Abstract
Glucocorticoids exert their effects in target tissues predominantly through their interaction with the glucocorticoid receptor, a member of the nuclear receptor superfamily of transcription factors. Over the years many studies have linked hormone responsiveness, both in vitro and in vivo, to the levels of both glucocorticoid and glucocorticoid receptor; furthermore, an impact of glucocorticoid receptor subcellular trafficking on hormone response has been revealed. This review will focus on the molecular mechanisms responsible for the regulation of glucocorticoid receptor trafficking and expression, and will highlight work that revealed selective physiological effects of altered glucocorticoid receptor expression. The role of alterations in glucocorticoid levels and glucocorticoid receptor function in the metabolic syndrome will also be discussed.
Key Points
-
The glucocorticoid receptor shuttles between the nuclear and cytoplasmic compartments and uses distinct transport proteins for this trafficking
-
Degradation of glucocorticoid receptor protein by the ubiquitin–proteasome protein degradation system influences the steady-state level of receptor protein, and can be stimulated in some cells by hormone binding to the receptor
-
The absolute level of glucocorticoid receptor protein expressed in a given cell can have a dramatic impact on cellular responsiveness to glucocorticoids, and can ultimately influence a wide range of physiological responses to glucocorticoids
-
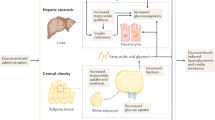
Accumulating data suggest that regulation of intracellular cortisol levels influences the pathogenesis of the metabolic syndrome
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Chrousos GP and Kino T (2005) Intracellular glucocorticoid signaling: a formerly simple system turns stochastic. Sci STKE 2005 (304): pe48
Zitt MJ (2005) Properties of ideal corticosteroid therapy. Allergy Asthma Proc 26: 173–182
Uings IJ and Farrow SN (2005) A pharmacological approach to enhancing the therapeutic index of corticosteroids in airway inflammatory disease. Curr Opin Pharmacol 5: 221–226
Walker BR et al. (2000) Independent effects of obesity and cortisol in predicting cardiovascular risk factors in men and women. J Intern Med 247: 198–204
Hollenberg SM et al. (1985) Primary structure and expression of a functional human glucocorticoid receptor cDNA. Nature 318: 635–641
Bamberger CM et al. (1995) Glucocorticoid receptor β, a potential endogenous inhibitor of glucocorticoid action in humans. J Clin Invest 95: 2435–2441
Zhou J and Cidlowski JA (2005) The human glucocorticoid receptor: one gene, multiple proteins and diverse responses. Steroids 70: 407–417
Lu NZ and Cidlowski JA (2005) Translational regulatory mechanisms generate N-terminal glucocorticoid receptor isoforms with unique transcriptional target genes. Mol Cell 18: 331–342
Mangelsdorf DJ et al. (1995) The nuclear receptor superfamily: the second decade. Cell 83: 835–839
Claessens F and Gewirth DT (2004) DNA recognition by nuclear receptors. Essays Biochem 40: 59–72
Yang-Yen HF et al. (1990) Transcriptional interference between c-Jun and the glucocorticoid receptor: mutual inhibition of DNA binding due to direct protein-protein interaction. Cell 62: 1205–1215
Rosenfeld MG et al. (2006) Sensors and signals: a coactivator/corepressor/epigenetic code for integrating signal-dependent programs of transcriptional response. Genes Dev 20: 1405–1428
Picard D and Yamamoto KR (1987) Two signals mediate hormone-dependent nuclear localization of the glucocorticoid receptor. EMBO J 6: 3333–3340
DeFranco DB et al. (1995) Nucleocytoplasmic shuttling of steroid receptors. Vitam Horm 51: 315–338
Silverstein AM et al. (1999) Different regions of the immunophilin FKBP52 determine its association with the glucocorticoid receptor, hsp90, and cytoplasmic dynein. J Biol Chem 274: 36980–36986
Wakui H et al. (1997) Interaction of the ligand-activated glucocorticoid receptor with the 14-3-3 η protein. J Biol Chem 272: 8153–8156
Schaaf MJ and Cidlowski JA (2002) Molecular mechanisms of glucocorticoid action and resistance. J Steroid Biochem Mol Biol 83: 37–48
Matthews JG et al. (2004) Defective glucocorticoid receptor nuclear translocation and altered histone acetylation patterns in glucocorticoid-resistant patients. J Allergy Clin Immunol 113: 1100–1108
Yamamoto KR et al. (1974) Receptors from glucocorticoid-sensitive lymphoma cells and two classes of insensitive clones: physical and DNA-binding properties. Proc Natl Acad Sci USA 71: 3901–3905
Irusen E et al. (2002) p38 Mitogen-activated protein kinase-induced glucocorticoid receptor phosphorylation reduces its activity: role in steroid-insensitive asthma. J Allergy Clin Immunol 109: 649–657
Itoh M et al. (2002) Nuclear export of glucocorticoid receptor is enhanced by c-Jun N-terminal kinase-mediated phosphorylation. Mol Endocrinol 16: 2382–2392
Lee H and Bai W (2002) Regulation of estrogen receptor nuclear export by ligand-induced and p38-mediated receptor phosphorylation. Mol Cell Biol 22: 5835–5845
Holaska JM et al. (2002) Ca2+-dependent nuclear export mediated by calreticulin. Mol Cell Biol 22: 6286–6297
Webster JC and Cidlowski JA (1994) Downregulation of the glucocorticoid receptor. A mechanism for physiological adaptation to hormones. Ann NY Acad Sci 746: 216–220
Ramdas J et al. (1999) Glucocorticoid-induced cell death requires autoinduction of glucocorticoid receptor expression in human leukemic T cells. Cancer Res 59: 1378–1385
Wallace AD and Cidlowski JA (2001) Proteasome-mediated glucocorticoid receptor degradation restricts transcriptional signaling by glucocorticoids. J Biol Chem 276: 42714–42721
Voges D et al. (1999) The 26S proteasome: a molecular machine designed for controlled proteolysis. Annu Rev Biochem 68: 1015–1068
Ciechanover A et al. (2000) Ubiquitin-mediated proteolysis: biological regulation via destruction. Bioessays 22: 442–451
Sengupta S and Wasylyk B (2001) Ligand-dependent interaction of the glucocorticoid receptor with p53 enhances their degradation by Hdm2. Genes Dev 15: 2367–2380
Kinyamu HK and Archer TK (2003) Estrogen receptor-dependent proteasomal degradation of the glucocorticoid receptor is coupled to an increase in mdm2 protein expression. Mol Cell Biol 23: 5867–5881
Connell P et al. (2001) The co-chaperone CHIP regulates protein triage decisions mediated by heat-shock proteins. Nat Cell Biol 3: 93–96
Wang X and DeFranco DB (2005) Alternative effects of the ubiquitin-proteasome pathway on glucocorticoid receptor down-regulation and transactivation are mediated by CHIP, an E3 ligase. Mol Endocrinol 19: 1474–1482
Ghosh B et al. (2000) Glucocorticoid receptor regulation in the rat embryo: a potential site for developmental toxicity? Toxicol Appl Pharmacol 164: 221–229
Wang X et al. (2002) Glucocorticoid receptors in hippocampal neurons that do not engage proteasomes escape from hormone-dependent down-regulation but maintain transactivation activity. Mol Endocrinol 16: 1987–1998
Reichardt HM et al. (2000) Mice with an increased glucocorticoid receptor gene dosage show enhanced resistance to stress and endotoxic shock. Mol Cell Biol 20: 9009–9017
Ridder S et al. (2005) Mice with genetically altered glucocorticoid receptor expression show altered sensitivity for stress-induced depressive reactions. J Neurosci 25: 6243–6250
Boyle MP et al. (2005) Acquired deficit of forebrain glucocorticoid receptor produces depression-like changes in adrenal axis regulation and behavior. Proc Natl Acad Sci USA 102: 473–478
Wei Q et al. (2004) Glucocorticoid receptor overexpression in forebrain: a mouse model of increased emotional lability. Proc Natl Acad Sci USA 101: 11851–11856
Liggins GC and Howie RN (1972) A controlled trial of antepartum glucocorticoid treatment for prevention of the respiratory distress syndrome in premature infants. Pediatrics 50: 515–525
Yeh TF et al. (2004) Outcomes at school age after postnatal dexamethasone therapy for lung disease of prematurity. N Engl J Med 350: 1304–1313
Friedman TC et al. (1996) Carbohydrate metabolism in endogenous hypercortisolism: shared features with metabolic syndrome X and NIDDM. Endocr J 43: 645–655
Lyon CJ et al. (2003) Adiposity, inflammation, and atherosclerosis. Endocrinology 144: 2195–2200
Xu H et al. (2003) Chronic inflammation in fat plays a crucial role in the development of obesity-related insulin resistance. J Clin Invest 112: 1821–1830
Dandona P et al. (2004) Inflammation: the link between insulin resistance, obesity and diabetes. Trends Immunol 25: 4–7
Fain JN et al. (2004) Comparison of the release of adipocytokines by adipose tissue, adipose tissue matrix, and adipocytes from visceral and subcutaneous abdominal adipose tissues of obese humans. Endocrinology 145: 2273–2282
Arkan MC et al. (2005) IKK-β links inflammation to obesity-induced insulin resistance. Nat Med 11: 191–198
Opherk C et al. (2004) Inactivation of the glucocorticoid receptor in hepatocytes leads to fasting hypoglycemia and ameliorates hyperglycemia in streptozotocin-induced diabetes mellitus. Mol Endocrinol 18: 1346–1353
Garvey WT et al. (1989) Dexamethasone regulates the glucose transport system in primary cultured adipocytes: different mechanisms of insulin resistance after acute and chronic exposure. Endocrinology 124: 2063–2073
Gremlich S et al. (1997) Dexamethasone induces posttranslational degradation of GLUT2 and inhibition of insulin secretion in isolated pancreatic β cells. Comparison with the effects of fatty acids. J Biol Chem 272: 3216–3222
Davani B et al. (2004) Aged transgenic mice with increased glucocorticoid sensitivity in pancreatic β-cells develop diabetes. Diabetes 53 (Suppl 1): S51–S59
Gesina E et al. (2004) Dissecting the role of glucocorticoids on pancreas development. Diabetes 53: 2322–2329
Seckl JR (2004) 11β-hydroxysteroid dehydrogenases: changing glucocorticoid action. Curr Opin Pharmacol 4: 597–602
Whorwood CB et al. (2002) Increased glucocorticoid receptor expression in human skeletal muscle cells may contribute to the pathogenesis of the metabolic syndrome. Diabetes 51: 1066–1075
Wake DJ et al. (2003) Local and systemic impact of transcriptional up-regulation of 11β-hydroxysteroid dehydrogenase type 1 in adipose tissue in human obesity. J Clin Endocrinol Metab 88: 3983–3988
Kannisto K et al. (2004) Overexpression of 11β-hydroxysteroid dehydrogenase-1 in adipose tissue is associated with acquired obesity and features of insulin resistance: studies in young adult monozygotic twins. J Clin Endocrinol Metab 89: 4414–4421
Tomlinson JW et al. (2001) Regulation of expression of 11β-hydroxysteroid dehydrogenase type 1 in adipose tissue: tissue-specific induction by cytokines. Endocrinology 142: 1982–1989
Holmes MC et al. (2001) Phenotypic analysis of mice bearing targeted deletions of 11β-hydroxysteroid dehydrogenases 1 and 2 genes. Mol Cell Endocrinol 71: 15–20
Morton NM et al. (2001) Improved lipid and lipoprotein profile, hepatic insulin sensitivity, and glucose tolerance in 11β-hydroxysteroid dehydrogenase type 1 null mice. J Biol Chem 276: 41293–41300
Hermanowski-Vosatka A et al. (2005) 11β-HSD1 inhibition ameliorates metabolic syndrome and prevents progression of atherosclerosis in mice. J Exp Med 202: 517–527
Masuzaki H et al. (2003) Transgenic amplification of glucocorticoid action in adipose tissue causes high blood pressure in mice. J Clin Invest 112: 83–90
Paterson JM et al. (2004) Metabolic syndrome without obesity: Hepatic overexpression of 11β-hydroxysteroid dehydrogenase type 1 in transgenic mice. Proc Natl Acad Sci USA 101: 7088–7093
Kershaw EE et al. (2005) Adipocyte-specific glucocorticoid inactivation protects against diet-induced obesity. Diabetes 54: 1023–1031
Morton NM et al. (2005) A polygenic model of the metabolic syndrome with reduced circulating and intra-adipose glucocorticoid action. Diabetes 54: 3371–3378
Walker BR and Seckl JR (2003) 11β-hydroxysteroid dehydrogenase type 1 as a novel therapeutic target in metabolic and neurodegenerative disease. Expert Opin Ther Target 7: 771–783
van Rossum EF and Lamberts SW (2004) Polymorphisms in the glucocorticoid receptor gene and their associations with metabolic parameters and body composition. Recent Prog Horm Res 59: 333–357
van Rossum EF et al. (2002) A polymorphism in the glucocorticoid receptor gene, which decreases sensitivity to glucocorticoids in vivo, is associated with low insulin and cholesterol levels. Diabetes 51: 3128–3134
Russcher H et al. (2005) Increased expression of the glucocorticoid receptor—a translational isoform as a result of the ER22/23EK polymorphism. Mol Endocrinol 19: 1687–1696
Rosmond R (2002) The glucocorticoid receptor gene and its association to metabolic syndrome. Obes Res 10: 1078–1086
Phillips DIW (1998) Birth weight and the future development of diabetes. Diabetes Care 21: B150–B155
Reynolds RM et al. (2001) Altered control of cortisol secretion in adult men with low birth weight and cardiovascular risk factors. J Clin Endocrinol Metab 86: 245–250
Dave-Sharma S et al. (1998) Extensive personal experience—examination of genotype and phenotype relationships in 14 patients with apparent mineralocorticoid excess. J Clin Endocrinol Metab 83: 2244–2254
French NP and Hagan R et al. (2004) Repeated antenatal corticosteroids: effects on cerebral palsy and childhood behavior. Am J Obstet Gynecol 190: 588–595
Nyirenda MF et al. (1998) Glucocorticoid exposure in late gestation permanently programs rat hepatic phosphoenol-pyruvate carboxykinase and glucocorticoid receptor expression and causes glucose intolerance in adult offspring. J Clin Invest 101: 2174–2181
Cleasby ME et al. (2003) Programming of rat muscle and fat metabolism by in utero overexposure to glucocorticoids. Endocrinology 144: 999–1007
Boullu-Ciocca S et al. (2005) Postnatal diet-induced obesity in rats upregulates systemic and adipose tissue glucocorticoid metabolism during development and adulthood. Diabetes 54: 197–203
Elbi C et al. (2004) Molecular chaperones function as steroid receptor nuclear mobility factors. Proc Natl Acad Sci USA 101: 2876–2881
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
About this article
Cite this article
Witchel, S., DeFranco, D. Mechanisms of Disease: regulation of glucocorticoid and receptor levels—impact on the metabolic syndrome. Nat Rev Endocrinol 2, 621–631 (2006). https://doi.org/10.1038/ncpendmet0323
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/ncpendmet0323
This article is cited by
-
Role of glucocorticoid receptor phosphorylation-mediated synaptic plasticity in anxiogenic and depressive behaviors induced by monosodium glutamate
Naunyn-Schmiedeberg's Archives of Pharmacology (2021)
-
Circadian clocks and energy metabolism
Cellular and Molecular Life Sciences (2014)
-
High-end normal adrenocorticotropic hormone and cortisol levels are associated with specific cardiovascular risk factors in pediatric obesity: a cross-sectional study
BMC Medicine (2013)
-
Impaired nuclear translocation of glucocorticoid receptors: novel findings from psoriatic epidermal keratinocytes
Cellular and Molecular Life Sciences (2013)