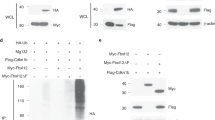
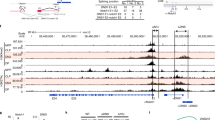
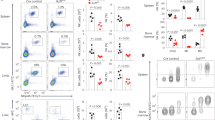
Abstract
Interleukin 7 (IL-7) has a critical role in the development of early CD4−CD8− double-negative (DN) thymocytes. Although the transcription factor STAT5 is an important component of IL-7 signaling, differences in the phenotypes of mice deficient in STAT5, IL-7, IL-7 receptor alpha (IL-7rα) or the kinase Jak3 suggest the existence of STAT5-independent IL-7 signaling. Here we found that IL-7–Jak3 signals activated the transcription factor NFATc1 in DN thymocytes by phosphorylating Tyr371 in the regulatory region of NFATc1. This NFAT-activation pathway was critical for the survival and development of DN thymocytes, as deficiency in NFATc1 blocked thymocyte development at the DN1 stage, leading to T cell lymphopenia. In addition, our results demonstrated a cooperative function for NFATc1 and STAT5 in guiding thymocyte development in response to IL-7 signals.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
References
Godfrey, D.I., Kennedy, J., Suda, T. & Zlotnik, A. A developmental pathway involving four phenotypically and functionally distinct subsets of CD3−CD4−CD8− triple-negative adult mouse thymocytes defined by CD44 and CD25 expression. J. Immunol. 150, 4244–4252 (1993).
Dudley, E.C., Petrie, H.T., Shah, L.M., Owen, M.J. & Hayday, A.C. T cell receptor β chain gene rearrangement and selection during thymocyte development in adult mice. Immunity 1, 83–93 (1994).
Graef, I.A., Chen, F. & Crabtree, G.R. NFAT signaling in vertebrate development. Curr. Opin. Genet. Dev. 11, 505–512 (2001).
Crabtree, G.R. & Olson, E.N. NFAT signaling: choreographing the social lives of cells. Cell 109 (suppl. 1), S67–S79 (2002).
Hogan, P.G., Chen, L., Nardone, J. & Rao, A. Transcriptional regulation by calcium, calcineurin, and NFAT. Genes Dev. 17, 2205–2232 (2003).
Macian, F. NFAT proteins: key regulators of T-cell development and function. Nat. Rev. Immunol. 5, 472–484 (2005).
Adachi, S., Amasaki, Y., Miyatake, S., Arai, N. & Iwata, M. Successive expression and activation of NFAT family members during thymocyte differentiation. J. Biol. Chem. 275, 14708–14716 (2000).
Oukka, M. et al. The transcription factor NFAT4 is involved in the generation and survival of T cells. Immunity 9, 295–304 (1998).
Yoshida, H. et al. The transcription factor NF-ATc1 regulates lymphocyte proliferation and Th2 cytokine production. Immunity 8, 115–124 (1998).
Xanthoudakis, S. et al. An enhanced immune response in mice lacking the transcription factor NFAT1. Science 272, 892–895 (1996).
Ranger, A.M., Oukka, M., Rengarajan, J. & Glimcher, L.H. Inhibitory function of two NFAT family members in lymphoid homeostasis and Th2 development. Immunity 9, 627–635 (1998).
Jiang, Q. et al. Cell biology of IL-7, a key lymphotrophin. Cytokine Growth Factor Rev. 16, 513–533 (2005).
Foxwell, B.M., Beadling, C., Guschin, D., Kerr, I. & Cantrell, D. Interleukin-7 can induce the activation of Jak1, Jak3 and STAT5 proteins in murine T cells. Eur. J. Immunol. 25, 3041–3046 (1995).
Lin, J.X. & Leonard, W.J. The role of Stat5a and Stat5b in signaling by IL-2 family cytokines. Oncogene 19, 2566–2576 (2000).
von Freeden-Jeffry, U. et al. Lymphopenia in interleukin (IL)-7 gene-deleted mice identifies IL-7 as a nonredundant cytokine. J. Exp. Med. 181, 1519–1526 (1995).
Peschon, J.J. et al. Early lymphocyte expansion is severely impaired in interleukin 7 receptor-deficient mice. J. Exp. Med. 180, 1955–1960 (1994).
Puel, A., Ziegler, S.F., Buckley, R.H. & Leonard, W.J. Defective IL7R expression in T−B+NK+ severe combined immunodeficiency. Nat. Genet. 20, 394–397 (1998).
DiSanto, J.P., Muller, W., Guy-Grand, D., Fischer, A. & Rajewsky, K. Lymphoid development in mice with a targeted deletion of the interleukin 2 receptor γ chain. Proc. Natl. Acad. Sci. USA 92, 377–381 (1995).
Cao, X. et al. Defective lymphoid development in mice lacking expression of the common cytokine receptor γ chain. Immunity 2, 223–238 (1995).
Nosaka, T. et al. Defective lymphoid development in mice lacking Jak3. Science 270, 800–802 (1995).
Park, S.Y. et al. Developmental defects of lymphoid cells in Jak3 kinase-deficient mice. Immunity 3, 771–782 (1995).
Thomis, D.C., Gurniak, C.B., Tivol, E., Sharpe, A.H. & Berg, L.J. Defects in B lymphocyte maturation and T lymphocyte activation in mice lacking Jak3. Science 270, 794–797 (1995).
Yao, Z. et al. Stat5a/b are essential for normal lymphoid development and differentiation. Proc. Natl. Acad. Sci. USA 103, 1000–1005 (2006).
Akashi, K., Kondo, M., von Freeden-Jeffry, U., Murray, R. & Weissman, I.L. Bcl-2 rescues T lymphopoiesis in interleukin-7 receptor-deficient mice. Cell 89, 1033–1041 (1997).
Maraskovsky, E. et al. Bcl-2 can rescue T lymphocyte development in interleukin-7 receptor-deficient mice but not in mutant rag-1−/− mice. Cell 89, 1011–1019 (1997).
Kondo, M., Akashi, K., Domen, J., Sugamura, K. & Weissman, I.L. Bcl-2 rescues T lymphopoiesis, but not B or NK cell development, in common γ chain-deficient mice. Immunity 7, 155–162 (1997).
Aifantis, I., Gounari, F., Scorrano, L., Borowski, C. & von Boehmer, H. Constitutive pre-TCR signaling promotes differentiation through Ca2+ mobilization and activation of NF-κB and NFAT. Nat. Immunol. 2, 403–409 (2001).
Patra, A.K. et al. PKB rescues calcineurin/NFAT-induced arrest of Rag expression and pre-T cell differentiation. J. Immunol. 177, 4567–4576 (2006).
Zhang, B.W. et al. T cell responses in calcineurin A α-deficient mice. J. Exp. Med. 183, 413–420 (1996).
Neilson, J.R., Winslow, M.M., Hur, E.M. & Crabtree, G.R. Calcineurin B1 is essential for positive but not negative selection during thymocyte development. Immunity 20, 255–266 (2004).
Goodman, P.A., Niehoff, L.B. & Uckun, F.M. Role of tyrosine kinases in induction of the c-jun proto-oncogene in irradiated B-lineage lymphoid cells. J. Biol. Chem. 273, 17742–17748 (1998).
Sudbeck, E.A. et al. Structure-based design of specific inhibitors of Janus kinase 3 as apoptosis-inducing antileukemic agents. Clin. Cancer Res. 5, 1569–1582 (1999).
Tibbles, H.E. et al. Role of a JAK3-dependent biochemical signaling pathway in platelet activation and aggregation. J. Biol. Chem. 276, 17815–17822 (2001).
Beals, C.R., Clipstone, N.A., Ho, S.N. & Crabtree, G.R. Nuclear localization of NF-ATc by a calcineurin-dependent, cyclosporin-sensitive intramolecular interaction. Genes Dev. 11, 824–834 (1997).
Aringer, M. et al. Characterization and analysis of the proximal Janus kinase 3 promoter. J. Immunol. 170, 6057–6064 (2003).
von Freeden-Jeffry, U., Solvason, N., Howard, M. & Murray, R. The earliest T lineage-committed cells depend on IL-7 for Bcl-2 expression and normal cell cycle progression. Immunity 7, 147–154 (1997).
Kim, K., Lee, C.K., Sayers, T.J., Muegge, K. & Durum, S.K. The trophic action of IL-7 on pro-T cells: inhibition of apoptosis of pro-T1, -T2, and -T3 cells correlates with Bcl-2 and Bax levels and is independent of Fas and p53 pathways. J. Immunol. 160, 5735–5741 (1998).
Kawamura, T. et al. Endothelin-1-dependent nuclear factor of activated T lymphocyte signaling associates with transcriptional coactivator p300 in the activation of the B cell leukemia-2 promoter in cardiac myocytes. Circ. Res. 94, 1492–1499 (2004).
Changelian, P.S. et al. The specificity of JAK3 kinase inhibitors. Blood 111, 2155–2157 (2008).
Bhattacharyya, S. et al. NFATc1 affects mouse splenic B cell function by controlling the calcineurin–NFAT signaling network. J. Exp. Med. 208, 823–839 (2011).
de Boer, J. et al. Transgenic mice with hematopoietic and lymphoid specific expression of Cre. Eur. J. Immunol. 33, 314–325 (2003).
Hosoya, T. et al. GATA-3 is required for early T lineage progenitor development. J. Exp. Med. 206, 2987–3000 (2009).
Li, L., Leid, M. & Rothenberg, E.V. An early T cell lineage commitment checkpoint dependent on the transcription factor Bcl11b. Science 329, 89–93 (2010).
Weber, B.N. et al. A critical role for TCF-1 in T-lineage specification and differentiation. Nature 476, 63–68 (2011).
Zhou, B. et al. Characterization of Nfatc1 regulation identifies an enhancer required for gene expression that is specific to pro-valve endocardial cells in the developing heart. Development 132, 1137–1146 (2005).
Schluns, K.S., Kieper, W.C., Jameson, S.C. & Lefrancois, L. Interleukin-7 mediates the homeostasis of naive and memory CD8 T cells in vivo. Nat. Immunol. 1, 426–432 (2000).
Kaech, S.M. et al. Selective expression of the interleukin 7 receptor identifies effector CD8 T cells that give rise to long-lived memory cells. Nat. Immunol. 4, 1191–1198 (2003).
Li, J., Huston, G. & Swain, S.L. IL-7 promotes the transition of CD4 effectors to persistent memory cells. J. Exp. Med. 198, 1807–1815 (2003).
Kondrack, R.M. et al. Interleukin 7 regulates the survival and generation of memory CD4 cells. J. Exp. Med. 198, 1797–1806 (2003).
Chuvpilo, S. et al. Alternative polyadenylation events contribute to the induction of NF-ATc in effector T cells. Immunity 10, 261–269 (1999).
Acknowledgements
We thank C. Linden for cell sorting; S.Y. Na, B. Kneitz and T. Hünig (University of Würzburg) for Rag1−/− and Il2−/− mice; A. Rao (La Jolla Institute for Allergy and Immunology) for Nfatc1fl/fl mice; D. Kioussis (Medical Research Council National Institute for Medical Research, London) for Vav-Cre mice; S. Chuvpilo (TheraMAB LLC, Moscow) for GST-NFATc1 constructs; R. Rost (Institute of Molecular Biology and Biochemistry, Graz, Austria) for NFATc1 expression vector; W.J. Leonard (US National Institutes of Health) for expression vectors for human IL-2Rγ and mouse IL-7Rα; J.J. O'Shea (US National Institutes of Health) for expression vectors for human Jak3; K. Hasegawa (Kyoto Medical Center) for the BCL2 luciferase construct; J.C. Zũniga-Pflücker (University of Toronto) for OP9 and OP9-DL1 cells; J. Schneider-Schaulies (University of Würzburg) for help in lentiviral transduction experiments; and members of the Serfling laboratory for comments on the manuscript. Supported by Deutsche Forschungsgemeinschaft (TRR52 to A.A. and E.S., and DFG-SFB854-TP9 to U.B.), the Wilhelm-Sander Foundation (E.S.) and the Mildred Scheel Foundation (E.S.).
Author information
Authors and Affiliations
Contributions
A.K.P. conceived of the project and designed and did the experiments; A.A. assisted in OP9-DL1 coculture experiments and GST-protein purification; R.P.Z. and A.S. did mass spectrometry analysis for mapping of phosphorylated tyrosines; T.S. helped with Il7r−/− mice; U.B. contributed to immunofluorescence staining and discussions; E.S. contributed to the study organization; and A.K.P. and E.S. wrote the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–7 and Table 1 (PDF 1004 kb)
Rights and permissions
About this article
Cite this article
Patra, A., Avots, A., Zahedi, R. et al. An alternative NFAT-activation pathway mediated by IL-7 is critical for early thymocyte development. Nat Immunol 14, 127–135 (2013). https://doi.org/10.1038/ni.2507
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ni.2507
This article is cited by
-
The Rho/MRTF pathway inhibitor CCG-222740 reduces stellate cell activation and modulates immune cell populations in KrasG12D; Pdx1-Cre (KC) mice
Scientific Reports (2019)
-
B cell development is critically dependent on NFATc1 activity
Cellular & Molecular Immunology (2019)
-
A threshold level of NFATc1 activity facilitates thymocyte differentiation and opposes notch-driven leukaemia development
Nature Communications (2016)
-
Cell cycle and apoptosis regulation by NFAT transcription factors: new roles for an old player
Cell Death & Disease (2016)
-
Transcription factors and target genes of pre-TCR signaling
Cellular and Molecular Life Sciences (2015)