Abstract
Improved understanding of the pathogenesis of type 1 diabetes mellitus has completely changed our view of this disease in the past 25 years—from an acute, fulminant disease, to a chronic, autoimmune process. Information on genetic and serologic markers has increased our ability to identify individuals at risk. Prospectively gathered data indicate that, with a combination of immunologic and metabolic studies, children with a 6-year risk of disease higher than 90% can be identified due to an ongoing immune process. They differ from children with overt disease only in the time it will take for glucose levels to rise above a diagnostic threshold. Therapies to change the progression of β-cell loss have been tested in patients with newly diagnosed type 1 diabetes. With improved predictive capabilities and agents that can have longer-lasting effects than those tested more than 10 years ago, new prevention studies are underway. These studies are large and costly but the risks posed by such interventions compare favorably with those of developing hyperglycemia and of future complications portended by the diagnosis of diabetes. In this Review we discuss risk-stratification techniques and how they are applied, other diagnostic criteria, and outcomes from diabetes-prevention trials.
Key Points
-
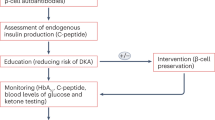
Screening of relatives with type 1 diabetes can identify individuals with an extraordinarily high risk of developing overt type 1 diabetes mellitus within 5–6 years
-
Diagnostic criteria for diabetes are not associated with the disease process but rather with glycemic levels associated with the development of complications
-
Prevention trials will target individuals with greater functional β-cell mass, who have not yet demonstrated overt hyperglycemia but who are at high risk of disease development
-
Future prevention trials will include the use of therapies that are antigen specific or antigen nonspecific, or that are used in combination
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout

Similar content being viewed by others
References
Atkinson MA (2005) ADA Outstanding Scientific Achievement Lecture 2004. Thirty years of investigating the autoimmune basis for type 1 diabetes: why can't we prevent or reverse this disease? Diabetes 54: 1253–1263
Eisenbarth GS (1986) Type I diabetes mellitus. A chronic autoimmune disease. N Engl J Med 314: 1360–1368
Palmer JP et al. (2004) C-peptide is the appropriate outcome measure for type 1 diabetes clinical trials to preserve beta-cell function: report of an ADA workshop, 21-22 October 2001. Diabetes 53: 250–264
Steffes MW et al. (2003) beta-Cell function and the development of diabetes-related complications in the diabetes control and complications trial. Diabetes Care 26: 832–836
Bougneres PF et al. (1988) Factors associated with early remission of type I diabetes in children treated with cyclosporine. N Engl J Med 318: 663–670
Silverstein J et al. (1988) Immunosuppression with azathioprine and prednisone in recent-onset insulin-dependent diabetes mellitus. N Engl J Med 319: 599–604
Keymeulen B et al. (2005) Insulin needs after CD3-antibody therapy in new-onset type 1 diabetes. N Engl J Med 352: 2598–2608
Herold KC et al. (2002) Anti-CD3 monoclonal antibody in new-onset type 1 diabetes mellitus. N Engl J Med 346: 1692–1698
Herold KC et al. (2005) A single course of anti-CD3 monoclonal antibody hOKT3γ1(Ala-Ala) results in improvement in C-peptide responses and clinical parameters for at least 2 years after onset of type 1 diabetes. Diabetes 54: 1763–1769
Hagopian WA et al. (1995) Glutamate decarboxylase-, insulin-, and islet cell-antibodies and HLA typing to detect diabetes in a general population-based study of Swedish children. J Clin Invest 95: 1505–1511
Eisenbarth GS et al. (1998) Dual-parameter model for prediction of type I diabetes mellitus. Proc Assoc Am Physicians 110: 126–135
Verge CF et al. (1996) Prediction of type I diabetes in first-degree relatives using a combination of insulin, GAD, and ICA512bdc/IA-2 autoantibodies. Diabetes 45: 926–933
Bingley PJ (1996) Interactions of age, islet cell antibodies, insulin autoantibodies, and first-phase insulin response in predicting risk of progression to IDDM in ICA+ relatives: the ICARUS data set. Islet Cell Antibody Register Users Study. Diabetes 45: 1720–1728
Barker JM et al. (2004) Prediction of autoantibody positivity and progression to type 1 diabetes: Diabetes Autoimmunity Study in the Young (DAISY). J Clin Endocrinol Metab 89: 3896–3902
Merriman TR and Todd JA (1995) Genetics of autoimmune disease. Curr Opin Immunol 7: 786–792
Todd JA et al. (1987) HLA-DQβ gene contributes to susceptibility and resistance to insulin dependent diabetes mellitus. Nature 329: 599–604
Atkinson MA and Eisenbarth GS (2001) Type 1 diabetes: new perspectives on disease pathogenesis and treatment. Lancet 358: 221–229
Todd JA et al. (2007) Robust associations of four new chromosome regions from genome-wide analyses of type 1 diabetes. Nat Genet 39: 857–864
Rewers M et al. (1996) Beta-cell autoantibodies in infants and toddlers without IDDM relatives: diabetes autoimmunity study in the young (DAISY). J Autoimmun 9: 405–410
Rewers M et al. (1996) Newborn screening for HLA markers associated with IDDM: diabetes autoimmunity study in the young (DAISY). Diabetologia 39: 807–812
Lambert AP et al. (2004) Absolute risk of childhood-onset type 1 diabetes defined by human leukocyte antigen class II genotype: a population-based study in the United Kingdom. J Clin Endocrinol Metab 89: 4037–4043
Bell GI et al. (1984) A polymorphic locus near the human insulin gene is associated with insulin dependent diabetes mellitus. Diabetes 33: 2176–2183
Bottini N et al. (2004) A functional variant of lymphoid tyrosine phosphatase is associated with type 1 diabetes. Nature 36: 337–338
Nejentsev S et al. (2007) Localization of type 1 diabetes susceptibility to the MHC class I genes HLA-B and HLA-A. Nature 450: 887–892
Yu L et al. (1996) Anti-islet autoantibodies usually develop sequentially rather than simultaneously. J Clin Endocrinol Metab 81: 4264–4267
Yu L et al. (2001) Expression of GAD65 and islet cell antibody (ICA512) autoantibodies among cytoplasmic ICA+ relatives is associated with eligibility for the Diabetes Prevention Trial-Type 1. Diabetes 50: 1735–1740
Irvine WJ et al. (1976) Pancreatic islet-cell antibody as a marker for asymptomatic and latent diabetes and prediabetes. Lancet 2: 1097–1102
Landin-Olsson M (1990) Precision of the islet-cell antibody assay depends on the pancreas. J Clin Lab Anal 4: 289–294
Wenzlau JM et al. (2007) The cation efflux transporter ZnT8 (Slc30A8) is a major autoantigen in human type 1 diabetes. Proc Natl Acad Sci U S A 104: 17040–17045
Krischer JP et al. (2003) Screening strategies for the identification of multiple antibody-positive relatives of individuals with type 1 diabetes. J Clin Endocrinol Metab 88: 103–108
Achenbach P et al. (2006) Type 1 diabetes risk assessment: improvement by follow-up measurements in young islet autoantibody-positive relatives. Diabetologia 49: 2969–2976
Bingley PJ et al. (1997) Prediction of IDDM in the general population: strategies based on combinations of autoantibody markers. Diabetes 46: 1701–1710
Sosenko JM et al. (2006) Patterns of metabolic progression to type 1 diabetes in the Diabetes Prevention Trial-Type 1. Diabetes Care 29: 643–649
Tsai EB et al. (2006) The rise and fall of insulin secretion in type 1 diabetes mellitus. Diabetologia 49: 261–270
Sosenko JM et al. (2007) Increasing the accuracy of oral glucose tolerance testing and extending its application to individuals with normal glucose tolerance for the prediction of type 1 diabetes: the Diabetes Prevention Trial-Type 1. Diabetes Care 30: 38–42
Xu P et al. (2007) Role of insulin resistance in predicting progression to type 1 diabetes. Diabetes Care 30: 2314–2320
Sosenko JM et al. (2007) A risk score for type 1 diabetes derived from autoantibody positive participants in the diabetes prevention trial-type 1. Diabetes Care 31: 528–533
Stiller CR et al. (1984) Effects of cyclosporine immunosuppression in insulin-dependent diabetes mellitus of recent onset. Science 223: 1362–1367
Stiller CR et al. (1987) Effects of cyclosporine in recent-onset juvenile type 1 diabetes: impact of age and duration of disease. J Pediatr 111: 1069–1072
Carel JC et al. (1996) Cyclosporine delays but does not prevent clinical onset in glucose intolerant pre-type 1 diabetic children. J Autoimmun 9: 739–745
Parving HH et al. (1999) Cyclosporine nephrotoxicity in type 1 diabetic patients. A 7-year follow-up study. Diabetes Care 22: 478–483
Gale EA et al. (2004) European Nicotinamide Diabetes Intervention Trial (ENDIT): a randomized controlled trial of intervention before the onset of type 1 diabetes. Lancet 363: 925–931
Gale EA (2003) Intervening before the onset of type 1 diabetes: baseline data from the European Nicotinamide Diabetes Intervention Trial (ENDIT). Diabetologia 46: 339–346
Diabetes Prevention Trial: Type 1 Diabetes Study Group (2002) Effects of insulin in relatives of patients with type 1 diabetes mellitus. N Engl J Med 346: 1685–1691
Skyler JS et al. (2005) Effects of oral insulin in relatives of patients with type 1 diabetes: The Diabetes Prevention Trial—Type 1. Diabetes Care 28: 1068–1076
Zhang ZJ et al. (1991) Suppression of diabetes in nonobese diabetic mice by oral administration of porcine insulin. Proc Natl Acad Sci U S A 88: 10252–10256
Yamada K et al. (1982) Preventive and therapeutic effects of large-dose nicotinamide injections on diabetes associated with insulitis. An observation in nonobese diabetic (NOD) mice. Diabetes 31: 749–753
Pozzilli P et al. (1996) Meta-analysis of nicotinamide treatment in patients with recent-onset IDDM. The Nicotinamide Trialists. Diabetes Care 19: 1357–1363
Keller RJ et al. (1993) Insulin prophylaxis in individuals at high risk of type I diabetes. Lancet 341: 927–928
Pociot F (1993) Nicotinamide-biological actions and therapeutic potential in diabetes prevention. Diabetologia 36: 574–576
National Diabetes Data Group (1979) Classification and diagnosis of diabetes mellitus and other categories of glucose intolerance. Diabetes 28: 1039–1057
Expert Committee on the Diagnosis and Classification of Diabetes Mellitus (2003) Report of the Expert Committee on the Diagnosis and Classification of Diabetes Mellitus. Diabetes Care 26: 3160–3167
Expert Committee on the Diagnosis and Classification of Diabetes Mellitus (2007) Report of the Expert Committee on the Diagnosis and Classification of Diabetes Mellitus. Diabetes Care 30 (Suppl): S42–S47
Sherry NA et al. (2006) Effects of autoimmunity and immune therapy on beta-cell turnover in type 1 diabetes. Diabetes 55: 3238–3245
The Diabetes Control and Complications Trial Research Group (1993) The effect of intensive treatment of diabetes on the development and progression of long-term complications in insulin-dependent diabetes mellitus. N Engl J Med 329: 977–986
Lehmann PV et al. (1992) Spreading of T-cell autoimmunity to cryptic determinants of an autoantigen. Nature 358: 155–157
Rish R et al. (1999) Induction of glutamic acid decarboxylase 65-specific Th2 cells and suppression of autoimmune diabetes at late stages of disease is epitope dependent. J Immunol 163: 1178–1187
Agardh C et al. (2005) Clinical evidence for the safety of GAD65 immunomodulation in adult onset autoimmune diabetes. J Diabetes Complications 19: 238–246
Hu CY et al. (2007) Treatment with CD20-specific antibody prevents and reverses autoimmune diabetes in mice. J Clin Invest 117: 3857–3867
Bresson D et al. (2006) Anti-CD3 and nasal proinsulin combination therapy enhances remission from recent-onset autoimmune diabetes by inducing Tregs. J Clin Invest 116: 1371–1381
Nir T et al. (2007) Recovery from diabetes in mice by beta cell regeneration. J Clin Invest 117: 2553–2561
Dor Y et al. (2004) Adult pancreatic beta-cells are formed by self-duplication rather than stem-cell differentiation. Nature 429: 41–46
Sherry N et al. (2007) Exendin-4 improves reversal of diabetes in NOD mice treated with anti-CD3 mAb by enhancing recovery of β cells. Endocrinology 148: 5136–5144
Hadjiyanni I et al. (2008) Exendin-4 modulates diabetes onset in non obese diabetic mice. Endocrinology 149: 1338–1349
Fourlanos S et al. (2004) Insulin resistance is a risk factor for progression to type 1 diabetes. Diabetologia 47: 1661–1667
Bingley PJ and Gale EA (2006) Progression to type 1 diabetes in islet cell antibody-positive relatives in the European Nicotinamide Diabetes Intervention Trial: the role of additional immune, genetic and metabolic markers of risk. Diabetologia 49: 881–890
Achenbach P et al. (2004) Stratification of type 1 diabetes risk on the basis of islet autoantibody characteristics. Diabetes 53: 384–392
Dahlquist G and Gothefors L (1995) The cumulative incidence of childhood diabetes mellitus in Sweden unaffected by BCG-vaccination. Diabetologia 38: 873–874
Parent ME et al. (1997) Bacille Calmette-Guerin vaccination and incidence of IDDM in Montreal, Canada. Diabetes Care 20: 767–772
Huppmann M et al. (2005) Neonatal Bacille Calmette-Guerin vaccination and type 1 diabetes. Diabetes Care 28: 1204–1206
Bohmer KP et al. (1994) Linear loss of insulin secretory capacity during the last six months preceding IDDM. No effect of antiedematous therapy with ketotifen. Diabetes Care 17: 138–141
Hummel M et al. (2002) Elimination of dietary gluten does not reduce titers of type 1 diabetes-associated autoantibodies in high-risk subjects. Diabetes Care 25: 1111–1116
Lampeter EF et al. (1998) The Deutsche Nicotinamide Intervention Study: an attempt to prevent type 1 diabetes. DENIS Group. Diabetes 47: 980–984
Kupila A et al. (2003) Intranasally administered insulin intended for prevention of type 1 diabetes—a safety study in healthy adults. Diabetes Metab Res Rev 19: 415–420
TRIGR Study Group (2007) Study design of the Trial to Reduce IDDM in the Genetically at Risk (TRIGR). Pediatr Diabetes 8: 117–137
Acknowledgements
K Herold was awarded grants from the NIH, the Juvenile Diabetes Research Foundation, and the Brehm Foundation to support work related to this article.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
About this article
Cite this article
Sherr, J., Sosenko, J., Skyler, J. et al. Prevention of type 1 diabetes: the time has come. Nat Rev Endocrinol 4, 334–343 (2008). https://doi.org/10.1038/ncpendmet0832
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ncpendmet0832
This article is cited by
-
Microparticles bearing encephalitogenic peptides induce T-cell tolerance and ameliorate experimental autoimmune encephalomyelitis
Nature Biotechnology (2012)
-
Immunomodulatory therapy to preserve pancreatic β-cell function in type 1 diabetes
Nature Reviews Drug Discovery (2011)
-
A Short Pulse of IL-4 Delivered by DCs Electroporated With Modified mRNA Can Both Prevent and Treat Autoimmune Diabetes in NOD Mice
Molecular Therapy (2010)
-
Four-year metabolic outcome of a randomised controlled CD3-antibody trial in recent-onset type 1 diabetic patients depends on their age and baseline residual beta cell mass
Diabetologia (2010)
-
I marker di efficacia nel disegno dei trial immunoterapici: le lezioni emergenti dal diabete di tipo 1
L'Endocrinologo (2010)