Abstract
Detailed understanding of the signaling intermediates that confer the sensing of intracellular viral nucleic acids for induction of type I interferons is critical for strategies to curtail viral mechanisms that impede innate immune defenses. Here we show that the activation of the microtubule-associated guanine nucleotide exchange factor GEF-H1, encoded by Arhgef2, is essential for sensing of foreign RNA by RIG-I–like receptors. Activation of GEF-H1 controls RIG-I–dependent and Mda5-dependent phosphorylation of IRF3 and induction of IFN-β expression in macrophages. Generation of Arhgef2−/− mice revealed a pronounced signaling defect that prevented antiviral host responses to encephalomyocarditis virus and influenza A virus. Microtubule networks sequester GEF-H1 that upon activation is released to enable antiviral signaling by intracellular nucleic acid detection pathways.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Takeuchi, O. & Akira, S. Pattern recognition receptors and inflammation. Cell 140, 805–820 (2010).
Desmet, C.J. & Ishii, K.J. Nucleic acid sensing at the interface between innate and adaptive immunity in vaccination. Nat. Rev. Immunol. 12, 479–491 (2012).
Merino-Gracia, J., Garcia-Mayoral, M.F. & Rodriguez-Crespo, I. The association of viral proteins with host cell dynein components during virus infection. FEBS J. 278, 2997–3011 (2011).
Krendel, M., Zenke, F.T. & Bokoch, G.M. Nucleotide exchange factor GEF-H1 mediates cross-talk between microtubules and the actin cytoskeleton. Nat. Cell Biol. 4, 294–301 (2002).
Meiri, D. et al. Mechanistic insight into the microtubule and actin cytoskeleton coupling through dynein-dependent RhoGEF inhibition. Mol. Cell 45, 642–655 (2012).
Matsuzawa, T., Kuwae, A., Yoshida, S., Sasakawa, C. & Abe, A. Enteropathogenic Escherichia coli activates the RhoA signaling pathway via the stimulation of GEF-H1. EMBO J. 23, 3570–3582 (2004).
Fukazawa, A. et al. GEF-H1 mediated control of NOD1 dependent NF-kappaB activation by Shigella effectors. PLoS Pathog. 4, e1000228 (2008).
Zhao, Y. et al. Control of NOD2 and Rip2-dependent innate immune activation by GEF-H1. Inflamm. Bowel Dis. 18, 603–612 (2012).
Yoneyama, M. & Fujita, T. RNA recognition and signal transduction by RIG-I-like receptors. Immunol. Rev. 227, 54–65 (2009).
Lund, J.M. et al. Recognition of single-stranded RNA viruses by Toll-like receptor 7. Proc. Natl. Acad. Sci. USA 101, 5598–5603 (2004).
Alexopoulou, L., Holt, A.C., Medzhitov, R. & Flavell, R.A. Recognition of double-stranded RNA and activation of NF-κB by Toll-like receptor 3. Nature 413, 732–738 (2001).
Takeuchi, O. & Akira, S. Innate immunity to virus infection. Immunol. Rev. 227, 75–86 (2009).
Yoneyama, M. et al. Shared and unique functions of the DExD/H-box helicases RIG-I, MDA5, and LGP2 in antiviral innate immunity. J. Immunol. 175, 2851–2858 (2005).
Zhang, Z. et al. The helicase DDX41 senses intracellular DNA mediated by the adaptor STING in dendritic cells. Nat. Immunol. 12, 959–965 (2011).
Gitlin, L. et al. Essential role of mda-5 in type I IFN responses to polyriboinosinic:polyribocytidylic acid and encephalomyocarditis picornavirus. Proc. Natl. Acad. Sci. USA 103, 8459–8464 (2006).
Kato, H. et al. Differential roles of MDA5 and RIG-I helicases in the recognition of RNA viruses. Nature 441, 101–105 (2006).
Kato, H. et al. Length-dependent recognition of double-stranded ribonucleic acids by retinoic acid-inducible gene-I and melanoma differentiation-associated gene 5. J. Exp. Med. 205, 1601–1610 (2008).
Hornung, V. et al. 5′-Triphosphate RNA is the ligand for RIG-I. Science 314, 994–997 (2006).
Pichlmair, A. et al. RIG-I-mediated antiviral responses to single-stranded RNA bearing 5′-phosphates. Science 314, 997–1001 (2006).
Taniguchi, T. & Takaoka, A. The interferon-alpha/beta system in antiviral responses: a multimodal machinery of gene regulation by the IRF family of transcription factors. Curr. Opin. Immunol. 14, 111–116 (2002).
Ishikawa, H., Ma, Z. & Barber, G.N. STING regulates intracellular DNA-mediated, type I interferon-dependent innate immunity. Nature 461, 788–792 (2009).
Parvatiyar, K. et al. The helicase DDX41 recognizes the bacterial secondary messengers cyclic di-GMP and cyclic di-AMP to activate a type I interferon immune response. Nat. Immunol. 13, 1155–1161 (2012).
Fitzgerald, K.A. et al. IKKɛε and TBK1 are essential components of the IRF3 signaling pathway. Nat. Immunol. 4, 491–496 (2003).
Yang, K. et al. Hsp90 regulates activation of interferon regulatory factor 3 and TBK-1 stabilization in Sendai virus-infected cells. Mol. Biol. Cell 17, 1461–1471 (2006).
Birkenfeld, J., Nalbant, P., Yoon, S.H. & Bokoch, G.M. Cellular functions of GEF-H1, a microtubule-regulated Rho-GEF: is altered GEF-H1 activity a crucial determinant of disease pathogenesis? Trends Cell Biol. 18, 210–219 (2008).
Meiri, D. et al. Modulation of Rho guanine exchange factor Lfc activity by protein kinase A-mediated phosphorylation. Mol. Cell. Biol. 29, 5963–5973 (2009).
Zenke, F.T. et al. p21-activated kinase 1 phosphorylates and regulates 14–3-3 binding to GEF-H1, a microtubule-localized Rho exchange factor. J. Biol. Chem. 279, 18392–18400 (2004).
Garcia, A., Cayla, X. & Sontag, E. Protein phosphatase 2A: a definite player in viral and parasitic regulation. Microbes Infect. 2, 401–407 (2000).
Zhang, Z., Yuan, B., Lu, N., Facchinetti, V. & Liu, Y.J. DHX9 pairs with IPS-1 to sense double-stranded RNA in myeloid dendritic cells. J. Immunol. 187, 4501–4508 (2011).
Diebold, S.S., Kaisho, T., Hemmi, H., Akira, S. & Reis e Sousa, C. Innate antiviral responses by means of TLR7-mediated recognition of single-stranded RNA. Science 303, 1529–1531 (2004).
Garcia-Sastre, A. et al. Influenza A virus lacking the NS1 gene replicates in interferon-deficient systems. Virology 252, 324–330 (1998).
Manicassamy, B. et al. Analysis of in vivo dynamics of influenza virus infection in mice using a GFP reporter virus. Proc. Natl. Acad. Sci. USA 107, 11531–11536 (2010).
Georgel, P. et al. Vesicular stomatitis virus glycoprotein G activates a specific antiviral Toll-like receptor 4-dependent pathway. Virology 362, 304–313 (2007).
Kumar, H. et al. Essential role of IPS-1 in innate immune responses against RNA viruses. J. Exp. Med. 203, 1795–1803 (2006).
Arpaia, N. et al. TLR signaling is required for Salmonella typhimurium virulence. Cell 144, 675–688 (2011).
Wittmann, T. & Waterman-Storer, C.M. Cell motility: can Rho GTPases and microtubules point the way? J. Cell Sci. 114, 3795–3803 (2001).
Misawa, T. et al. Microtubule-driven spatial arrangement of mitochondria promotes activation of the NLRP3 inflammasome. Nat. Immunol. 14, 454–460 (2013).
Ohman, T., Rintahaka, J., Kalkkinen, N., Matikainen, S. & Nyman, T.A. Actin and RIG-I/MAVS signaling components translocate to mitochondria upon influenza A virus infection of human primary macrophages. J. Immunol. 182, 5682–5692 (2009).
Palazzo, A.F., Cook, T.A., Alberts, A.S. & Gundersen, G.G. mDia mediates Rho-regulated formation and orientation of stable microtubules. Nat. Cell Biol. 3, 723–729 (2001).
Minin, A.A. et al. Regulation of mitochondria distribution by RhoA and formins. J. Cell Sci. 119, 659–670 (2006).
Perry, A.K., Chow, E.K., Goodnough, J.B., Yeh, W.C. & Cheng, G. Differential requirement for TANK-binding kinase-1 in type I interferon responses to toll-like receptor activation and viral infection. J. Exp. Med. 199, 1651–1658 (2004).
Sakaguchi, S. et al. Essential role of IRF-3 in lipopolysaccharide-induced interferon-beta gene expression and endotoxin shock. Biochem. Biophys. Res. Commun. 306, 860–866 (2003).
Aksoy, E. et al. Inhibition of phosphoinositide 3-kinase enhances TRIF-dependent NF-kappa B activation and IFN-beta synthesis downstream of Toll-like receptor 3 and 4. Eur. J. Immunol. 35, 2200–2209 (2005).
Honda, K., Yanai, H., Takaoka, A. & Taniguchi, T. Regulation of the type I IFN induction: a current view. Int. Immunol. 17, 1367–1378 (2005).
Peters, K.L., Smith, H.L., Stark, G.R. & Sen, G.C. IRF-3-dependent, NFkappa B- and JNK-independent activation of the 561 and IFN-beta genes in response to double-stranded RNA. Proc. Natl. Acad. Sci. USA 99, 6322–6327 (2002).
Wang, X. et al. Lack of essential role of NF-κB p50, RelA, and cRel subunits in virus-induced type 1 IFN expression. J. Immunol. 178, 6770–6776 (2007).
Akira, S., Uematsu, S. & Takeuchi, O. Pathogen recognition and innate immunity. Cell 124, 783–801 (2006).
Hale, B.G., Randall, R.E., Ortin, J. & Jackson, D. The multifunctional NS1 protein of influenza A viruses. J. Gen. Virol. 89, 2359–2376 (2008).
Hengge, R. Principles of c-di-GMP signalling in bacteria. Nat. Rev. Microbiol. 7, 263–273 (2009).
Paludan, S.R., Bowie, A.G., Horan, K.A. & Fitzgerald, K.A. Recognition of herpesviruses by the innate immune system. Nat. Rev. Immunol. 11, 143–154 (2011).
Acknowledgements
This work was supported by grants AI-093588 (H.-C.R.), DK-068181 (H.-C.R.), DK-033506 (H.-C.R.), DK-043351 (H.-C.R. and C.T.) and DK-52510 (C.T.) from the US National Institutes of Health.
Author information
Authors and Affiliations
Contributions
H.-S.C., Y.Z., J.-H.S., S.L., N.W. and M.B. carried out experiments; K.J., A.H.S. and C.T. supported the development of research tools and mice; K.L.J. provided virus and advised on virus infection experiments; H.-C.R. conceived of and directed all research, and along with H.-S.C. prepared the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Integrated supplementary information
Supplementary Figure 1 Normal distribution of T cells, B cells and mononuclear phagocytes in mesenteric lymph nodes and spleens of Arhgef2−/− mice.
Flow cytometry analysis of T cells, B cells, and mononuclear phagocytes in mesenteric lymph nodes (MLN) and spleens of C57BL/6 wild-type, Arhgef2+/–, and Arhgef2−/− mice (n=3 per group).
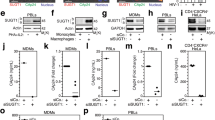
Supplementary Figure 2 TLR4-mediated IFN-β and proinflammatory cytokine production in wild-type and GEF-H1–deficient macrophages.
(a) IFN-β in supernatants of WT or Arhgef2−/− macrophages incubated with LPS for 24 h. (b) Quantitative RT-PCR analysis of Ifnb1 mRNA expression in WT, Arhgef2+/– and Arhgef2−/− macrophages in the presence of LPS (0.2 μg/ml) for 24 h. (c) Quantitative RT-PCR analysis of Il6 and Tnf mRNA expression in WT, Arhgef2+/– and Arhgef2−/− macrophages incubated for 24 h with LPS (0.2 μg/ml). Results are presented relative to the expression of GAPDH. UT, untreated. ND, not detectable. Data are from one experiment representative of three independent experiments and reflect mean ± SD.
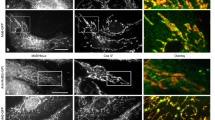
Supplementary Figure 3 Poly(I:C) uptake, microtubule and actin networks in C57BL/6 wild-type and Arhgef2−/− macrophages.
(a) Confocal microscopic images of wild-type and Arhgef2−/− macrophages in the presence of 0.5 μg/ml poly(I:C)-Rhodamine. (b) Flow cytometry analysis of wild-type (blue line) and Arhgef2−/− (red line) macrophages following an incubation with Rhodamine-labeled poly(I:C) (0.5 μg/ml). Data are from one experiment representative of three independent experiments. (c, d) Confocal images of α-tubulin (c) and F-actin (d) in macrophages form Arhgef2−/− mice and their wild-type littermate control in the presence of 0.5 μg/ml poly(I:C) for 3 h. (e) Confocal images of nocodazole-treated (10μM, 40 minutes) wild-type and Arhgef2−/− macrophages in the presence of 0.5 μg/ml poly(I:C)-Rhodamine for 3 h. UT, untreated. Confocal images are from one experiment representative of three independent experiments.
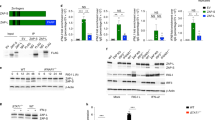
Supplementary Figure 4 GEF-H1–deficient macrophages are susceptible to Influenza A and VSV infection.
(a) Viral replication was visualized by confocoal microscopy of WT or Arhgef2-/- macrophages 12 h after infection (MOI=1) with a recombinant influenza A/PR/8/34 carrying RNA for an NS1-GFP fusion protein. At least 200 cells of WT or Arhgef2−/− macrophages were imaged. Mean fluorescence intensity of each cell is shown in the scatter plot. (b) Plaque assays of the supernatants of VSV-infected C56BL/6 WT and GEF-H1-deficient macrophages. The bar graph represents the viral titers in the supernatants of macrophages after VSV infection (MOI=1). (c) ELISA of IFN-β in the supernatants of VSV-infected C56BL/6 WT and GEF-H1-deficient macrophages (MOI=1). (d) Quantitative RT-PCR analysis of Ifnb1 mRNA expression in VSV-infected C56BL/6 WT and MAVS-deficient macrophages (MOI=1). Results are presented relative to the expression of GAPDH. ND, not detectable. Data reflect mean ± SD. **, P < 0.01, *, P < 0.05 (Student's t-test). (e) Immunoblot analysis of STAT1 phosphorylation (tyrosine 701) in C57BL/6 wild-type or Arhgef2-/- bone marrow-derived macrophages treated with IFN-β (500 U/ml) for 10 minutes, 30 minutes, 4 hours or 16 hours. β-actin serves as a loading control. Data are from one experiment representative of three independent experiments
Supplementary Figure 5 GEF-H1 is dispensable for Ifnb1 and Tnf mRNA induction during S. typhimurium infection.
(a, b) Quantitative RT-PCR analysis of (a) Ifnb1 mRNA and (b) Tnf expression in S. typhimurim-infected (MOI=10) C56BL/6 WT and GEF-H1-deficient macrophages. (c) Intracellular CFU was measured by a gentamicin protection assay in WT and GEF-H1 deficient macrophages infected with S. typhimurim at MOI=10. Results are presented relative to the expression of GAPDH. ND, not detectable (Student's t-test). Data reflect mean ± SD. Data are from one experiment representative of three independent experiments.
Supplementary Figure 6 Graphical abstract.
GEF-H1 emerges as a critical signaling intermediate for cytoplasmic recognition of nucleic acid through the MAVS and STING pathway. GEF-H1 is required for sensing of 5'-triphosphate dsRNA by RIG-I and poly(I:C) by MDA5, and mediates IFN-β expression upon DDX41 activation. GEF-H1 elicits this selective function through its nucleotide exchange activity after release from a microtubule bound reservoir by dephosphorylation. GEF-H1 subsequently becomes part of TBK1 containing signaling complexes that mediate phosphorylation of IRF3 and subsequent Ifnb1 promoter induction. Disruption of the microtubule network prevents activation of GEF-H1 and MAVS signaling. In contrast, deletion of GEF-H1 or disruption of microtubule function in macrophages does not prevent NF-κB activation by surface and endosomal TLRs involved in the detection of viral RNA and viral glycoproteins. GEF-H1 is required for IFN-β secretion by macrophages in response to infection by EMCV and influenza A and controls influenza A virus infection in mice.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–6 (PDF 7622 kb)
Rights and permissions
About this article
Cite this article
Chiang, HS., Zhao, Y., Song, JH. et al. GEF-H1 controls microtubule-dependent sensing of nucleic acids for antiviral host defenses. Nat Immunol 15, 63–71 (2014). https://doi.org/10.1038/ni.2766
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ni.2766