity at developing climbing fiber-Purkinje neuron synapses. Eur. J. Neurosci. 28, 2393–2400 (2008).
Arata, A. & Ito, M. Purkinje cell functions in the in vitro cerebellum isolated from neonatal rats in a block with the pons and medulla. Neurosci. Res. 50, 361–367 (2004).
Wang, W. et al. Distinct cerebellar engrams in short-term and long-term motor learning. Proc. Natl. Acad. Sci. USA 111, E188–E193 (2014). This study shows that in the adult cerebellum motor learning can be associated with an elimination of parallel fiber synapses.
Nägerl, U.V., Eberhorn, N., Cambridge, S.B. & Bonhoeffer, T. Bidirectional activity-dependent morphological plasticity in hippocampal neurons. Neuron 44, 759–767 (2004).
Wiegert, J.S. & Oertner, T.G. Long-term depression triggers the selective elimination of weakly integrated synapses. Proc. Natl. Acad. Sci. USA 110, E4510–E4519 (2013).
Lo, Y.J. & Poo, M.M. Activity-dependent synaptic competition in vitro: heterosynaptic suppression of developing synapses. Science 254, 1019–1022 (1991).
Etherington, S.J. & Everett, A.W. Postsynaptic production of nitric oxide implicated in long-term depression at the mature amphibian (Bufo marinus) neuromuscular junction. J. Physiol. (Lond.) 559, 507–517 (2004).
Lo, Y.J., Lin, Y.C., Sanes, D.H. & Poo, M.M. Depression of developing neuromuscular synapses induced by repetitive postsynaptic depolarizations. J. Neurosci. 14, 4694–4704 (1994).
Cash, S., Dan, Y., Poo, M.M. & Zucker, R. Postsynaptic elevation of calcium induces persistent depression of developing neuromuscular synapses. Neuron 16, 745–754 (1996).
Wang, T., Xie, Z. & Lu, B. Nitric oxide mediates activity-dependent synaptic suppression at developing neuromuscular synapses. Nature 374, 262–266 (1995).
Wan, J. & Poo, M. Activity-induced potentiation of developing neuromuscular synapses. Science 285, 1725–1728 (1999).
Balice-Gordon, R.J. & Lichtman, J.W. In vivo observations of pre- and postsynaptic changes during the transition from multiple to single innervation at developing neuromuscular junctions. J. Neurosci. 13, 834–855 (1993).
Balice-Gordon, R.J. & Lichtman, J.W. Long-term synapse loss induced by focal blockade of postsynaptic receptors. Nature 372, 519–524 (1994).
Penzes, P., Cahill, M.E., Jones, K.A., VanLeeuwen, J.E. & Woolfrey, K.M. Dendritic spine pathology in neuropsychiatric disorders. Nat. Neurosci. 14, 285–293 (2011).
Hutsler, J.J. & Zhang, H. Increased dendritic spine densities on cortical projection neurons in autism spectrum disorders. Brain Res. 1309, 83–94 (2010).
Tang, G. et al. Loss of mTOR-dependent macroautophagy causes autistic-like synaptic pruning deficits. Neuron 83, 1131–1143 (2014). Using post-mortem histology, this study demonstrates deficits in spine pruning in autistic individuals. This developmental deficit is linked to hyperactive mTOR signaling.
Verheij, C. et al. Characterization and localization of the FMR-1 gene product associated with fragile X syndrome. Nature 363, 722–724 (1993).
Contractor, A., Klyachko, V.A. & Portera-Cailliau, C. Altered neuronal and circuit excitability in Fragile X syndrome. Neuron 87, 699–715 (2015).
Comery, T.A. et al. Abnormal dendritic spines in fragile X knockout mice: maturation and pruning deficits. Proc. Natl. Acad. Sci. USA 94, 5401–5404 (1997).
Huber, K.M., Gallagher, S.M., Warren, S.T. & Bear, M.F. Altered synaptic plasticity in a mouse model of fragile X mental retardation. Proc. Natl. Acad. Sci. USA 99, 7746–7750 (2002).
Weiler, I.J. & Greenough, W.T. Metabotropic glutamate receptors trigger postsynaptic protein synthesis. Proc. Natl. Acad. Sci. USA 90, 7168–7171 (1993).
Bear, M.F., Huber, K.M. & Warren, S.T. The mGluR theory of fragile X mental retardation. Trends Neurosci. 27, 370–377 (2004).
Santini, E. et al. Exaggerated translation causes synaptic and behavioural aberrations associated with autism. Nature 493, 411–415 (2013). This paper shows that genetic interference with translation control pathways causes enhanced protein synthesis and autism-like synaptic and behavioral phenotypes.
Aguilar-Valles, A. et al. Inhibition of group I metabotropic glutamate receptors reverses autistic-like phenotypes caused by deficiency of the translation repressor eIF4E binding protein 2. J. Neurosci. 35, 11125–11132 (2015).
Barnes, S.A. et al. Convergence of hippocampal pathophysiology in Syngap+/− and Fmr1-/y mice. J. Neurosci. 35, 15073–15081 (2015).
Santini, E. & Klann, E. Reciprocal signaling between translational control pathways and synaptic proteins in autism spectrum disorders. Sci. Signal. 7, re10 (2014).
Abrahams, B.S. & Geschwind, D.H. Advances in autism genetics: on the threshold of a new neurobiology. Nat. Rev. Genet. 9, 341–355 (2008).
Auerbach, B.D., Osterweil, E.K. & Bear, M.F. Mutations causing syndromic autism define an axis of synaptic pathophysiology. Nature 480, 63–68 (2011).Characterizing synaptic plasticity and levels of protein synthesis in Tsc2+/− and Fmr1−/y mice, this paper shows that synaptic dysfunction caused by autism-relevant mutations can fall at opposite ends of the alteration spectrum and can be rescued in mice that carry both mutations.
Dölen, G. et al. Correction of fragile X syndrome in mice. Neuron 56, 955–962 (2007).
Dölen, G. & Bear, M.F. Role for metabotropic glutamate receptor 5 (mGluR5) in the pathogenesis of fragile X syndrome. J. Physiol. (Lond.) 586, 1503–1508 (2008).
Tsai, P.T. et al. Autistic-like behaviour and cerebellar dysfunction in Purkinje cell Tsc1 mutant mice. Nature 488, 647–651 (2012).
Reith, R.M. et al. Loss of Tsc2 in Purkinje cells is associated with autistic-like behavior in a mouse model of tuberous sclerosis complex. Neurobiol. Dis. 51, 93–103 (2013).
Koekkoek, S.K. et al. Deletion of FMR1 in Purkinje cells enhances parallel fiber LTD, enlarges spines, and attenuates cerebellar eyelid conditioning in Fragile X syndrome. Neuron 47, 339–352 (2005).
Baudouin, S.J. et al. Shared synaptic pathophysiology in syndromic and nonsyndromic rodent models of autism. Science 338, 128–132 (2012).
Zhang, B. et al. Neuroligins sculpt cerebellar Purkinje-cell circuits by differential control of distinct classes of synapses. Neuron 87, 781–796 (2015).
Lai, E. et al. An autism-associated neuroligin-3 mutation impairs developmental synapse elimination in the cerebellum. Soc. Neurosci. Abstract 718.03 (2013).
Piochon, C. et al. Cerebellar plasticity and motor learning deficits in a copy-number variation mouse model of autism. Nat. Commun. 5, 5586 (2014). This study demonstrates deficits in cerebellar LTD and synaptic pruning in a mouse model for the human 15q11–13 duplication. Moreover, the paper shows impaired delay eyeblink conditioning, a form of motor learning that requires an intact cerebellum and that is affected in autistic individuals. As LTD is one of several plasticity types that contributes to eyeblink conditioning, this study is a rare example of a synaptic dysfunction that can be linked to an autism-typical behavioral alteration.
Sears, L.L., Finn, P.R. & Steinmetz, J.E. Abnormal classical eye-blink conditioning in autism. J. Autism Dev. Disord. 24, 737–751 (1994).
Oristaglio, J. et al. Children with autism spectrum disorders show abnormal conditioned response timing on delay, but not trace, eyeblink conditioning. Neuroscience 248, 708–718 (2013).
Freeman, J.H. Cerebellar learning mechanisms. Brain Res. 1621, 260–269 (2015).
Bruinsma, C.F. et al. Dissociation of locomotor and cerebellar deficits in a murine Angelman syndrome model. J. Clin. Invest. 125, 4305–4315 (2015).
Kakizawa, S., Yamada, K., Iino, M., Watanabe, M. & Kano, M. Effects of insulin-like growth factor I on climbing fibre synapse elimination during cerebellar development. Eur. J. Neurosci. 17, 545–554 (2003).
Chen, X.R. et al. Mature Purkinje cells require the retinoic acid-related orphan receptor-α (RORα) to maintain climbing fiber mono-innervation and other adult characteristics. J. Neurosci. 33, 9546–9562 (2013).
Sarachana, T. & Hu, V.W. Genome-wide identification of transcriptional targets of RORA reveals direct regulation of multiple genes associated with autism spectrum disorder. Mol. Autism 4, 14 (2013).
Pfeiffer, B.E. et al. Fragile X mental retardation protein is required for synapse elimination by the activity-dependent transcription factor MEF2. Neuron 66, 191–197 (2010).
Sahin, M. & Sur, M. Genes, circuits, and precision therapies for autism and related neurodevelopmental disorders. Science 350, aab3897 (2015).
Lohmann, C. & Kessels, H.W. The developmental stages of synaptic plasticity. J. Physiol. (Lond.) 592, 13–31 (2014).
Bavelier, D., Levi, D.M., Li, R.W., Dan, Y. & Hensch, T.K. Removing brakes on adult brain plasticity: from molecular to behavioral interventions. J. Neurosci. 30, 14964–14971 (2010).
Yuste, R. & Bonhoeffer, T. Morphological changes in dendritic spines associated with long-term synaptic plasticity. Annu. Rev. Neurosci. 24, 1071–1089 (2001).
Holtmaat, A.J. et al. Transient and persistent dendritic spines in the neocortex in vivo. Neuron 45, 279–291 (2005).
Acknowledgements
We thank B. Kasthuri and members of the Hansel laboratory for comments. M.K. is supported by a Grant-in-Aid for Scientific Research (25000015) from JSPS, Japan, and the Brain Mapping by Integrated Neurotechnologies for Disease Studies (Brain/MINDS) from AMED, Japan. C.H. is supported by the Simons Foundation (SFARI 203507 and 311232) and the National Institutes of Health (NS62771).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
About this article
Cite this article
Piochon, C., Kano, M. & Hansel, C. LTD-like molecular pathways in developmental synaptic pruning. Nat Neurosci 19, 1299–1310 (2016). https://doi.org/10.1038/nn.4389
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nn.4389
This article is cited by
-
Effect of Tai Chi on Young Adults with Subthreshold Depression via a Stress–Reward Complex: A Randomized Controlled Trial
Sports Medicine - Open (2023)
-
Dendritic autophagy degrades postsynaptic proteins and is required for long-term synaptic depression in mice
Nature Communications (2022)
-
Role of JAK-STAT and PPAR-Gamma Signalling Modulators in the Prevention of Autism and Neurological Dysfunctions
Molecular Neurobiology (2022)
-
Altered ribosomal function and protein synthesis caused by tau
Acta Neuropathologica Communications (2021)
-
Dopamine D2 receptor regulates cortical synaptic pruning in rodents
Nature Communications (2021)