Abstract
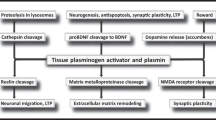
Tissue-plasminogen activator (t-PA) is now available for the treatment of thrombo-embolic stroke but adverse effects have been reported in some patients, particularly hemorrhaging. In contrast, the results of animal studies have indicated that t-PA could increase neuronal damage after focal cerebral ischemia. Here we report for the first time that t-PA potentiates signaling mediated by glutamatergic receptors by modifying the properties of the N-methyl-D-aspartate (NMDA) receptor. When depolarized, cortical neurons release bio-active t-PA that interacts with and cleaves the NR1 subunit of the NMDA receptor. Moreover, the treatment with recombinant t-PA leads to a 37% increase in NMDA-stimulated fura-2 fluorescence, which may reflect an increased NMDA-receptor function. These results were confirmed in vivo by the intrastriatal injection of recombinant-PA, which potentiated the excitotoxic lesions induced by NMDA. These data provide insight into the regulation of NMDA-receptor–mediated signaling and could initiate therapeutic strategies to improve the efficacy of t-PA treatment in man.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Choi, D.W. Glutamate neurotoxicity and diseases of the nervous system. Neuron 1, 623–634 (1988).
Lee, J.M., Zipfel, G.J. & Choi, D.W. The changing landscape of ischemic brain injury mechanisms. Nature 399, 7–14 (1999).
The National Institute of Neurological Disorders and Stroke rt-PA Stroke Study Group. Tissue-plasminogen activator for acute ischemic stroke. N. Engl. J. Med. 333, 1581–1587 (1995).
Lijnen, H.R., Hoylaerts, M. & Collen, D. Isolation and characterization of a human plasma protein with affinity for the lysine binding sites in plasminogen. Role in the regulation of fibrinolysis and identification as histidine-rich glycoprotein. J. Biol. Chem. 255, 10214–10222 (1980).
Sappino, A.P. et al. Extracellular proteolysis in the adult murine brain. J. Clin. Invest. 92, 679–685 (1993).
Baranes, D. et al. Tissue-plasminogen activator contributes to the late phase of LTP and to synaptic growth in the hippocampal mossy fiber pathway. Neuron 21, 813–825 (1998).
Friedman, G.C. & Seeds, N.W. Tissue-plasminogen activator expression in the embryonic nervous system. Brain Res. Dev. 81, 41–49 (1994).
Tsirka, S.E., Gualandris, A., Amaral, D.G. & Strickland, S. Excitotoxin-induced neuronal degeneration and seizure are mediated by tissue-plasminogen activator. Nature 377, 340–344 (1995).
Wang, Y.F. et al. Tissue-plasminogen activator (t-PA) increases neuronal damage after focal cerebral ischemia in wild-type and tPA-deficient mice. Nature Med. 4, 228–231 (1998).
Kirkegaard, T. et al. Engineering of conformations of plasminogen activator inhibitor-1. A crucial role of β-strand 5A residues in the transition of active form to latent and substrate forms. Eur. J. Biochem. 263, 577–586 (1999).
Gualandris, A., Jones, T.E., Strickland, S. & Tsirka, S.E. Membrane depolarization induces calcium-dependent secretion of tissue-plasminogen activator. J. Neurosci. 16, 2220–2225 (1996).
Verderio, C. et al. Tetanus toxin blocks the exocytosis of synaptic vesicles clustered at synapses but not of synaptic vesicles in isolated axons. J. Neurosci. 19, 6723–6732 (1999).
Carmeliet, P. et al. Physiological consequences of loss of plasminogen activator gene function in mice. Nature 368, 419–424 (1994).
Nowack, L., Bregestovki, P. Ascher, P, Herbet. A & Prochaintz,A. Magnesium gates glutamate-activated channels in mouse central neurons. Nature 307, 462–465 (1984).
Sattler, R., Charlton, M.P., Hafner, M. & Tymiansky, M. Distinct influx pathways, not calcium load, determine neuronal vulnerability to calcium neurotoxicity. J. Neurochem. 71, 2349–2364 (1998).
Mori, H. & Mishina, M. Structure and function of the NMDA receptor channel. Neuropharmacology 34, 1219–1237 (1995).
Carmeliet, P. & Collen, D. Molecular analysis of blood vessel formation and disease. Am. J. Physiol. 273, H2091–H2104 (1997).
Kim, Y.H., Park, J.H., Hong, S.H. & Koh, J.Y. Nonproteolytic neuroprotection by human recombinant tissue-plasminogen activator. Science 284, 647–650 (1999).
Rogove, A.D. & Tsirka, S.E. Neurotoxic responses by microglia elicited by excitotoxic injury in the mouse hippocampus. Curr. Biol. 8, 19–25 (1998).
Parmer, R.J. et al. Tissue-plasminogen activator (t-PA) is targeted to the regulated secretory pathway. Catecholamine storage vesicles as a reservoir for the rapid release of t-PA. J. Biol. Chem. 272, 976–982 (1997).
Gils, A. & Declerck, J.P. Proteinase specificity and functional diversity in point mutants of plasminogen activator inhibitor 1. J. Biol. Chem. 272, 12662–12666 (1997).
Ding, L. et al. Origins of the specificity of tissue-type plasminogen activator. Proc. Natl. Acad. Sci. USA 15, 7627–7631 (1995).
Hirai, H., Kirsch, J., Laube, B., Betz, H. & Kuhse, J. The glycine binding site of the N-methyl-D-aspartate receptor subunit NR1: identification of novel determinants of co-agonist potentiation in the extracellular M3–M4 loop region. Proc. Natl. Acad. Sci. USA 11, 6031–6036 (1996).
Rose, K., Goldberg, M.P. & Choi, D.W. Cytotoxicity in murine cortical cell culture. in In Vitro Biological Methods (Tyson C.A. & Frazier J.M., eds), 46–60 (Academic Press, San Diego, California, 1993).
Buisson, A. et al. Up-regulation of a serine protease inhibitor in astrocytes mediates the neuroprotective activity of transforming growth factor beta1. FASEB J. 12, 1683–1691 (1998).
Koh, J.Y. & Choi, D.W. Quantitative determination of glutamate mediated cortical neuronal injury in cell culture by lactate dehydrogenase efflux assay. J. Neurosci. Meth. 20, 83–90 (1987).
Paxinos, G. & Watson, C. The rat brain stereotaxic coordinates. (Academic Press, Australia, 1982).
Acknowledgements
We thank L. Plawinski for his technical assistance and A. Ruocco for his involvement in this study. We were supported by grants from the CNRS and the University of Caen. The following received doctoral bursaries: O.N. (Commissariat à l'Energie Atomique-CEA), F.D. (Regional Council of Lower Normandy), and C.A. (Ministry of Education and Research). The investigation was carried out within the framework of the Functional Neuroimaging Institute (IFR 47).
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Nicole, O., Docagne, F., Ali, C. et al. The proteolytic activity of tissue-plasminogen activator enhances NMDA receptor-mediated signaling. Nat Med 7, 59–64 (2001). https://doi.org/10.1038/83358
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/83358
This article is cited by
-
Effects of the New Thrombolytic Compound LT3001 on Acute Brain Tissue Damage After Focal Embolic Stroke in Rats
Translational Stroke Research (2024)
-
Blood tissue Plasminogen Activator (tPA) of liver origin contributes to neurovascular coupling involving brain endothelial N-Methyl-D-Aspartate (NMDA) receptors
Fluids and Barriers of the CNS (2023)
-
Tranexamic acid for haemostasis and beyond: does dose matter?
Thrombosis Journal (2023)
-
Modulations of the neuronal trafficking of tissue-type plasminogen activator (tPA) influences glutamate release
Cell Death & Disease (2023)
-
Surgical Robotics for Intracerebral Hemorrhage Treatment: State of the Art and Future Directions
Annals of Biomedical Engineering (2023)