Abstract
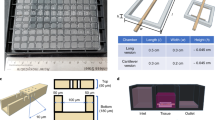
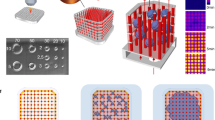
We report the fabrication of a scaffold (hereafter referred to as AngioChip) that supports the assembly of parenchymal cells on a mechanically tunable matrix surrounding a perfusable, branched, three-dimensional microchannel network coated with endothelial cells. The design of AngioChip decouples the material choices for the engineered vessel network and for cell seeding in the parenchyma, enabling extensive remodelling while maintaining an open-vessel lumen. The incorporation of nanopores and micro-holes in the vessel walls enhances permeability, and permits intercellular crosstalk and extravasation of monocytes and endothelial cells on biomolecular stimulation. We also show that vascularized hepatic tissues and cardiac tissues engineered by using AngioChips process clinically relevant drugs delivered through the vasculature, and that millimetre-thick cardiac tissues can be engineered in a scalable manner. Moreover, we demonstrate that AngioChip cardiac tissues implanted with direct surgical anastomosis to the femoral vessels of rat hindlimbs establish immediate blood perfusion.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Huh, D. et al. Reconstituting organ-level lung functions on a chip. Science 328, 1662–1668 (2010).
Kim, H. J., Huh, D., Hamilton, G. & Ingber, D. E. Human gut-on-a-chip inhabited by microbial flora that experiences intestinal peristalsis-like motions and flow. Lab Chip 12, 2165–2174 (2012).
Kolesky, D. B. et al. 3D bioprinting of vascularized, heterogeneous cell-laden tissue constructs. Adv. Mater. 26, 3124–3130 (2014).
Zimmermann, W.-H. et al. Engineered heart tissue grafts improve systolic and diastolic function in infarcted rat hearts. Nature Med. 12, 452–458 (2006).
Nunes, S. S. et al. Biowire: a platform for maturation of human pluripotent stem cell-derived cardiomyocytes. Nature Methods 10, 781–787 (2013).
Yang, X., Pabon, L. & Murry, C. E. Engineering adolescence maturation of human pluripotent stem cell-derived cardiomyocytes. Circ. Res. 114, 511–523 (2014).
Bian, W., Badie, N., Himel IV, H. D. & Bursac, N. Robust T-tubulation and maturation of cardiomyocytes using tissue-engineered epicardial mimetics. Biomaterials 35, 3819–3828 (2014).
Takebe, T. et al. Vascularized and functional human liver from an iPSC-derived organ bud transplant. Nature 499, 481–484 (2013).
Thavandiran, N. et al. Design and formulation of functional pluripotent stem cell-derived cardiac microtissues. Proc. Natl Acad. Sci. USA 110, E4698–E4707 (2013).
Legant, W. R. et al. Microfabricated tissue gauges to measure and manipulate forces from 3D microtissues. Proc. Natl Acad. Sci. USA 106, 10097–10102 (2009).
Bian, W. Engineered skeletal muscle tissue networks with controllable architecture. Biomaterials 30, 1401–1412 (2009).
Kim, S., Lee, H., Chung, M. & Jeon, N. L. Engineering of functional, perfusable 3D microvascular networks on a chip. Lab Chip 13, 1489–1500 (2013).
Zheng, Y. et al. In vitro microvessels for the study of angiogenesis and thrombosis. Proc. Natl Acad. Sci. USA 109, 9342–9347 (2012).
Zhang, B., Peticone, C., Murthy, S. K. & Radisic, M. A standalone perfusion platform for drug testing and target validation in micro-vessel networks. Biomicrofluidics 7, 044125 (2013).
Atala, A., Kasper, F. K. & Mikos, A. G. Engineering complex tissues. Sci. Transl. Med. 4, 160rv112 (2012).
Bae, H. et al. Building vascular networks. Sci. Transl. Med. 4, 160ps123 (2012).
Ye, L., Zimmermann, W.-H., Garry, D. J. & Zhang, J. Patching the heart cardiac repair from within and outside. Circ. Res. 113, 922–932 (2013).
Baranski, J. D. et al. Geometric control of vascular networks to enhance engineered tissue integration and function. Proc. Natl Acad. Sci. USA 110, 7586–7591 (2013).
Sekine, H. et al. In vitro fabrication of functional three-dimensional tissues with perfusable blood vessels. Nature Commun. 4, 1399 (2013).
Shandalov, Y. et al. An engineered muscle flap for reconstruction of large soft tissue defects. Proc. Natl Acad. Sci. USA 111, 6010–6015 (2014).
Miller, J. S. et al. Rapid casting of patterned vascular networks for perfusable engineered three-dimensional tissues. Nature Mater. 11, 768–774 (2012).
Vollert, I. et al. In-vitro perfusion of engineered heart tissue through endothelialized channels. Tissue Eng. 20, 854–863 (2013).
Tang, M. D., Golden, A. P. & Tien, J. Fabrication of collagen gels that contain patterned, micrometer-scale cavities. Adv. Mater. 16, 1345–1348 (2004).
Ye, X. et al. A biodegradable microvessel scaffold as a framework to enable vascular support of engineered tissues. Biomaterials 34, 10007–10015 (2013).
Bettinger, C. J. J. et al. Three-dimensional microfluidic tissue-engineering scaffolds using a flexible biodegradable polymer. Adv. Mater. 18, 165–169 (2006).
Bettinger, C. J. et al. Silk fibroin microfluidic devices. Adv. Mater. 19, 2847–2850 (2007).
Tran, R. T. et al. Synthesis and characterization of a biodegradable elastomer featuring a dual crosslinking mechanism. Soft Matter 6, 2449–2461 (2010).
Yang, J., Webb, A. R. & Ameer, G. A. Novel citric acid-based biodegradable elastomers for tissue engineering. Adv. Mater. 16, 511–516 (2004).
Spiller, K., Freytes, D. & Vunjak-Novakovic, G. Macrophages modulate engineered human tissues for enhanced vascularization and healing. Ann. Biomed. Eng. 43, 616–627 (2014).
Kibbe, M. R. et al. Citric acid-based elastomers provide a biocompatible interface for vascular grafts. J. Biomed. Mater. Res. A 93A, 314–324 (2010).
Motlagh, D. et al. Hemocompatibility evaluation of poly(diol citrate) in vitro for vascular tissue engineering. J. Biomed. Mater. Res. A 82A, 907–916 (2007).
Dendukuri, D., Pregibon, D. C., Collins, J., Hatton, T. A. & Doyle, P. S. Continuous-flow lithography for high-throughput microparticle synthesis. Nature Mater. 5, 365–369 (2006).
Derby, B. Printing and prototyping of tissues and scaffolds. Science 338, 921–926 (2012).
Hoshi, R. A. Nanoporous biodegradable elastomers. Adv. Mater. 21, 188–192 (2009).
Nagueh, S. F. et al. Altered titin expression, myocardial stiffness, and left ventricular function in patients with dilated cardiomyopathy. Circulation 110, 155–162 (2004).
Weis, S. M. et al. Myocardial mechanics and collagen structure in the osteogenesis imperfecta murine (oim). Circ. Res. 87, 663–669 (2000).
Coirault, C. et al. Increased compliance in diaphragm muscle of the cardiomyopathic Syrian hamster. J. Appl. Physiol. 85, 1762–1769 (1998).
Omens, J. H. Stress and strain as regulators of myocardial growth. Prog. Biophys. Mol. Biol. 69, 559–572 (1998).
Yeh, W. C. et al. Elastic modulus measurements of human liver and correlation with pathology. Ultrasound Med. Biol. 28, 467–474 (2002).
Merkel, T. C., Bondar, V. I., Nagai, K., Freeman, B. D. & Pinnau, I. Gas sorption, diffusion, and permeation in poly(dimethylsiloxane). J. Polym. Sci. B 38, 415–434 (2000).
Toepke, M. W. & Beebe, D. J. PDMS absorption of small molecules and consequences in microfluidic applications. Lab Chip 6, 1484–1486 (2006).
Gaillard, P. J. et al. Establishment and functional characterization of an in vitro model of the blood-brain barrier, comprising a co-culture of brain capillary endothelial cells and astrocytes. Eur. J. Pharm. Sci. 12, 215–222 (2001).
Yuan, W., Lv, Y., Zeng, M. & Fu, B. M. Non-invasive measurement of solute permeability in cerebral microvessels of the rat. Microvasc. Res. 77, 166–173 (2009).
Adamson, R. H., Huxley, V. H. & Curry, F. E. Single capillary permeability to proteins having similar size but different charge. Am. J. Physiol. 254, H304–H312 (1988).
Woosley, R. L., Chen, Y., Freiman, J. P. & Gillis, R. A. Mechanism of the cardiotoxic actions of terfenadine. JAMA 269, 1532–1536 (1993).
Ogawa, S. et al. Three-dimensional culture and cAMP signaling promote the maturation of human pluripotent stem cell-derived hepatocytes. Development 140, 3285–3296 (2013).
Boudou, T. et al. A microfabricated platform to measure and manipulate the mechanics of engineered cardiac microtissues. Tissue Eng. A 18, 910–919 (2012).
Radisic, M. et al. Functional assembly of engineered myocardium by electrical stimulation of cardiac myocytes cultured on scaffolds. Proc. Natl Acad. Sci. USA 101, 18129–18134 (2004).
Tulloch, N. L. et al. Growth of engineered human myocardium with mechanical loading and vascular coculture. Circ. Res. 109, 47–59 (2011).
Wu, W., Allen, R. A. & Wang, Y. Fast-degrading elastomer enables rapid remodeling of a cell-free synthetic graft into a neoartery. Nature Med. 18, 1148–1153 (2012).
Bhatia, S., Balis, U., Yarmush, M. & Toner, M. Effect of cell–cell interactions in preservation of cellular phenotype: cocultivation of hepatocytes and nonparenchymal cells. FASEB J. 13, 1883–1900 (1999).
Mazza, E. & Ehret, A. E. Mechanical biocompatibility of highly deformable biomedical materials. J. Mech. Behav. Biomed. Mater. 48, 100–124 (2015).
Engelmayr, G. C. et al. Accordion-like honeycombs for tissue engineering of cardiac anisotropy. Nature Mater. 7, 1003–1010 (2008).
Nava, A., Mazza, E., Furrer, M., Villiger, P. & Reinhart, W. In vivo mechanical characterization of human liver. Med. Image Anal. 12, 203–216 (2008).
Hoshi, R. A. et al. The blood and vascular cell compatibility of heparin-modified ePTFE vascular grafts. Biomaterials 34, 30–41 (2013).
Sefton, M. V., Gemmell, C. H. & Gorbet, M. B. in Biomaterials Science 3rd edn (eds Ratner, B. D., Hoffman, A. S., Schoen, F. J. & Lemons, J. E.) 758–760 (Academic, 2013).
Zhang, B., Green, J. V., Murthy, S. K. & Radisic, M. Label-free enrichment of functional cardiomyocytes using microfluidic deterministic lateral flow displacement. PLoS ONE 7, e37619 (2012).
Ogawa, M. et al. Directed differentiation of cholangiocytes from human pluripotent stem cells. Nature Biotech. 33, 853–861 (2015).
Berry, M. & Friend, D. High-yield preparation of isolated rat liver parenchymal cells: a biochemical and fine structural study. J. Cell Biol. 43, 506–520 (1969).
Kennedy, M., D’Souza, S. L., Lynch-Kattman, M., Schwantz, S. & Keller, G. Development of the hemangioblast defines the onset of hematopoiesis in human ES cell differentiation cultures. Blood 109, 2679–2687 (2007).
Lian, X. et al. Directed cardiomyocyte differentiation from human pluripotent stem cells by modulating Wnt/β-catenin signaling under fully defined conditions. Nature Protocols 8, 162–175 (2013).
Zeebregts, C., Heijmen, R., Van Den Dungen, J. & Van Schilfgaarde, R. Non-methods of vascular anastomosis. Br. J. Surg. 90, 261–271 (2003).
Acknowledgements
We thank K. Marjan and P. Lai from the University Health Network, Toronto, for their help in the optical mapping analysis. We thank Y. Liu from Osaka University, Japan, for her help in quantifying the platelet coverage on the AngioChip in the blood perfusion study. We thank J. W. Miklas and Y. Xiao for their helpful discussion regarding human cardiomyocyte culture and cell seeding. We thank A. Sofla for his help with the POMaC synthesis. We thank A. Keating and I. Rashedi for providing hMSCs and Y. Zhao for her help in culturing and expanding hMSCs. We thank J. Yang for suggestions regarding POMaC synthesis. This work was made possible by the National Sciences and Engineering Research Council of Canada (NSERC) Steacie Fellowship to M.R. This work was also financially supported by the Canadian Institutes of Health Research (CIHR) Operating Grants (MOP-126027 and MOP-137107), the Heart and Stroke Foundation GIA T6946, NSERC–CIHR Collaborative Health Research Grant (CHRPJ 385981-10), NSERC Discovery Grant (RGPIN 326982-10), NSERC Discovery Accelerator Supplement (RGPAS 396125-10) and National Institutes of Health Grant 2R01 HL076485.
Author information
Authors and Affiliations
Contributions
B.Z. developed the AngioChip concept, designed and performed experiments, analysed data and prepared the manuscript. M.M. contributed to mechanical testing, polymer characterization, sprouting assay, blood perfusion experiments, and vascular anastomosis surgery. M.D.C. performed the primary rat hepatocyte isolation and urea assay. S.O. differentiated hESC-derived hepatocytes. A.K. performed polymer mechanical testing. A.P. differentiated hESC-derived cardiomyocytes and contributed to the whole blood perfusion experiment and optical mapping. L.A.W. performed extraction of human whole blood. S.M. and K.N. performed optical mapping measurements and analysis. J.K. performed mass spectrometry analysis. L.R. contributed to the direct vascular anastomosis surgery; A.M. performed the direct vascular anastomosis surgery; S.S.N. contributed to the direct vascular anastomosis surgery and writing of the manuscript. A.R.W. contributed to the writing of the manuscript. G.K. contributed to the writing of the manuscript. M.V.S. contributed to writing of the manuscript. M.R. developed the AngioChip concept, supervised the work and wrote the manuscript.
Corresponding author
Ethics declarations
Competing interests
M.R. and B.Z. are amongst co-founders of TARA Biosystems and they hold equity in this company.
Supplementary information
Supplementary Information
Supplementary Information (PDF 25074 kb)
Supplementary Movie 1
Supplementary Movie 1 (MOV 4710 kb)
Supplementary Movie 2
Supplementary Movie 2 (MOV 2917 kb)
Supplementary Movie 3
Supplementary Movie 3 (MOV 4817 kb)
Supplementary Movie 4
Supplementary Movie 4 (MOV 8083 kb)
Supplementary Movie 5
Supplementary Movie 5 (MOV 6213 kb)
Supplementary Movie 6
Supplementary Movie 6 (MOV 511 kb)
Supplementary Movie 7
Supplementary Movie 7 (MOV 9570 kb)
Rights and permissions
About this article
Cite this article
Zhang, B., Montgomery, M., Chamberlain, M. et al. Biodegradable scaffold with built-in vasculature for organ-on-a-chip engineering and direct surgical anastomosis. Nature Mater 15, 669–678 (2016). https://doi.org/10.1038/nmat4570
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nmat4570
This article is cited by
-
Opportunities and challenges in cardiac tissue engineering from an analysis of two decades of advances
Nature Biomedical Engineering (2022)
-
Engineering the multiscale complexity of vascular networks
Nature Reviews Materials (2022)
-
Bioengineering approaches to treat the failing heart: from cell biology to 3D printing
Nature Reviews Cardiology (2022)
-
Challenges and opportunities for the next generation of cardiovascular tissue engineering
Nature Methods (2022)
-
Integrated Organ-on-a-chip with Human-induced Pluripotent Stem Cells Directional Differentiation for 3D Skin Model Generation
Journal of Wuhan University of Technology-Mater. Sci. Ed. (2022)