Abstract
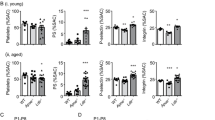
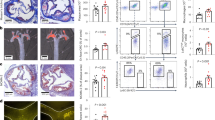
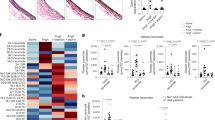
Dyslipidemia is associated with a prothrombotic phenotype; however, the mechanisms responsible for enhanced platelet reactivity remain unclear. Proatherosclerotic lipid abnormalities are associated with both enhanced oxidant stress and the generation of biologically active oxidized lipids, including potential ligands for the scavenger receptor CD36, a major platelet glycoprotein. Using multiple mouse in vivo thrombosis models, we now demonstrate that genetic deletion of Cd36 protects mice from hyperlipidemia-associated enhanced platelet reactivity and the accompanying prothrombotic phenotype. Structurally defined oxidized choline glycerophospholipids that serve as high-affinity ligands for CD36 were at markedly increased levels in the plasma of hyperlipidemic mice and in the plasma of humans with low HDL levels, were able to bind platelets via CD36 and, at pathophysiological levels, promoted platelet activation via CD36. Thus, interactions of platelet CD36 with specific endogenous oxidized lipids play a crucial role in the well-known clinical associations between dyslipidemia, oxidant stress and a prothrombotic phenotype.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Trip, M.D., Cats, V.M., van Capelle, F.J. & Vreeken, J. Platelet hyperreactivity and prognosis in survivors of myocardial infarction. N. Engl. J. Med. 322, 1549–1554 (1990).
Lacoste, L. et al. Hyperlipidemia and coronary disease. Correction of the increased thrombogenic potential with cholesterol reduction. Circulation 92, 3172–3177 (1995).
Kabbani, S.S. et al. Platelet reactivity characterized prospectively: a determinant of outcome 90 days after percutaneous coronary intervention. Circulation 104, 181–186 (2001).
Vanschoonbeek, K. et al. Thrombin-induced hyperactivity of platelets of young stroke patients: involvement of thrombin receptors in the subject-dependent variability in Ca2+ signal generation. Thromb. Haemost. 88, 931–937 (2002).
Kabbani, S.S. et al. Usefulness of platelet reactivity before percutaneous coronary intervention in determining cardiac risk one year later. Am. J. Cardiol. 91, 876–878 (2003).
Carvalho, A.C., Colman, R.W. & Lees, R.S. Platelet function in hyperlipoproteinemia. N. Engl. J. Med. 290, 434–438 (1974).
Stuart, M.J., Gerrard, J.M. & White, J.G. Effect of cholesterol on production of thromboxane b2 by platelets in vitro. N. Engl. J. Med. 302, 6–10 (1980).
Davi, G. et al. Increased thromboxane biosynthesis in type IIa hypercholesterolemia. Circulation 85, 1792–1798 (1992).
Davi, G. et al. Increased levels of soluble P-selectin in hypercholesterolemic patients. Circulation 97, 953–957 (1998).
Cipollone, F. et al. Association between enhanced soluble CD40L and prothrombotic state in hypercholesterolemia: effects of statin therapy. Circulation 106, 399–402 (2002).
Wang, T.H., Bhatt, D.L. & Topol, E.J. Aspirin and clopidogrel resistance: an emerging clinical entity. Eur. Heart J. (2005).
Salonen, J.T. et al. Effects of antioxidant supplementation on platelet function: a randomized pair-matched, placebo-controlled, double-blind trial in men with low antioxidant status. Am. J. Clin. Nutr. 53, 1222–1229 (1991).
Vericel, E., Januel, C., Carreras, M., Moulin, P. & Lagarde, M. Diabetic patients without vascular complications display enhanced basal platelet activation and decreased antioxidant status. Diabetes 53, 1046–1051 (2004).
Morita, H., Ikeda, H., Haramaki, N., Eguchi, H. & Imaizumi, T. Only two-week smoking cessation improves platelet aggregability and intraplatelet redox imbalance of long-term smokers. J. Am. Coll. Cardiol. 45, 589–594 (2005).
Berliner, J.A. & Watson, A.D. A role for oxidized phospholipids in atherosclerosis. N. Engl. J. Med. 353, 9–11 (2005).
Podrez, E.A. et al. Identification of a novel family of oxidized phospholipids that serve as ligands for the macrophage scavenger receptor CD36. J. Biol. Chem. 277, 38503–38516 (2002).
Podrez, E.A. et al. A novel family of atherogenic oxidized phospholipids promotes macrophage foam cell formation via the scavenger receptor CD36 and is enriched in atherosclerotic lesions. J. Biol. Chem. 277, 38517–38523 (2002).
Sun, M. et al. Light-induced oxidation of photoreceptor outer segment phospholipids generates ligands for CD36-mediated phagocytosis by retinal pigment epithelium: a potential mechanism for modulating outer segment phagocytosis under oxidant stress conditions. J. Biol. Chem. 281, 4222–4230 (2006).
Podrez, E.A. et al. Macrophage scavenger receptor CD36 is the major receptor for LDL modified by monocyte-generated reactive nitrogen species. J. Clin. Invest. 105, 1095–1108 (2000).
Febbraio, M., Hajjar, D.P. & Silverstein, R.L. CD36: a class B scavenger receptor involved in angiogenesis, atherosclerosis, inflammation, and lipid metabolism. J. Clin. Invest. 108, 785–791 (2001).
Febbraio, M. et al. Targeted disruption of the class B scavenger receptor CD36 protects against atherosclerotic lesion development in mice. J. Clin. Invest. 105, 1049–1056 (2000).
Tandon, N.N., Lipsky, R.H., Burgess, W.H. & Jamieson, G.A. Isolation and characterization of platelet glycoprotein IV (CD36). J. Biol. Chem. 264, 7570–7575 (1989).
Hoebe, K. et al. CD36 is a sensor of diacylglycerides. Nature 433, 523–527 (2005).
Podrez, E.A., Schmitt, D., Hoff, H.F. & Hazen, S.L. Myeloperoxidase-generated reactive nitrogen species convert LDL into an atherogenic form in vitro. J. Clin. Invest. 103, 1547–1560 (1999).
Kieffer, N. et al. Developmentally-regulated expression of a 78-kDa erythroblast membrane glycoprotein immunologically related to the platelet thrombospondin receptor. Biochem. J. 262, 835–842 (1989).
Watson, A.D. et al. Structural identification by mass spectrometry of oxidized phospholipids in minimally-oxidized low-density lipoproteins that induce monocyte/endothelial interactions and evidence for their presence in vivo. J. Biol. Chem. 272, 13597–13607 (1997).
Boullier, A. et al. Phosphocholine as a pattern recognition ligand for CD36. J. Lipid Res. 46, 969–976 (2005).
Simon, D.I. et al. Decreased neointimal formation in Mac1−/− mice reveals a role for inflammation in vascular repair after angioplasty. J. Clin. Invest. 105, 293–300 (2000).
Sarma, J. et al. Increased platelet binding to circulating monocytes in acute coronary syndromes. Circulation 105, 2166–2171 (2002).
Huo, Y. et al. Circulating activated platelets exacerbate atherosclerosis in mice deficient in apolipoprotein E. Nat. Med. 9, 61–67 (2003).
Pearce, S.F. et al. Recombinant glutathione S-transferase/CD36 fusion proteins define an oxidized low-density lipoprotein–binding domain. J. Biol. Chem. 273, 34875–34881 (1998).
Febbraio, M., Guy, E. & Silverstein, R.L. Stem cell transplantation reveals that absence of macrophage CD36 is protective against atherosclerosis. Arterioscler. Thromb. Vasc. Biol. 24, 2333–2338 (2004).
Eitzman, D.T., Westrick, R.J., Xu, Z., Tyson, J. & Ginsburg, D. Hyperlipidemia promotes thrombosis after injury to atherosclerotic vessels in apolipoprotein E–deficient mice. Arterioscler. Thromb. Vasc. Biol. 20, 1831–1834 (2000).
Schafer, K. et al. Enhanced thrombosis in atherosclerosis-prone mice is associated with increased arterial expression of plasminogen activator inhibitor-1. Arterioscler. Thromb. Vasc. Biol. 23, 2097–2103 (2003).
Barter, P.J. et al. Antiinflammatory properties of HDL. Circ. Res. 95, 764–772 (2004).
Bodart, V. et al. CD36 mediates the cardiovascular action of growth hormone-releasing peptides in the heart. Circ. Res. 90, 844–849 (2002).
Philips, J.A., Rubin, E.J. & Perrimon, N. Drosophila RNAi screen reveals CD36 family member required for mycobacterial infection. Science 309, 1251–1253 (2005).
Tandon, N.N., Ockenhouse, C.F., Greco, N.J. & Jamieson, G.A. Adhesive functions of platelets lacking glycoprotein IV (CD36). Blood 78, 2809–2813 (1991).
Englyst, N.A., Taube, J.M., Aitman, T.J., Baglin, T.P. & Byrne, C.D. A novel role for CD36 in VLDL-enhanced platelet activation. Diabetes 52, 1248–1255 (2003).
Huang, M.M., Bolen, J.B., Barnwell, J.W., Shattil, S.J. & Brugge, J.S. Membrane glycoprotein IV (CD36) is physically associated with the Fyn, Lyn, and Yes protein-tyrosine kinases in human platelets. Proc. Natl. Acad. Sci. USA 88, 7844–7848 (1991).
Maschberger, P. et al. Mildly oxidized low-density lipoprotein rapidly stimulates via activation of the lysophosphatidic acid receptor Src family and Syk tyrosine kinases and Ca2+ influx in human platelets. J. Biol. Chem. 275, 19159–19166 (2000).
Angelillo-Scherrer, A. et al. Deficiency or inhibition of Gas6 causes platelet dysfunction and protects mice against thrombosis. Nat. Med. 7, 215–221 (2001).
Andre, P. et al. CD40L stabilizes arterial thrombi by a β3 integrin–dependent mechanism. Nat. Med. 8, 247–252 (2002).
Prevost, N. et al. Eph kinases and ephrins support thrombus growth and stability by regulating integrin outside-in signaling in platelets. Proc. Natl. Acad. Sci. USA 102, 9820–9825 (2005).
Plow, E.F. et al. Related binding mechanisms for fibrinogen, fibronectin, von Willebrand factor, and thrombospondin on thrombin-stimulated human platelets. Blood 66, 724–727 (1985).
Acknowledgements
We thank E. Plow for thoughtful comments and criticisms and W. Feng, W. Li and V. Verbovetskaya for technical assistance. This work was supported in part by National Institutes of Health grants HL077213 and HL053315 (E.A.P.) and by a Scientist Development Grant from the American Heart Association (E.A.P.), HL70621 and HL076491 (S.L.H.), HL 70083 (M.F.), HL072942 and HL46403 (R.L.S. and M.F.), HL071625, HL073311 and HL077107 (T.V.B.), HL53315 (R.G.S.), Cleveland Clinic Specialized Centers for Clinically Oriented Research (P01 HL077107, S.L.H.; and P50 HL81011, R.L.S. and M.F.) and the Cleveland Clinic Foundation General Clinical Research Center (M01 RR018390).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Text and Figures
Supplementary Methods (PDF 128 kb)
Rights and permissions
About this article
Cite this article
Podrez, E., Byzova, T., Febbraio, M. et al. Platelet CD36 links hyperlipidemia, oxidant stress and a prothrombotic phenotype. Nat Med 13, 1086–1095 (2007). https://doi.org/10.1038/nm1626
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nm1626
This article is cited by
-
Research progress of nephrotic syndrome accompanied by thromboembolism
International Urology and Nephrology (2023)
-
On-treatment platelet reactivity through the thromboxane A2 or P2Y12 platelet receptor pathways is not affected by pelacarsen
Journal of Thrombosis and Thrombolysis (2023)
-
Efficient megakaryopoiesis and platelet production require phospholipid remodeling and PUFA uptake through CD36
Nature Cardiovascular Research (2023)
-
Association of FXI activity with thrombo-inflammation, extracellular matrix, lipid metabolism and apoptosis in venous thrombosis
Scientific Reports (2022)
-
Mild hyperlipidemia in mice aggravates platelet responsiveness in thrombus formation and exploration of platelet proteome and lipidome
Scientific Reports (2020)