Abstract
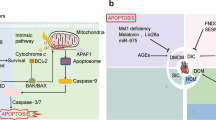
Background Despite widespread activation of proapoptotic stimuli and mediators, the degree of apoptosis in failing hearts is not very high. Endogenous antiapoptotic mechanisms are thought to be triggered by the heart-failure process. We investigated whether activation of endogenous apoptosis inhibitors plays a part when death receptor and mitochondrial apoptotic pathways have been triggered.
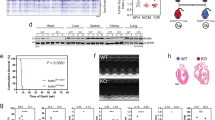
Methods We evaluated various proapoptotic and antiapoptotic factors in myocardial tissue specimens obtained from normal and explanted end-stage ischemic and dilated cardiomyopathic hearts. Caspases (CASPs) 3, 8 and 9, total and activated Bcl-2 homology domain 3-interacting domain death agonist, the X-linked inhibitor of apoptosis (XIAP), and DNA fragmentation factor (DFF) proteins were analyzed by western blotting. Expression of messenger RNA was measured by reverse-transcription polymerase chain reaction for the XIAP, DIABLO, CFLAR and DFF genes. We also assessed CASP3, CASP8 and CASP9 and DFF activity. Cytochrome c1 localization in myocytes was analyzed by immunohistochemistry and immunoelectron microscopy.
Results We collected myocardial tissue from eight cardiomyopathic hearts and five normal hearts. Cytochrome c1 was released from mitochondria into the cytosol in the cardiomyopathic hearts but CASP9 was not activated. CASP8 activity was increased compared with that in normal myocardium. Although CASP3 was cleaved, activity was not greatly increased because of an increase in XIAP and decrease in DIABLO expression. DFF proteins were conspicuously absent.
Conclusions Concurrent upregulation of endogenous antiapoptotic mechanisms can interrupt the apoptotic cascade and prevents cell loss despite the presence of multiple proapoptotic factors. This period might offer a therapeutic window for restoration of myocardial function in heart failure.
Key Points
-
Apoptosis plays an important part in heart failure, and release of cytochrome c1 release from mitochondria to cytoplasm is a prominent feature
-
Although the apoptotic cascade is initiated in heart failure, it might not complete (so-called apoptosis interruptus)
-
Interruption of apoptotic process is most likely due to concurrent upregulation of antiapoptotic and proapoptotic factors, while AIFM1 and DFF concentrations are substantially reduced
-
Interrupted apoptosis suggests that heart failure could often represent a reversible disease state and comprise an attractive therapeutic target
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout








Similar content being viewed by others
References
Narula J et al. (1996) Apoptosis in myocytes in end-stage heart failure. N Engl J Med 335: 1182–1189
Olivetti G et al. (1997) Apoptosis in the failing human heart. N Engl J Med 336: 1131–1141
Narula J et al. (1999) Apoptosis in heart failure: release of cytochrome c from mitochondria and activation of caspase-3 in human cardiomyopathy. Proc Natl Acad Sci USA 96: 8144–8149
Narula J et al. (2006) Mechanisms of Disease: apoptosis in heart failure—seeing hope in death. Nat Clin Pract Cardiovasc Med 3: 681–688
Kanoh M et al. (1999) Significance of myocytes with positive DNA in situ nick end-labeling (TUNEL) in hearts with dilated cardiomyopathy: not apoptosis but DNA repair. Circulation 99: 2757–2764
Narula N et al. (2005) Is the myofibrillarlytic myocyte a forme fruste apoptotic myocyte? Ann Thorac Surg 79: 1333–1337
Reed JC and Paternostro G (1999) Postmitochondrial regulation of apoptosis during heart failure. Proc Natl Acad Sci U S A 96: 7614–7616
Kietselaer BL et al. (2007) Noninvasive detection of programmed cell loss with 99mTc-labeled annexin A5 in heart failure. J Nucl Med 48: 562–567
Quantity One® version 4.1.1. (Bio-rad, Hercules, CA)
Scheubel RJ et al. (2002) Apoptotic pathway activation from mitochondria and death receptors without caspase-3 cleavage in failing human myocardium: fragile balance of myocyte survival? J Am Coll Cardiol 39: 481–488
Haider NKS et al. (1999) Caspase 3 mediated cleavage of troponin c at evolutionary conserved calcium binding site: Relevance of apoptosis in heart failure. Circulation 100 (Suppl 1): I-283
Gu J et al. (1999) Functional interaction of DFF35 and DFF45 with caspase-activated DNA fragmentation nuclease DFF40. J Biol Chem 274: 20759–20762
Datta R et al. (2000) XIAP regulates DNA damage-induced apoptosis downstream of caspase-9 cleavage. J Biol Chem 275: 31733–31738
Deveraux QL et al. (1998) IAPs block apoptotic events induced by caspase-8 and cytochrome c by direct inhibition of distinct caspases. EMBO J 17: 2215–2223
Salvesen GS and Duckett CS (2002) IAP proteins: blocking the road to death's door. Nat Rev Mol Cell Biol 3: 401–410
Han Z et al. (1997) A sequential two-step mechanism for the production of the mature p17:p12 form of caspase-3 in vitro. J Biol Chem 272: 13432–13436
von Coelln R et al. (2001) Rescue from death but not from functional impairment: caspase inhibition protects dopaminergic cells against 6-hydroxydopamine-induced apoptosis but not against the loss of their terminals. J Neurochem 77: 263–273
Communal C et al. (2002) Functional consequences of caspase activation in cardiac myocytes. Proc Natl Acad Sci USA 99: 6252–6256
Birks EJ et al. (2006) Left ventricular assist device and drug therapy for the reversal of heart failure. N Engl J Med 355: 1873–1884
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
About this article
Cite this article
Haider, N., Arbustini, E., Gupta, S. et al. Concurrent upregulation of endogenous proapoptotic and antiapoptotic factors in failing human hearts. Nat Rev Cardiol 6, 250–261 (2009). https://doi.org/10.1038/ncpcardio1452
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/ncpcardio1452
This article is cited by
-
Novel XIAP mutation with early-onset Crohn’s disease complicated with acute heart failure: a case report
BMC Cardiovascular Disorders (2023)
-
Cardiac function in BRCA1/2 mutation carriers with history of breast cancer treated with anthracyclines
Breast Cancer Research and Treatment (2016)
-
Variations in the protein level of Omi/HtrA2 in the heart of aged rats may contribute to the increased susceptibility of cardiomyocytes to ischemia/reperfusion injury and cell death
AGE (2013)